Abstract
Chemical flooding is of increasing interest and importance due to high oil prices and the need to increase oil production. Research in nanotechnology in the petroleum industry is advancing rapidly, and an enormous progress in the application of nanotechnology in this area is to be expected. The nanotechnology has been widely used in several other industries, and the interest in the oil industry is increasing. Nanotechnology has the potential to profoundly change enhanced oil recovery and to improve mechanism of recovery, and it is chosen as an alternative method to unlock the remaining oil resources and applied as a new enhanced oil recovery method in last decade. This paper therefore focuses on the reviews of the application of nanotechnology in chemical flooding process in oil recovery and reviews the applications of nanomaterials for improving oil recovery that have been proposed to explain oil displacement by polymer flooding within oil reservoirs, and also this paper highlights the research advances of polymer in oil recovery. Nanochemical flooding is an immature method from an application point of view.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the past decade, rapid development in nanotechnology has produced several aspects for the scientists and technologists to look into [1]. Nanoparticles are particles with dimensions in the order of 1–100 nm (nanometer). They possess unique properties due to their small sizes and greater surface area per unit volume [2]. Various nanofluids can be designed by the addition of nanoparticles to different base fluids. The stability or dispersion of nanoparticles in solutions relies on the functionality (or surface activity) of the nanoparticles. To this end, the surfaces of nanoparticles are usually treated or functionalized, thereby putting shields around them. These shields hinder particle–particle interaction, and the possibility of nanoparticles aggregation is reduced. Nanotechnology in the petroleum industry has gained enormous interest in the recent years, which is reflected in the amount of literature available. Nanoparticles for EOR purposes seem gradually to become the cutting-edge technology [3].
Reservoir engineering, however, has received the most attention for nanotechnology applications. Several experiments have been conducted to investigate flow behavior of nanoparticle suspensions through porous media. Rodriguez et al. [4] investigated the migration of concentrated surface-treated silica nanoparticles in sedimentary rocks. Kanj et al. [5] identified the usable size of nanoparticles in reservoir rocks through nanofluid core flooding experiments. Using hydrophilic/hydrophobic synthesized nanoparticles, Zhang [6] made oil in water emulsions and stabilized CO2 foams with quite high stability.
The nanotechnology has been widely used in several other industries, and the interest in the oil industry is increasing. Silica nanoparticles are the most widely tested and have shown good EOR applications. Recently, studies have explored the potential of Al2O3, MgO and Fe2O3 in addition to SiO2 nanoparticles. The results showed that some combinations have yielded better results than SiO2. Based on the current knowledge, it is expected that both chemical EOR and specifically micellar flooding will make huge benefits from nanotechnology and nanoemulsion in particular in the future [7]. Adding nanoparticles to fluids may significantly benefit enhanced oil recovery and improve well drilling, such as changing the properties of the fluid, wettability alternation of rocks, advanced drag reduction, strengthening sand consolidation, reducing the interfacial tension and increasing the mobility of the capillary-trapped oil [8].
In the past decade, most investigations have shown that nanoparticles (NPs) offer promise for future enhanced oil recovery (EOR) processes where silica-based NPs have been most commonly used [9–13]. Although the oil displacement mechanism with NPs is not yet clearly understood [14–16], the nanotechnology is now chosen as an alternative method to unlock the remaining oil resources and applied as a new enhanced oil recovery (EOR) method in the last decade [10–12, 17–20]. Polymeric microspheres and nanospheres have been applied as water mobility control both as a pilot and full field and showed fantastic results in reducing water cut, increasing sweep efficiency and improving oil recovery [21–23]. They reported that polymeric microspheres and nanospheres can swell when meet with water and then reduce water permeability due to its ability to reduce the capillary force and change water flow path. Consequently water goes into bypassed area and enhances displacement efficiency. Tian et al. also reported that polymeric microspheres and nanospheres have some advantages such as no degradation at high temperature and salinity. Some papers also address experiments where combinations of nanoparticles and surfactant solutions are tested. Le et al. studied synergistic blends of SiO2 nanoparticles and surfactants for EOR in high-temperature reservoirs.
Hamedi Shokrlu and Babadagli investigated the effects on nanometals on viscosity reduction in heavy oil and bitumen for thermal oil recovery applications. Based on their obtained results, various parameters such as nanoparticle types, size and concentration could affect on the mechanism of the viscosity reduction in heavy oil/bitumen. Moreover, they figured out the optimum values of the addressed parameters in their studies [24]. In addition to previous applications of nanotechnology in petroleum upstreams, enormous researches have been made on the title of implementing of nanoparticles on the enhanced oil recovery (EOR) from petroleum reservoirs [25]. Hence, in some studies the applications of nanoparticles in oil industry have been reported and classified based on priority. Researchers have concluded that nanotechnology has the greatest usage in chemical EOR methods [26]. Therefore, in some studies, the role of nanoparticles in EOR operations has been reported [27–29]. The nanofluids are created by the addition of nanoparticles to fluids for intensification and improvement of some properties at low volume concentration of the dispersing medium. Then, the main feature of nanofluids is that their properties greatly depend on the dimensions of nanoparticles added to them [30].
The objective of this paper is to investigate the potential of nanoparticles in chemical flooding for oil recovery. This study reviewed and assessed some of the recent advances.
Enhanced oil recovery
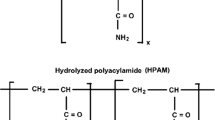
In enhanced oil recovery, a dilute aqueous solution of partially hydrolyzed polyacrylamide is used as a pushing fluid in the injection wells to sweep oil in the reservoir into the production well. Mobility reduction or viscosity behavior of partially hydrolyzed polyacrylamide polyelectrolyte solutions plays an important role in such applications. The properties of polyelectrolytes in aqueous solutions have been widely investigated in the past decade [31–39]. Oil production has three different stages: primary (production by natural reservoir energy), secondary (on the supply of external energy into the reservoir in the form of injecting fluids to increase reservoir pressure) and tertiary production (enhanced oil recovery methods increase the mobility of the oil in order to increase production). Over the lifetime of the oil field, the pressure will fall, and at some point, there will be insufficient underground pressure to lift the oil to the surface. In order to maintain the reservoir pressure and sweep out oil in a more efficient way, water flooding as secondary oil production is a practical and effective way for many reservoir formations. A problem with water flooding is that in many cases oil has higher viscosity than water, and this, however, may lead to an unstable displacement. Increasing the viscosity of injected water by adding a polymer will improve sweep during water flooding process. It is necessary to study and evaluate the rheological properties of the polymers before implementation as polymer flooding [40].
Nano- and chemical flooding
Enhanced oil recovery (EOR) processes
The ultimate goal of EOR processes is to increase the overall oil displacement efficiency, which is a function of microscopic and macroscopic displacement efficiency. Based on the overall materials balance of the reservoir, the overall oil recovery efficiency (E ro) can be defined as:
where N is the original oil in place and Np is the accumulative oil recovered after recovery process. The overall efficiency consists of microscopic or displacement efficiency (E do) and macroscopic or volumetric sweep efficiency (E vo) as given in equation below:
where E a is the areal displacement efficiency and E v is the vertical displacement efficiency.
Microscopic or displacement efficiency refers to the displacement or mobilization of oil at the pore scale and measures the effectiveness of the displacing fluid in moving the oil at those places in the rock when the displacing fluid contacts the oil [41]. It is the ratio of the amount of oil recovered to the oil initially present in the swept volume and can be expressed in terms of saturation as:
where S oi is the initial oil saturation and S or is the residual oil saturation after oil recovery process.
Capillary forces mechanism
The capillary forces have great influence on oil recovery efficiency, but the influence differs fundamentally for non-fractured and fractured reservoirs. In a non-fractured reservoir, strong capillary forces during water flooding will trap oil and cause relatively high residual oil saturation. Therefore, to remobilize residual oil, there is a need to reduce the oil water IFT. While in fractured reservoirs, spontaneous imbibition of water due to strong capillary forces is an important and necessary mechanism to attain high displacement efficiency [42]. Generally, capillary forces are overcome by either viscous or gravity forces. The two dimensionless numbers that are used to compare these forces include capillary number (N c) and bond number (N b).
The capillary number (N c) is a ratio of viscous to capillary forces and is given as:
where v is the brine velocity (Darcy’s velocity), µ w is the brine viscosity, σ ow is the oil/water interfacial tension, and θ is the contact angle between the wetting phase and the rock [43].
Flooding
Nanoparticles along with surfactants cause releasing trapped oil in pores and throats. This happens by many factors such as reducing IFT between oil and water, spontaneous emulsion formation, the wettability alteration of porous media and the modification of flow character, which ultimately increase oil recovery significantly.
Results of Mohajeri show that the presence of nanoparticles in surfactant increases oil recovery significantly. This enhancement generally for C12TAB/ZrO2 is greater than SDS/ZrO2 in terms of oil recovery. This is due to the higher activity of C12TAB as a cationic surfactant than SDS as an anionic surfactant [44]. The presence of nanoparticles changes rheological properties and increases the effect of surfactant solution on oil recovery processes. The observed reduction in interfacial tension and wettability is the result of nanoparticle present at the interfacial layers. As a result, using nanoparticles shows a 19 % increase in oil recovery when compared to surfactants used alone [45].
Wanga et al. carried out simulation experiments to study the effects of HPAM and Na–montmorillonite (Na–Mt) particles on emulsion stability (Fig. 1). The oil–water interfacial properties, such as the zeta potentials, interfacial tension and dilational viscoelasticity, were also studied. As a result, the addition of Na–Mt particles and HPAM to simulated polymer flooding water showed that with the increase in Na–Mt particle concentration (<200 mg/L), the Na–Mt particles could cause the zeta potential of the oil droplets to decrease rapidly, the interfacial tension (IFT) to decline slightly, the dilational viscoelasticity to increase and stable oil–mineral aggregates (OMA) structures to appear. It is noticeable that the polymer flooding produced water was stable at this stage and therefore difficult to treat. When the Na–Mt particle concentration was more than 250 mg/L, the zeta potential of the oil droplets was stable, while the interfacial tension and dilational viscoelasticity showed a slight increase. It could be seen that the system formed a large oil–mineral aggregate structure, so the oil droplets were apt to form aggregates which would enhance the treatment of polymer flooding produced water [46].
Adsorption of the Na–Mt particles on oil droplets a 250 mg/L Na–Mt, b 300 mg/L Na–Mt [46]
A comprehensive laboratory study on hydrophilic silica dioxide nanoparticles (NPs) for EOR purposes was performed at different reservoir temperatures and different reservoir wettabilities (water-, intermediate- and oil-wet systems). Relationships between the temperature, initial wettability and additional oil recovery and the nanofluid flooding (nano-EOR) process were investigated. The pore blocking leads to differential pressure increases during nanoflooding. Hence, providing a stable nanofluid for a longer duration and at a higher temperature to maintain a nanometer size is interesting and challenging to avoid early particle aggregation and would be more favorable for field application. Overall, these results reveal a potential way to use silica-based nanofluid for enhanced oil recovery purposes for a wide range of reservoir wettabilities at a particular reservoir temperature [44].
Lipophobic and hydrophilic polysilicon nanoparticles (LHP) can be adsorbed on pore walls and can give rise to blockage of pore throats, leading to change in wettability of sandrock surfaces. The mechanism for enhanced oil recovery consists of change of wettability of reservoir rock from hydrophobic to hydrophilic under the influence of adsorbed lipophobic and hydrophilic polysilicon. Oil recovery can be enhanced obviously by flooding with lipophobic and hydrophilic polysilicon. Figure 2 plots the recovery curves for five runs lipophobic, and hydrophilic polysilicon concentration of 2.0–3.0 % by volume is suggested for improving oil recovery. This can be seen in Fig. 2 [48].
Recoveries flooded by LHP solution with different concentrations of LHP at different injection PV [48]
Flooding experiments revealed that nanoparticles can efficiently enhance surfactant flooding, and greater additional oil recovery can be obtained by the inclusion of nanoparticles into surfactant solutions. The injection scenario also affected the efficiency of similar agents with equal surfactant and nanoparticle concentrations. The improvement in surfactant flooding efficiency was more considerable for slightly hydrophobic nanoparticle-augmented surfactant solutions with respect to hydrophilic ones [49].
A novel copolymer containing nano-SiO2 was synthesized by free radical polymerization using AM, AA and nano-SiO2 functional monomer (NSFM) as raw materials. The AM/AA/NSFM copolymer was characterized by IR spectrum, 1H NMR spectrum, elemental analysis and scanning electron microscope. The solution properties, such as rheological property, viscoelasticity, temperature tolerance, salt tolerance, mobility control ability and oil displacement efficiency of the copolymer, were investigated under different conditions. The results indicated that the copolymer containing nano-SiO2 possessed moderate or good shear resistance, temperature tolerance and mobility control ability as EOR chemical. The synthesis of AM/AA/NSFM is shown in Figs. 3 and 4 [50, 51].
The synthesis of AM/AA/NSFM [51]
SEM images of a AM/AA and b AM/AA/NSFM [51]
Results of Hendraningrat study show that polymeric nanospheres can enhance residual oil recovery in the rock samples, and its concentration showed a significant impact into incremental oil. By evaluating the contact angle, it was observed that wettability alteration also might be involved in the oil displacement mechanism in this process together with reduction in mobility ratio and permeability to water that might divert injected fluid into unswept areas (bypassed oil) and enhance the oil recovery. These investigations promote polymeric nanospheres suspension as a potential displacement fluid in EOR. Further study is required, for instance, stability tests, repeat coreflood testing by increasing nanospheres concentration, and various parameter effects to EOR process such as initial wettability of rock, temperature, different salinity and salt compositions [47].
Viscosity of nanosuspension considerably increases with nanoparticle weight fraction. More particles in solution have direct effect on fluid shear stress. The best sample is around 0.9 wt% nanoclay mix with 3150 ppm hydrolyzed polyacrylamide. Cheraghian et al. indicated ultimate oil recovery by nanoclay polymer flooding enhances by a factor of 5 % in comparison with polymer flooding. SEM images of samples (Fig. 5) depict the typical chains of polymer nanoparticles due to significant interaction among nanosized particles and the presence of micro- and nanosized grains/nanoparticles rock [52].
Scanning electron microscopy (SEM) images of sand samples of a 0.45 wt%, b 0.90 wt%, clay containing nanocomposites after polymer flooding process [52]
Hydrophilic silica nanoparticles (Fig. 6) have through experimental work showed to propagate through a porous media. The particles caused permeability impairment, which was highly dependent on varying parameters, especially concentration. Both permeability reduction experiments and SEM analysis showed similar results, where both indicated entrainment and adsorption onto rock surfaces. Large concentrations yielded too high impairment, and a nanoparticle concentration should not exceed 0.1 wt%. The silica nanoparticles had various effects on enhancing oil recovery. The nanoparticle solution utilized did not mobilize sufficient oil when applied as a tertiary recovery method, where it only increased recovery with 0–1 % above secondary baseline. Interesting results were, however, obtained when nanoparticle solution was used as a secondary recovery method. Here, a significant decrease in residual oil saturation varying from 2 to 13 % was observed for the three floods, and an increase in recovery factor varying from 0 to 8 % was obtained. The potential for hydrophilic silica nanoparticles as an EOR-agent exists, but it is clear that water-wet reservoirs are not the best target area. Applying hydrophilic silica nanoparticles in a different wetting system will possibly show a much more promising result [53].
SEM picture of Aerosil 300 nanoparticles on clean surface, magnified by a 40,000 and b 80,000 [53]
Oil recovery was improved after the application of nanosilica fluid in core samples. However, these nanoparticles are more efficient in light oil reservoirs [11]. Flooding experiments revealed that nanoparticles can efficiently enhance surfactant flooding, and greater additional oil recovery (see also Table 1) can be obtained by the inclusion of nanoparticles into surfactant solutions [49].
A systematic study of hydrophilic metal oxide nanoparticles (NPs) for EOR purposes was performed, and the stability of metal oxide-based nanofluids was successfully improved by adding polyvinylpyrrolidone at 1 wt%. Hydrophilic metal oxide nanoparticles listed in Table 2 were screened. The combinations of metal oxide nanofluids and polyvinylpyrrolidone improved oil recovery to a greater extent than silica-based nanofluid and dispersant alone, and the TiO2-based nanofluid was especially effective for all wettability systems. The additional oil recovery (NF) of metal oxide-based nanofluids decreased as the wettability changed from water- to intermediate- and oil-wet systems. Therefore, wettability alteration plays a dominant role in the oil displacement mechanism using nano-EOR. Finding a suitable stabiliser for each nanofluid and its optimum concentration might be important in future work as well as repeating the experiment with representative formation water at reservoir conditions [47].
Nanosized silica particles propagate through water-wet Berea sandstone cores. Silica nanosized particles did not mobilize oil in water-wet Berea sandstone cores. When silica particles were dispersed in a 600 ppm hydrolyzed polyacrylamide (HPAM) solution, more than 20 % reduction in S or was observed [27]. Aluminum oxide nanoparticles that are dispersed in propanol have more tendencies to enhance oil recovery than iron oxide nanoparticles. However, both of them are applicable. Also silicon oxide treated by silane that is dispersed in propanol has the most tendencies to enhance oil recovery through the interfacial tension reduction and wettability alterations. Total oil recoveries by the nanofluids are more 88 %. It indicates that if nanofluid injection begins earlier, the oil recoveries will be higher and less amount of oil is trapped within the porous media [54].
It seems adding 0.1 wt% nanoparticles can improve pseudoplasticity behavior of polymer solutions in low shear rate values. In all tests, an increase in viscosity of nanosuspension in comparison with polymer viscosity was observed. This increase can be a reason for increasing oil recovery during flooding test by nanosuspension in comparison with polymer solution flooding. It means nanosuspension can be used instead of polymer solution to enhance oil recovery because they could increase viscosity of polymer solution. Possibility of improving pseudoplasticity behavior of polymer solutions after adding high percentage of nanoparticles can be evaluated in the future [55–57].
Effects of nanoparticles on oil recovery were investigated in Rahimi et al.’s works by using different concentrations of SDS surfactant solution and water-wet nanoparticles in a sandstone core plug. Ultimate oil recovery is increased by injecting surfactant solution enriched by Aerosil 200 through a sandstone core. The amount of additional oil recovery for a surfactant solution with 2500 ppm SDS concentration and 1000 ppm Aerosil 200 concentration was 11 %. This 11 % additional recovered oil can lead to significant increase in oil production if reservoir volume is taken into calculation. Increased oil recovery may be attributed to the contribution of the surfactant molecules, which was supposed previously to be adsorbed on the rock surface, in the interfacial tension reduction process and prevention of the changing rock wettability from water wet to oil wet [58, 59].
Conclusion
Chemical EOR methods have made a relatively small contribution to the world’s oil production during the last decades. China is the country with the largest oil production coming from chemical EOR projects. However, there are an increasing number of ongoing and planned SP and ASP evaluations at pilot scale, especially in Canada and the USA. Polymer flooding is gaining interest for heavy crude oil reservoirs (i.e., Canada) and offshore fields. However, chemical EOR is not expected to impact world’s oil production for at least two decades, if it is ever implemented at commercial scales. Despite the growing research interest on chemically assisted methods (e.g., spontaneous imbibition, wettability modifiers and ITF reductions) and surfactant polymer (SP) flooding to improve oil recoveries in carbonate formations, these projects are not expected to impact global oil production in the near future [60].
Nanotechnology has the potential to have a positive effect on the chemical EOR process. The aims of this paper were firstly to compile an up-to-date database for implemented nanochemical injection projects reported in the literature over the last 15 years. Altogether, nanotechnology can be an effective enhancement option for an oil recovery method in an oil reservoir which is technically sensitive to the chemical recovery method. Although the future of nanotechnology is completely uncharted territory, certainly nanotechnology will revolutionize the oil industry in several important ways.
References
Mishra, P.C., Mukherjee, S., Kumar Nayak, S., Panda, A.: A brief review on viscosity of nano fluids. J. Int. Nano. Lett. 4, 109–120 (2014)
Singh, S., Ahmed, R.: Vital role of nanopolymers in drilling and stimulations fluid applications. Paper SPE 130413 presented at the SPE annual technical conference and exhibition, Florence, Italy (2010)
Engeset, B.: The potential of hydrophilic silica nanoparticles for EOR purposes: a literature review and an experimental study. Norway: Department of Petroleum Engineering and Applied Geophysics, Norwegian University of Science and Technology, Trondheim, Master thesis (2012)
Rodriguez, E., Roberts, M.R., Yu, H., Huh, H., Bryant, S.L.: Enhanced Migration of Surface-Treated Nanoparticles in Sedimentary Rocks. SPE Annual Technical Conference and Exhibition, New Orleans, Louisiana (2009)
Kanj, M.Y., Funk, J.J., Al-Yousif, Z.: Nanofluid Coreflood Experiments in the ARAB-D. SPE Saudi Arabia Section Technical Symposium and Exhibition, AlKhobar (2009)
Zhang, T.: Engineered Nanoparticles as Harsh-Condition Emulsion and Foam Stabilizers and as Novel Sensors. Offshore Technology Conference, Houston, Texas (2011)
Mokhatab, S., Fresky, M.A., Islam, M.R.: Application of Nanotechnology in Oil and Gas E&P. JPT, Society of Petroleum Engineers (2006)
Cheraghian, G., Khalili Nezhad, S., Kamari, M., Hemmati, M., Masihi, M., Bazgir, S.: Adsorption polymer on reservoir rock and role of the nanoparticles clay and SiO2. Int. Nano. Lett (2014). doi:10.1007/s40089-014-0114-7
Torsater, O., Li, S., Hendraningrat, L.: Effect of Some Parameters Influencing Enhanced Oil Recovery Process using Silica Nanoparticles: An Experimental Investigation. SPE Reservoir Characterization and Simulation Conference and Exhibition, Abu Dhabi, UAE (2006). doi: 10.2118/165955-MS
Miranda, C.R., De Lara, L.S., Tonetto, B.C.: Stability and mobility of functionalized silica nanoparticles for enhanced oil recovery application. Soc. Pet. Eng. SPE 157033 (2012)
Roustaei, A., Saffarzadeh, S., Mohammadi, M.: An evaluation of modified silica nanoparticles, efficiency in enhancing oil recovery of light and intermediate oil reservoirs. Egypt. J. Petrol 22, 427–433 (2013)
Cheraghian, G.: Application of Nano Fumed Silica in heavy oil recovery. Petrol. Sci. Technol. (2015). doi:10.1080/10916466.2015.1114497
Hendraningrat, L., Li, S., Torsæter, O.: Enhancing oil recovery of low-permeability berea sandstone through optimised nanofluids concentration. In: SPE enhanced oil recovery conference, Kuala Lumpur, Malaysia (2013)
Wasan, D.T., Nikolov, A.: Spreading of nanofluids on solids. Nature 423, 156–159 (2003)
Wasan, D.T., Nikolov, A., Kondiparty, K.: The wetting and spreading of nanofluids on solids: role of the structural disjoining pressure. Curr. Opin. Colloid Interface Sci. 16, 344–349 (2011)
Chengara, A., Nikolov, A., Wasan, D.T., Trokhymchuck, A., Henderson, D.: Spreading of nanofluids driven by the structural disjoining pressure gradient. J. Colloid Interface Sci. 280, 192–201 (2004)
Cheraghian, G., Khalili Nezhad, S.: Experimental Investigation of Polymer Solutions Used in Enhanced Oil Recovery: Thermal properties Improved by Nanoclay. 77th EAGE Conference and Exhibition (2015). doi:10.3997/2214-4609.201412504
Ogolo, N.A., Olafuyi, O.A., Onyekonwu, M.O.: Enhanced oil recovery using nanoparticles. Soc. Pet. Eng. SPE 160847 (2012)
Hendraningrat, L., Torsæter, O.: Metal oxide-based nanoparticles: revealing their possibility to enhance the oil recovery at different wettability systems. Appl NanoSci. Springer. (2014). doi:10.1007/s13204-014-0305-6
Hendraningrat, L., Torsæter, O.: Effects of the initial rock wettability on silica-based nanofluid-enhanced oil recovery processes at reservoir temperatures. Energy Fuels 28(10), 6228–6241 (2014)
Yuan, W., Liu, X., Wei, H., Liu, J., Yang, H., Hu, S., Li, Y., Wang, D.: Research and application effect of polymeric microsphere in Wen-10 of Sinopec Zhongyuan Oil field. Inner. Mong. Petrochem. 12, 122 (2010)
Li, X., Ying, Z., Jia, Y., Liu, X., Yang, T., Ma, L.: Application of nanosphere deep profile control and displacement technology in Chanqing oilfield. Oilfield Chem. 29(4), 419–422 (2012)
Tian, Y., Wang, L., Tang, Y., Liu, C., Ma, C., Wang, T.: Research and application of nano polymer microspheres diversion technique of deep fluid. Soc. Pet. Eng. SPE 156999 (2012)
Hamedi Shokrlu, Y., Babadagli, T.: Effects of nano sized metals on viscosity reduction of heavy oil/bitumen during thermal applications. Canadian Unconventional Resources & International Petroleum Conference Held in Calgary, Alberta, Canada (2010)
Hendraningrat, L., Engeset, B., Suwarno, S., Torsæter, O.: Improved oil recovery by nanofluids flooding: an experimental study. In: SPE Kuwait international petroleum conference and exhibition Kuwait city, Kuwait (2012)
Pourafshary, P., Azimipour, S.S., Motamedi, P., Samet, M., Taheri, S.A., Bargozin, H., Hendi, S.S.: Priority assessment of the investment in development of nanotechnology in upstream petroleum industry. In: Proceedings of the Saudi Arabia Section Technical Symposium and Exhibition. A1Khobar, Saudi Arabia, SPE No. 126101 (2009)
Skauge, T., Hetland, S., Spildo, K., Skauge, A.: Nano-Sized Particles for EOR. SPE 129933, SPE Improved Oil Recovery Symposium, Oklahoma, USA (2010)
Cheraghian, G., Tardasti, S.: Improved Oil Recovery by the Efficiency of Nano-particle in Imbibition Mechanism. 74th EAGE Conference and Exhibition incorporating EUROPEC (2012). doi: 10.3997/2214-4609.20148913
Cheraghian, G.: Thermal resistance and application of nanoclay on polymer flooding in heavy oil recovery. Pet. Sci. Technol. (2015). doi:10.1080/10916466.2015.1075036
Amanullah, Md., Al-Tahini, A.M.: Nanotechnology: Its Significance in Smart Fluid Development for Oil and Gas Field Application. SPE No. 126102 (2009)
Jousset, S., Bellissent, H., Galin, J.C.: Polyelectrolytes of high charge density in organic solvent: synthesis and viscometric behavior. Macromolecules 31, 4520–4530 (1998)
Rinaudo, M., Milas, M., Jouon, N., Borsali, R.: On some original properties of dilute polyelectrolyte solutions at low salt content: sodium hyaluronate example. Polymer 34, 3710–3715 (1993)
Muller, K.K., Vilgis, T.A.: Dynamics of dense polyelectrolyte solutions. Macromolecules 31, 5898–5903 (1998)
Antonietti, M., Briel, A., Forster, S.: Quantitative description of the intrinsic viscosity of branched polyelectrolytes. Macromolecules 30, 2700–2704 (1997)
Leibler, L., Pezron, E.: Viscosity behaviour of polymer solutions in the presence of complexing ions. Polymer 29, 1105–1109 (1988)
Ennari, J., Elomaa, M., Sundholm, F.: Modelling a polyelectrolyte system in water to estimate the ion-conductivity. Polymer 40, 5035–5041 (1999)
Vishalakshi, B., Ghosh, S., Kalpagam, V.: The effects of charge density and concentration on the composition of polyelectrolyte complexes. Polymer 34, 3270–3275 (1993)
Dobrynin, A.V., Colby, R.H., Rubintein, M.: Scaling theory of polyelectrolyte solutions. Macromolecules 28, 1859–1871 (1995)
Zeynali, M., Rabii, A., Baharvand, H.: Synthesis of partially hydrolyzed polyacrylamide and investigation of solution properties (viscosity behaviour). Iran Polym J 13(6), 479–484 (2004)
Sorbie, K.S.: Polymer Improved Oil Recovery. Blackie and Son Ltd, Glasgow (1991)
Green, D.W., Willhite, G.P.: Enhanced Oil Recovery, SPE Textbook Series, vol. 6. Henry L. Doherty Memorial Fund of AIME, Society of Petroleum Engineers, Richardson (1998)
Udegbunam, J.E.: EOR in chalk: optimization oil recovery with modified seawater. Master Thesis. University of Stavanger (2011)
Olajire, A.A.: Review of ASP EOR (alkaline surfactant polymer enhanced oil recovery) technology in the petroleum industry: prospects and challenges. Energy (2014). doi:10.1016/j.energy.2014.09.005
Mohajeri, M., Hemmati, M., SadatShekarabi, A.: An experimental study on using a nanosurfactant in an EOR process of heavy oil in a fractured micromodel. J. Petrol. Sci. Eng. 126, 162–173 (2015)
Rosen, M.J., Wang, H., Shen, P., Zhu, Y.: Ultralow interfacial tension for enhanced oil recovery at very low surfactant concentrations. Langmuir 21, 3749–3756 (2005)
Wanga, Y., Lua, F., Lia, Y., Wub, T., Sunb, D., Zhanga, G., Huanga, X., Wang, G.: Effects of Na–montmorillonite particles on the emulsification stability of polymer flooding produced water. Colloids Surf. A: Physicochem. Eng. Aspects 410, 125–129 (2012)
Hendraningrat, L., Zhang, J.: Polymeric nanospheres as a displacement fluid in enhanced oil recovery. Appl. Nano. Sci. Springer (2015). doi:10.1007/s13204-014-0399-x
Ju, B., Fan, T., Ma, M.: Enhanced oil recovery by flooding with hydrophilic nanoparticles. China Particuol. 4(1), 41–46 (2006)
Zargartalebi, M., Kharrat, R., Barati, N.: Enhancement of surfactant flooding performance by the use of silica nanoparticles. Fuel 143, 21–27 (2015)
Kim, B.J., Kang, K.S.: Fabrication of a crack-free large area photonic crystal with colloidal silica spheres modified with vinyltriethoxysilane. Cryst. Growth Des. 12(8), 4039–4042 (2012)
Ye, Z., Qin, X., Lai, N., Peng, Q., Li, X., Li, C.: Synthesis and performance of an acrylamide copolymer containing nano-SiO2 as enhanced oil recovery chemical. J. Chem. (2013). doi:10.1155/2013/437309
Cheraghian, G., Khalili Nezhad, S.: Effect of nanoclay on heavy oil recovery during polymer flooding. Pet. Sci. Technol. (2015). doi:10.1080/10916466.2015.1014962
Engeset, B.: The Potential of Hydrophilic Silica Nanoparticles for EOR Purposes: A Literateur Review and an Experimental Study. Norway: Department of Petroleum Engineering and Applied Geophysics, Norwegian University of Science and Technology, Trondheim, Master thesis (2012)
Joonaki, E., Ghanaatian, S.: The application of nanofluids for enhanced oil recovery: effects on interfacial tension and coreflooding process. Pet. Sci. Technol. 32(21), 2599–2607 (2014)
Maghzi, A., Mohebbi, A., Kharrat, R., Ghazanfari, M.H.: An experimental investigation of silica nanoparticles effect on the rheological behavior of polyacrylamide solution to enhance heavy oil recovery. Pet. Sci. Technol. 31(5), 500–508 (2013)
Cheraghian, G., Hemmati, M., Bazgir, S.: Application of TiO2 and fumed silica nanoparticles and improve the performance of drilling fluids. AIP Conf. Proc. London, Canada. 1590, 266 (2014). doi:10.1063/1.4870228
Cheraghian, G., KhaliliNezhad, S., Kamari, M., Hemmati, M., Masihi, M., Bazgir, S.: Effect of nanoclay on improved rheology properties of polyacrylamide solutions used in enhanced oil recovery. J. Petrol. Explor. Prod. Technol. (2014). doi:10.1007/s13202-014-0125-y
Rahimi, Kh, Adibifard, M.: Experimental study of the nanoparticles effect on surfactant absorption and oil recovery in one of the Iranian oil reservoirs. Petrol. Sci. Tech. 33(1), 79–85 (2015)
Cheraghian, G.: An experimental study of a surfactant polymer for enhanced heavy-oil recovery using a glass micromodel by adding nanoclay. Pet. Sci. Technol. 33(13–14), 1410–1417 (2015)
Alvarado, V., Manrique, E.: Enhanced oil recovery: an update review. Energies 3, 1529–1575 (2010)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Cheraghian, G., Hendraningrat, L. A review on applications of nanotechnology in the enhanced oil recovery part B: effects of nanoparticles on flooding. Int Nano Lett 6, 1–10 (2016). https://doi.org/10.1007/s40089-015-0170-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40089-015-0170-7