Abstract
The aim of this work is to find the effect of the adding graphene oxide nanoparticle ands dodecyl sulfate (SDS) on the absorption rate and solubility of CO2 in mono-ethanolamine (MEA) solvent. To this end, an experimental setup was designed and constructed to measure the gas solubility in the solvent and the absorption rate during the process. The experiments were conducted at ambient temperature and initial pressure of 20 bar. The parameters analyzed in this paper included GO-NPS at 0.0375, 0.05, 0.075 and 0.1%, SDS at the same percentages, and the simultaneous effects of both additives. In each experiment, the absorption rate and solubility were calculated by recording the temperature and pressure variations in the absorption and loading chambers. The results indicated that the highest effect of GO-NPS was in the concentration of 0.05%, increasing solubility by up to 7.62%. These values for SDS were 0.075% and 8.39%, respectively. In the experiment with both additives at 0.05% and 0.075% for GO-NPS and SDS, solubility increased by 6.32% and simultaneous presence of both compounds had the greatest effect on absorption rate. Comparison of the results of mole variations suggested that addition of nanoparticles and surfactants both increased the absorption rate.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Numerous studies have been conducted to preserve carbon dioxide at a higher efficiency and lower energy consumption [1]. From the time, Svante August Arrhenius (1859–1927) first discussed the effects of carbon dioxide on global warming until the present time that its concentration has reached 400 ppm due to excessive release of carbon dioxide into the atmosphere [2], and is the most important reason for the effect of greenhouse gases on global warming (IPCC 2007). Due to their high reaction rate and capacity for CO2 adsorption, amines have attracted greater interest from researchers. Amines are molecules containing nitrogen atoms bound to a carbon-based chain structure.
Lemoine et al. examined solubility of carbon dioxide and hydrogen sulfide in methyl diethanolamine (MDEA) aqueous solution. They investigated carbon dioxide solubility in MDEA at 63.23 wt% and temperature of 298 K, as well as solubility of hydrogen sulfide at 63.23 wt% and 313 K [3]. Dong Fu et al. examined solubility and viscosity in the absorption process of CO2 using an MEA-enhanced DEAE aqueous solution in the temperature range of 302.2–333.2 K [4]. Aronu et al. studied solubility of CO2 at 15, 30, 45 and 60 wt% of MEA in the temperature range of 40–120 °C. They introduced a new model using the developed UNIQAC thermodynamic model [5]. Arshad et al. examined the solubility of CO2 in 30 wt% MEA and M5 DEEA solutions at temperatures of 40, 80 and 120 °C and compared the results against those obtained by other researchers [6]. In some cases, researchers used amine compounds to increase the absorption rate of CO2 and reduce costs. Hagewiesche et al. examined and compared solubility of carbon dioxide in MDEA–MEA and MDEA–DEA mixtures [7]. Adopting the chromatography method for liquid analysis, Libreros et al. examined solubility of carbon dioxide in MDEA–DEA and MDEA–AMP mixtures at 313.15, 343.13 and 393.15 K and pressures of 3–2000 kPa [8]. RédaSidi-Boumedine et al. explored solubility of carbon dioxide in MDEA–DEA mixture in the temperature range of 298–348 K and pressure range of 61.2–2239 kPa [9]. Cheng et al. examined solubility of carbon dioxide in water–MEA–TEA mixture at temperatures of 33.2, 353.2 and 373.2 K and pressure range of 1–120 kPa [10]. In addition, researches on the prediction of CO2 gas solubility in MEA have been carried out [11].
Numerous studies have explored the effect of fine particles on increasing mass transfer from the gas into the liquid phase. In fact, different conditions should be examined to increase the rate of CO2 absorption in the presence of fine particles. In 1904, the idea of using particles at nanoscale dimensions was first proposed by Maxwell [12] setting in motion a great revolution in heat transfer in fluids. In fact, he proposed a new approach to solid–liquid suspension with nano-sized particles. Masuda et al. [13] were the first to dub this fluid containing suspended particles as nano fluid. Later on, Choi [14] developed the concept considerably at the Argonne National Laboratory. Concerning the absorption of carbon dioxide gas, laboratory results suggest that some nanoparticles can significantly increase carbon dioxide absorption [1]. Furthermore, addition of nanoparticles to the solvent can increase mass transfer intensity. In 2012, Pineda et al. studied and compared the effect of SiO2 and Al2O3 nanoparticles on CO2 absorption rate in methanol-based nanofluid using a column absorbent tray. Results explained that absorption capacity of SiO2 and Al2O3 nanofluids at the optimal volume load of 0.05 wt% of nanoparticles increased to 9.4% and 9.7% compared to their base fluids, respectively [15].
Kim et al. investigated solubility of carbon dioxide in SiO2/H2O nanofluid. They used three types of nanofluids including 30, 70, and 120 nm particles. Results demonstrated that the addition of nanoparticles led to a 76% increase in the average absorption rate during the first minute as well as a 24% increase in total absorption in water. They found that CO2 absorption rate in the nanofluid was four times greater than that for water without nanoparticles [16]. Salimi et al. explored removal of carbon dioxide gas by Al2O3 nanofluids and Al2O3–SiO2 mixture in a water-based fluid in a filled absorption tower. They examined the effects of concentration and type of nanoparticle on absorption rate. Results showed that the optimum value was 0.05 volume % for both nanofluids. It was also revealed that addition of nanoparticles increased mass transfer rate. They also found that the highest increase in mass transfer rates for Al2O3 and Al2O3–SiO2 nanoparticles occurred at the 0.05 volume % for both nanofluids (14% and 10%, respectively, compared to pure water) [17].
Shahraki et al. examined solubility of carbon dioxide in MDEA/PZ in the presence of SiO2 nanoparticles at temperatures of 40 °C and 120 °C and the pressure range of 1–26 atm [18]. Jiang et al. studied and compared the effect of adding different nanoparticles, TiO2, MgO, and Al2O3·SiO2, and of the size of nanoparticles in increasing adsorption rate of CO2 in MEA and MDEA solutions. In 2012, Pineda et al. studied adsorption of CO2 gas using the methanol-based nanofluids Al2O3 and SiO2 for the tray tower. Haghtalab et al. measured and compared solubility and absorption of CO2 in water-based ZnO and SiO2 nanofluids, and examined the effect of temperature and concentration of nanoparticles on increasing absorption. They measured the solubility of CO2 at 0.05, 0.1, 0.5 and 1 wt% of ZnO nano fluid at 5 °C and the pressure range of 1–25 bar and observed that absorption rate increased at greater ZnO masses in all experiments. Examining the solubility of CO2 in pure water and 0.1 wt% of two nanofluids, they concluded that the absorption rate of CO2 increased in the presence of nanoparticles and ZnO nanofluids had a greater effect than SiO2 nanofluids in all laboratory conditions [19].
Irani et al. examined the increase in CO2 absorption in MDEA in the presence of graphene oxide. They announced that GO/MDEA nanofluid showed high absorption capacities toward CO2 due to the high surface area and existence of OH groups on the GO surface and enhancement in mass transfer coefficient [20].
In the current study, we investigated simultaneous effect of nanoparticle and surfactant on the both solubility and absorption rate of CO2 in MEA Solvent. Therefore, GO-NPs and SDS in various concentrations from zero to 0.1 g were chosen as additives. Absorption of CO2 into MEA 25% was studied in a batch system.
Experiments
Setup
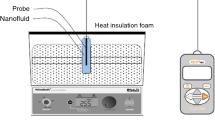
Numerous devices that differ in their measurement methods have been designed so far to measure gas solubility in a liquid (solvent). In these devices, the gas and solvent were kept in contact with each other and some gas molecules were absorbed by the solvent. This process continues until the solvent saturated with the solute (gas). The modern devices differ from the previous ones which were used to determine solubility. The pressure, temperature, nature of solvent and solute as well as the required experimental accuracy determine the type of device for solubility measurements. Most of these devices operate at a constant pressure and varying volume. Alternatively, the volume is constant and the pressure varies. The temperature of the system was kept constant throughout the experiment. This experiment involved a constant-volume device whose schematic view is shown in Fig. 1.
Schematic view of the laboratory device used in this study. 1 CO2 save capsule; 2 crank valve regulator, pressure regulator and pressure gauge; 3 loading cell; 4 bathroom circulation system for temperature and circulation; 5 needle valve; 6 absorption cell; 7 electromotor and agitator absorption cell; 8 temperature sensor; 9 pressure sensor; 10 power box, the temperature and pressure are displayed; 11 data acquisition, data transfer and registration (DAQ)
Materials
The representative SEM and AFM images of used graphene oxide have been shown below. Figure 2 shows that the graphene oxide sheet was successfully exfoliated and the size of GO-NPs is less than 2 nm (Table 1).
Description of the experiments
In this study, the experiments were conducted to measure solubility of CO2 in the MEA solvent in the presence and absence of GO-NPs and SDS. First, 25 ml of MEA solvent was poured in a 100-ml flask and the volume was raised to 100 ml by adding 75 ml of distilled water. The solution then was mixed with 0.05 g of GO-NPs and was fully blended and stabilized in an ultrasonic bath using a magnetic stirrer and a magnet for 20 min.
The pressure of the loading chamber has to reach the desired level before the experiment starts. First, CO2 is removed from the storage capsule and loaded into the chamber for the pressure in the loading chamber to be adjusted to the desired level (20 bar). Then, the solution containing 100 ml distilled water + 0.05 g of GO-NPs + MEA is injected into the absorption chamber with a syringe and the stirrer is set at 5 rpm. At the same time that the data recording software is switched on, the valve opened, and CO2 gas is discharged from the loading chamber into the absorption chamber and the absorption process is started. In this stage, since the solvent did not saturate with V and the gas is absorbed into the solvent, the pressure in the absorption chamber rapidly increases as the pressure in the loading chamber declines. The pressure drop in the loading chamber decreases with the passage of time until the solvent saturated with CO2 gas. Finally, the pressure stabilized and the absorption process ends. This final constant pressure is the equilibrium pressure. In this stage, data on the initial and final pressure and temperature of the chamber can used to calculate the number of CO2 gas moles absorbed in the solvent (details of experiment 2).
Theory
Thermodynamic calculations
Calculation of solubility in the laboratory is based on P–T data in liquid and gas equilibrium.
There are numerous methods to investigate fluid and gas equilibria, the most important of which is probably the equation of state. The present study used the Peng–Robinson equation that was proposed by Peng and Robinson in 1976 and in which the parameters of the state equation are obtained using the critical properties and the acentric factor.
In the above relations, V is the molar volume, Tc and Pc are the temperature and pressure at the critical point, respectively, R is the gas constant, and ω is the acentric factor.
a, b, α and m are the constants of the equation. T and P are the temperature and pressure of system, respectively; Tr and Pr are the reduced temperature and reduced pressure, respectively.
The Peng–Robinson equation can be formulated in terms of z-compressibility factor as follows:
A and B are the constants of the equation.
The third-order equation for the compressibility factor yields three values of Z in the two-phase zone. The highest and lowest values are associated with the gas phase and the liquid phase, respectively. The middle value is rejected.
The Z value obtained at the initial and final (equilibrium) temperature and pressure is used to calculate the number of initial and final (equilibrium) moles by employing the following equations:
where V is the volume.
where V is obtained from the following equation:
In this equation, VLC is the volume of the loading chamber and VAC is the volume of absorption cell.
The number of absorbed gas moles is obtained from the following equation:
Using the number of moles of gas absorbed, solubility in various units is obtained from the following equation:
In Eq. (7), standard volume refers to the volume (cm2) that the gas occupies at standard temperature (25 °C) and standard pressure (1 atm).
Kinetic calculations
To describe the mass transfer rate of CO2 into the liquid phase, the simple relation is used as shown below:
In Eq. (8), k is a function of mass transfer constant and t is the time (min). This simple equation is used for comparison of mass transfer rate in the experiments. The results show that by drawing (nt/n∞) versus t0.5, straight line with slope of k has been achieved. Therefore, k is calculated for each experiment. The bigger of k is according to higher mass transfer rate. By comparison, between k amounts in different experiments, the rate of absorption can be compared.
Results and discussion
At first, the experimental results of carbon dioxide solubility in MEA were examined at various concentrations of GO-NPs and SDS. Then, they compared it with the ideal solubility obtained in the modeling section. Finally, the effects of GO-NPs and SDS, as well as their simultaneous effects were compared (Table 2).
Solubility
Effect of GO-NPs
As shown in Fig. 3, addition of GO-NPs initially increases the solubility of CO2 in the solvent. After reaching the highest solubility at the optimum concentration of nanoparticles, further addition of nanoparticle decreases solubility. This trend has been reported for various nanoparticles in similar studies by Keshishian [21] and Jiang et al. [1]. As soon as the gas is absorbed in the nanofluid, the stable nanoparticles break the gas bubbles into smaller ones thus expanding the mass transfer zone. In fact, the solubility of small gas bubbles increases and this, ultimately, leads to an increase in total adsorption in the nanofluids [19, 22, 23]. Furthermore, it can be argued that mass transfer intensifies by increases in GO-NPs according to the effect of boundary mixture. In fact, GO-NPs that are around the bubbles prevent them from aggregating, and thus increase the surface for gas–liquid mass transfer. Nonetheless, since the boundary zone between liquid and gas is limited, when solid particles are loaded excessively, the adjacent particles may appear as a single plane and prevent mass transfer [24].
Effect of surfactant presence
According to Fig. 4, it can be argued that addition of SDS can reduce the surface tension of the solvent; i.e., accumulation at the interface of immiscible fluids can reduce surface tensions, and decrease resistance to mass transfer on the solvent surface to facilitate entry of CO2 and improve the kinetic behavior and increase the solubility of CO2 in the solvent. As with GO-NPs, addition of SDS initially increases the solubility of CO2 in the solvent. As the highest solubility is achieved at optimal SDS concentration, further addition of SDS decreases solubility. It can be said that due to the increased viscosity of the solution, the solubility decreases.
Effect of simultaneous presence of GO-NPs and SDS
According to Fig. 5a, it can be seen that addition of 0.075 g of SDS to 25% MEA solvent increases solubility by 8.39%. Addition of 0.05 g of GO-NPs to 25% MEA solvent increases solubility by 7.62%, and simultaneous addition of 0.075 g of SDS and 0.05 g of GO to 25% MEA solvent increases solubility by 6.31% compared to their base fluid concentrations.
As can be shown in Fig. 4b, mass transfer constant increased by adding GO and SDS. However, in simultaneously presence of two additives experiment, higher mass transfer constant has been achieved.
Absorption rate
Effect of GO-NPs on mass transfer coefficient
Addition of GO-NPs improves the kinetic behavior of absorption and, as shown in Fig. 6, the equilibrium is achieved sooner as the nanoparticle concentration increases from 0 to 0.1 g.
According to Fig. 7, it can be seen that adding GO to MEA caused increase in the mass transfer rate of about 35.8%.
Effect of surfactant concentration on mass transfer coefficient
Addition of SDS improves the kinetic behavior of absorption. As shown in Fig. 8, the equilibrium is achieved sooner as the concentration of nanoparticles increases from 0 to 0.1 g.
It can argued that addition of SDS can reduce the surface tension of the solvent; i.e., accumulation at the interface of immiscible fluids can reduce surface tensions, and decrease resistance to mass transfer on the solvent surface to facilitate entry of CO2 and improve the kinetic behavior and increase the solubility of CO2 in the solvent. Figure 9 shows that 38.9% increase has occurred by adding SDS to MEA.
At the last part of experiments, GO and SDS were added to MEA simultaneously. The mixture was prepared by adding 0.05% GO + 0.075% SDS. These concentrations were chosen based on the maximum solubility of each additive. The results of these experiments have been compared with GO and SDS in Fig. 10. This figure shows that using additives including GO nanoparticle and SDS can improve the mass transfer rate. And equilibrium time decreased when two additives are used simultaneously. However, the highest absorption rate at each instant was observed in simultaneous addition of 0.075 g of SDS and 0.05 g of GO to 25% MEA solvent.
According to the achieved results, we can say that by investigation of solubility and kinetic behavior of absorption, the better conclusion will be gained. Since in the absorption of gas in the solvent, both parameters, solubility and mass transfer rate have important roles in the performance of the system.
Conclusions
Graphene oxide nanoparticles and SDS adding effects on the solubility and absorption rate of CO2 in MEA was experimentally studied.
The results show that increases in the concentration of solvent MEA up to a certain level increased CO2 gas solubility, but beyond that lowered solubility of carbon dioxide gas. At 50 wt% concentration of solvent MEA, the solubility of CO2 increased by 30.97% compared to solubility of CO2 at 25 wt% concentration of MEA.
Addition of GO-NPs improved the kinetic behavior of absorption. In fact, the solubility of CO2 at 25% MEA solvent increased by 7.62% through addition of 0.05 g of GO-NPS.
Addition of SDS improved the kinetic behavior of absorption. In fact, the solubility of CO2 at 25% MEA solvent increased by 8.39% through addition of 0.075 g of SDS.
The highest absorption rate at each instant was observed in simultaneous addition of 0.075 g of SDS and 0.05 g of GO to 25% MEA solvent. The highest solubility was achieved by adding 0.075 g of SDS. According to these results, adding GO and SDS in a proper concentration can improve the kinetics and solubility of CO2 in MEA solvent.
References
Jiang J, Zhao B, Zhuo Y, Wang S (2014) Experimental study of CO2 absorption in aqueous MEA and MDEA solutions enhanced by nanoparticles. Int J Greenhouse Gas Control 29:135–141
Althuluth M, Mota-Martinez MT, Kroon MC, Peters CJ (2012) Solubility of carbon dioxide in the ionic liquid 1-ethyl-3-methylimidazolium tris(pentafluoroethyl)trifluorophosphate. J Chem Eng Data 57:3422–3425
Lemoine B, Li Y-G, Cadours R, Bouallou C, Richon D (2000) Partial vapor pressure of CO2 and H2S over aqueous methyldiethanolamine solutions. Fluid Phase Equilibria 172:261–277
Fu D, Wang L, Zhang P, Mi C (2016) Solubility and viscosity for CO2 capture process using MEA promoted DEAE aqueous solution. J Chem Thermodyn 95:136–141
Aronu UE, Gondal S, Hessen ET, Haug-Warberg T, Hartono A, Hoff KA (2011) Solubility of CO2 in 15, 30, 45 and 60 mass% MEA from 40 to 120 C and model representation using the extended UNIQUAC framework. Chem Eng Sci 66:6393–6406
Arshad MW, von Fosbol P, Solms LN, Svendsen HF, Thomsen K (2014) Equilibrium Solubility of CO2 in Alkanolamines. Energy Proc 51:217–223
Hagewiesche DP, Ashour SS, Al-Ghawas HA, Sandall OC (1995) Absorption of carbon dioxide into aqueous blends of monoethanolamine and N-methyldiethanolamine. Chem Eng Sci 50:1071–1079
Libreros MER, Trejo A (2004) Gas solubility of CO2 in aqueous solutions of N-methyldiethanolamine and diethanolamine with 2-amino-2-methyl-1-propanol. J Fluid Phase Equilib 218:261–267
Boumedine RS, Horstmann S, Fischer K, Provost E, Fürst W, Gmehling J (2004) Experimental determination of carbon dioxide solubility data in aqueous alkanolamine solutions. J Fluid Phase Equilib 218:85–94
Cheng MD, Caparanga AR, Soriano AN Hui, Li M (2010) Solubility of CO2 in the solvent system (water + monoethanolamine + triethanolamine). J Chem Thermodyn 42:342–347
Zhang Z, Lic H, Chang H, Pan Z, Luo X (2018) Machine learning predictive framework for CO2 thermodynamic properties in solution. J CO2 Utilization 26:496–502
Maxwell JC (1904) A treatise on electricity and magnetism. Claredon, Oxford
Murshed S, Leong K, Yang C (2008) Investigations of thermal conductivity and viscosity of nanofluids. Int J Therm Sci 47(5):560–568
Chol S (1995) Enhancing thermal conductivity of fluids with nanoparticles. ASME-Publications-Fed 231:99–106
Torres Pineda I, Lee JW, Jung I, Kang YT (2012) CO2 absorption enhancement by methanol-based Al2O3 and SiO2 nanofluids in a tray column absorber. Int J Refrig 35:1402–1409
Kim W, Kang HU, Jung K, Kim SH (2008) Synthesis of silica nanofluid and application to CO2 absorption. J Separation Sci Technol 43(11):3036–3055
Salimi J, Salimi F (2016) CO2 capture by water-based Al2O3-SiO2 mixture nanofluids in an absorbtion packed column. Revista Mexicana de Ingeniería Química 15:185–192
Shahraki H, Sadeghi J, Shahraki F, Kalhori DM (2016) Investigation on CO2 solubility in aqueous amine solution of MDEA/PZ with SiO2 nanoparticles additive as novel solvent. J Chem Eng Process Technol 7:37–43
Haghtalab A, Mohammadi M, Fakhroueian Z (2015) Absorption and solubility measurement of CO2 in water-based ZnO and SiO2 nanofluids. Fluid Phase Equilib 392:33–42
Irani V, Maleki A, Tavasoli A (2018) CO2 absorption enhancement in graphene-oxide/MDEA nanofluid. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2018
Keshishian N, Nasr Esfahany M, Etesami N (2013) Experimental investigation of mass transfer of active ions in silica nanofluids. Int. Commun Heat Mass 46:148–153
W-g Kim, Kang HU, K-m Jung, Kim SH (2008) Synthesis of silica nanofluid and application to CO2 absorption. Sep Sci Technol 43(30):36–55
Jung J-Y, Lee JW, Kang YT (2012) CO2 absorption characteristics of nanoparticle suspensions in methanol. J Mech Sci Technol 26(22):85–90
Karve S, Juvekar VA (1990) Gas absorption into slurries containing fine catalyst particles” Chem. Eng Sci 45(3):569–772
Acknowledgements
This paper was prepared from a Ph. D. thesis conducted in the Department of Chemical Engineering, Mahshahr branch, Islamic Azad University, Mahshahr, Iran.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Mohammadpour, A., Mirzaei, M., Azimi, A. et al. Solubility and absorption rate of CO2 in MEA in the presence of graphene oxide nanoparticle and sodium dodecyl sulfate. Int J Ind Chem 10, 205–212 (2019). https://doi.org/10.1007/s40090-019-0184-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40090-019-0184-5