Abstract
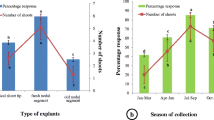
The current study evaluated for the first time plant regeneration in P. chaba. Adventitious shoot regeneration was achieved in Murashige and Skoog (MS) medium supplemented with three concentrations of 6-benzyladenine (BA; 0.25, 0.5 and 1 mg/l) and 40 mg/l ascorbic acid. Maximum number of 18 adventitious shoot buds was formed in 0.5 mg/l BA but it was accompanied by severe callusing. Therefore, for subsequent multiplication, shoots were subcultured in MS with 0.25 mg/l BA to prevent excessive callusing, though the number of shoots per culture was reduced to 12. A major problem with cultures of this plant was the excessive browning of the shoots and medium due to phenolic exudation. To control browning of tissue and medium two anti-browning agents, ascorbic acid (AA, 10, 40 and 100 mg/l) and activated charcoal (AC, 5 and 10 mg/l) were used individually or in combination with the multiplication medium. Univariate analysis (Kruskal–Wallis test) and principal component analysis plot were performed to assess impact of anti-browning agents on reducing browning but still maintaining consistent rates of proliferation (16.5 ± 0.42) indicated that AC (5 mg/l) was significantly better than either AA alone or any of the combinations of AC and AA. Single shoots were rooted in 0.25 mg/l indole butyric acid and successfully acclimatized under net-house conditions.





Similar content being viewed by others
Abbreviations
- AA:
-
Ascorbic acid
- AC:
-
Activated charcoal
- BA:
-
6-benzyladenine
- IBA:
-
Indole 3-butyric acid
- PCA:
-
Principal Component Analysis
References
Md Taufiq-Ur-Rahman, Shilpi JA, Ahmed M, Hossain CF (2005) Preliminary pharmacological studies on Piper chaba stem bark. J Ethnopharmacol 99:203–209
Bhandari SPS, Babu UV, Garg HS (1998) A lignan from Piper chaba stems. Phytochemistry 47:1435–1436
Parmar VS, Jain SC, Bisht KS, Jain R, Taneja P, Jha A, Tyagi OD, Prasad AK, Wengel J, Olsen CE, Boll PM (1997) Phytochemistry of the genus Piper. Phytochemistry 46:597–673
Parmar VS, Jain SC, Gupta S, Talwar S, Rajwanshi VK, Kumar R, Azim A, Malhotra S, Kumar N, Jain R, Sharma NK, Tyagi OD, Lawrie SJ, Errington W, Howarth OW, Olsen CE, Singh SK, Wengel J (1998) Polyphenols and alkaloids from Piper species. Phytochemistry 49:1069–1078
Morikawa T, Matsuda H, Yamaguchi I, Pongpiriyadacha Y, Yoshikawa M (2004) New amides and gastroprotective constituents from the fruit of Piper chaba. Planta Med 70:152–159
Krishnan MKS (1986) The useful plants of India. Publication and Information Directorate, CSIR, New Delhi
Naz T, Mosaddik A, Rahman MM, Muhammad I, Haque ME, Cho SK (2012) Antimicrobial, antileishmanial and cytotoxic compounds from Piper chaba. Nat Prod Res 26:979–986
Bhojwani SS, Dantu PK (2013) Plant tissue culture: an introductory text. Springer, Heidelberg
Bhat SR, Kackar A, Chandel KPS (1992) Plant regeneration from callus cultures of Piper longum L. by organogenesis. Plant Cell Rep 11:525–528
Rani D, Dantu PK (2012) Direct shoot regeneration from nodal, internodal and petiolar segments of Piper longum L. and in vitro conservation of indexed plantlets. Plant Cell Tiss Organ Cult 109:9–17
Philip VJ, Joseph D, Triggs GS, Dickinson NM (1992) Micropropagation of black pepper (Piper nigrum Linn.) through shoot tip cultures. Plant Cell Rep 12:41–44
Kelkar SM, Krishsnamurthy KV (1998) Adventitious shoot regeneration from root, internode, petiole and leaf explants of Piper colubrinum Link. Plant Cell Rep 17:721–725
Anand A, Rao CS (2000) A rapid in vitro propagation protocol for Piper barberi Gamble, a critically endangered plant. In vitro Cell Dev Biol Plant 36:61–64
Ved DK, Goraya GS (2007) Demand and supply of medicinal plants in India. NMPB, New Delhi and FRLHT
Anonymous (1999) Task force report on conservation and sustainable use of medicinal plants, Planning Commission, Govt. of India, No. 30015/9/99-S&T
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Karioti A, Hadjipavlou-Litina D, Mensah MLK, Fleischer TC, Saltsa H (2004) Composition and antioxidant activity of the essential oils of Xylopia aethiopica (Dun) A. Rich. (Annonaceae) leaves, stem bark, root bark, and fresh and dried fruits, growing in Ghana. J Agric Food Chem 52:8094–8098
Wang J, Liu K, Xu D, Wang Q, Bi K, Song Y, Li J, Zhang L (2012) Rapid micropropagation system in vitro and antioxidant activity of Scabiosa tschiliensis Grunning. Plant Growth Regul. doi:10.1007/s10725-012-9765-4
Hammer O, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electronica 4:1–9
Norelli JL, Aldwinckle HS (1993) The role of aminoglycoside antibiotics in the regeneration and selection of neomycin phosphotransferase-transgenic apple tissue. J Am Soc Hortic Sci 118:311–316
Soniya EV, Das MR (2002) In vitro micropropagation of Piper longum-an important medicinal plant. Plant Cell Tissue Org Cult 70:325–327
Bhat A, Kean OB, Keng CL (2012) Sucrose, benzylaminopurine and photoperiod effects on in vitro culture of Kaempferia galanga Linn. Plant Biosyst 146:900–905
Pan MJ, van Staden J (1998) The use of charcoal in in vitro culture—a review. Plant Growth Regul 26:155–163
Vengadesan G, Ganapathi A, Anand RP, Selvaraj N (2003) Shoot regeneration and ploidy variation in tissue culture of honeydew melon (Cucumis melo L. inodorus). In vitro Cell Dev Biol Plant 39:409–414
Yan H, Yang L, Li Y (2010) Axillary shoot proliferation and tuberization of Dioscorea fordii Prain et Burk. Plant Cell Tissue Org Cult 104:193–198
Madhusudhanan K, Rahiman BA (2000) The effect of activated charcoal supplemented media to browning of in vitro cultures of Piper species. Biol Plant 43:297–299
Linington IM (1991) In vitro propagation of Dipterocarpus intricatus. Plant Cell Tissue Org Cult 27:81–88
Philip S, Banerjee NS, Das MR (2000) Genetic variation and micropropagation in three varieties of Piper longum L. Curr Sci 78:169–173
Panda BM, Hazra S (2012) Micropropagation of Semecarpus anacardium L.: a medicinally important tree species. Plant Biosyst 146:61–68
Mahesh A, Jayachandran R (2012) Influence of plant growth regulators on micropropagation and in vitro flowering of Trichodesma indicum (Linn) R. Br Plant Biosyst 1:1–7
Acknowledgments
PKD gratefully acknowledges the University Grants Commission, New Delhi, for the financial help in the form of a Major Project vide letter no. F.32-382/2006 (SR) and Indian Institute of Spices Research, Calicut, Kerala, for the supply of the plant material. The authors thank Dr B. K. Sinha, Ranchi University, for helping in statistical analysis. The authors thank the Director of the Institute for providing the necessary laboratory facilities to carry out the work. The authors declare no competing financial interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rani, D., Dantu, P.K. Sustained Shoot Multiplication and Method for Overcoming In Vitro Browning in Medicinally Important Plant, Piper chaba Hunt. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 86, 407–413 (2016). https://doi.org/10.1007/s40011-014-0461-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-014-0461-1