Abstract
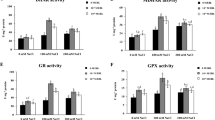
In the present investigation, the effects of 24-epibrassinolide and polyamines (putrescine and spermidine) on antioxidant enzymes (superoxide dismutase, catalase and guaiacol peroxidase), antioxidants (phenols and proline), lipid peroxidation, proteins and photosynthetic pigments in the Adiantum capillus-veneris under salt stress were studied. 24-epibrassinolide and polyamine treatment alone or in combination with salt stress modulate the activities of antioxidant enzymes. Modest decrease in proline content was observed for salt treatment alone. However, the combination of 24-epibrassinolide and polyamines with salt stress caused major increase in proline content over salt stress alone. The combined effect of salt with 24-epibrassinolide increased the phenolic content. Major enhancement in phenolic content was observed for 150 mM NaCl+10−8 EBL. A remarkable decrease in malondialdehyde content was observed in leaves treated with 24-epibrassinolide, putrescine and spermidine with/or without salinity stress. Proteins and photosynthetic pigments did not show any remarkable change under salt stress whereas, treatment of 24-epibrassinolide and polyamines enhanced the titers of protein and photosynthetic pigments of leaves with/or without salt stress. An increase in carotenoid content was observed with salt (150 and 300 mM) stress, which further improved remarkably by supplementation of polyamines and 24-epibrassinolide. Data presented here is one of the first detailed analysis of application of EBL, Put and Spd on the antioxidant system of Adiantum under salt stress.
Similar content being viewed by others
References
Gao S, Ouyang C, Wang S, Xu Y, Tang L, Chen F (2008) Effects of salt stress on growth, antioxidant enzyme and phenylalanine ammonia-lyase activities in Jatropha curcas L. seedlings. Plant Soil Environ 54:373–381
Nawaz K, Hussain K, Majeed A, Farah K, Afghan S, Ali K (2010) Fatality of salt stress to plants: morphological physiological and biochemical aspects. Afr J Biotech 9:5475–5480
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Kalaji HM, Bosa GK, Kościelniak J, Żuk-Gołaszewska K (2010) Effects of salt stress on photosystem II efficiency and CO2 assimilation of two Syrian barley landraces. Environ Exp Bot. doi:10.1016/j.envexpbot
Xia JR, Li YJ, Zou DH (2004) Effects of salinity stress on PSII in Ulva lactuca as probed by chlorophyll fluorescence measurements. Aquat Bot 80:129–137
Blokhina O, Virolainen E, Gagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot (Lond) 91:179–194
McCord LM (2000) The evolution of free radicals and oxidative stress. Am J Med 108:652–659
Wang W, Vincour B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218:1–14
Ahmad P, Sarwat M, Sharma S (2008) Reactive oxygen species, antioxidants and signallling in plants. J Plant Biol 5:167–173
Andre CM, Yvan L, Daniele E (2010) Dietary antioxidants and oxidative stress from a human and plant perspective: a review. Curr Nutr sci 6:2–12
Clouse SD (2011) Brassinosteroid signal transduction: from receptor kinase activation to transcriptional networks regulating plant development. Rev Plant Cell 23:1219–1230
Divi VK, Rahman T, Krishna P (2010) Brassinosteroids mediated stress tolerance in Arabidopsis shown interactions with ascorbic acid, ethylene and salicylic acid pathways. BMC Plant Biol 10:151
Hussain K, Nisar MF, Majeed A, Nawaz K, Bhatii KH, Afghan S, Shahazad A, Zia-ul-Hussnian S (2010) What molecular mechanism is adapted by plants during salt stress tolerance. Afr J Biotech 9:416–422
Choudhary SP, Kanwar M, Bhardwaj R, Yu J-Q, Tran L-S (2012) Chromium stress mitigation by polyamine-brassinosteroid application involves phytohormonal and physiological strategies in Raphanus sativus L. PLoS ONE. doi:10.1371/journal.pone.0033210
Clouse SD (1996) Molecular genetic studies confirm the role of brassinosteroids growth and development. Plant J 10:1–8
Fathutdinova RA, Shakirova FM, Chemeris AV, Sabirzhanov BE, Vakhitov VA (2002) NOR (nucleolar organizer region) reactivity in wheat with ploidy levels treated with phytohormones. Russ J Genet 38:1335–1338
Kagale S, Divi UK, Krochko JE, Keller WA, Krishna P (2007) Brassinosteroid confers tolerance in Arabidopsis thaliana and Brassica napus to a range of abiotic stresses. Planta 225:353–364
Slathia S, Sharma A, Choudhary SP (2012) Influence of exogenously applied epibrassinolide and putrescine on protein content, antioxidant enzymes and lipid peroxidation in Lycopersicon esculentum under salinity Stress. Am J Plant Sci 3:714–720
Anuradha S, Rao SSR (2001) Effect of brassinosteroids on salinity stress induced inhibition of seed germination and seedling growth of rice (Oryza sativa L.). Plant Growth Regul 33:151–153
Sirhindi G, Kumar S, Bhardwaj R, Kumar M (2009) Effects of 24-epibrassinolide and 28-homobrassinolide on the growth and antioxidant enzyme activities in the seedlings of Brassica juncea L. Physiol Mol Biol Plants 15:335–341
Ali B, Hayat S, Fariduddin Q, Ahmad A (2008) 24-epibrassinolide protects against the stress generated by salinity and nickel in Brassica juncea. Chemosphere 72:1387–1392
Cohen S (1998) A guide to the polyamines. Oxford University Press, Oxford
Igarashi K, Kashiwagi W (2000) Polyamines: mysterious modulators of cellular functions. Biochem Biophys Res Commun 271:559–664
Sawhney RK, Tiburcio AF, Altabella T, Galston AW (2003) Polyamines in plants: an overview. J Cell Mol Biol 2:1–12
Takahashi T, Kakehi JI (2009) Polyamines: ubiquitous polycations with unique roles in growth and stress responses. Ann Bot 105:1–6
Giridhar P, Mahendranath G, Venugopalan A, Ravishankar GA (2012) Enhanced yield of food colourant annatto from seeds of Bixia orellana L. The efficacy of polyamines floral spray. Proc Natl Acad Sci India Sect B Biol Sci 8:553–556
Kuznetsov V, Radyukina NL, Shevyakova NI (2006) Polyamines and stress: biological role, metabolism and regulation. Russ J Plant Physiol 53:583–604
Zhao FG, Qin P (2004) Protective role of exogenous polyamines on root tonoplast function against salt stress in barley seedlings. Plant Growth Regul 42:97–103
Jiu-ju D, Shi-Rong G, Yun-Yan K, Yan-Sheng J (2007) Effects of exogenous spermidine on polyamine content and antioxidant system in roots of cucumber under salinity stress. J Ecol Rural Environ 4:11–17
Limaye AS, Deore GB, Shinde BH, Laware SL (2010) Assesement of Adiantum trapeziforme L for antioxidant activities. Asian J Exp Biol Sci 1:75–80
Ahmad P, Jallel CA, Arooz MM, Nabi G (2009) Generation of ROS and non enzymatic antioxidants during abiotic stress in plants. Bot Res Int 2:11–20
Li X-W, Lei M, Chen T-B, Wan X-M (2009) Roles of sulfur in the arsenic tolerant plant Adiantum capillus-veneris and the hyperaccumulator Pteris vittata. J Korean Soc Appl Biol Chem 52:498–502
Singh N, Raj A, Khare PB, Tripathi RD, Jamil S (2010) Arsenic accumulation pattern in 12 Indian ferns and assessing the potential of Adiantum capillus-veneris, in comparison to Pteris vittata, as arsenic hyper accumulator. Bioresour Technol 101:8960–8968
Lowry OH, Rosebrough NJ, Farr AL (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Putter J (1974) Peroxidase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Verlag Chemie, Weinhan, pp 685–690
Aebi HE (1974) Catalase. In: Bergmeyer HU (ed) Methods of enzymatic analysis, vol 4. Verlag Chemie, Weinhan, pp 273–282
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–208
Ragazzi E, Veronese G (1973) Quantitative analysis of phenolics compounds after thin-layer chromatography separation. J Chromatogr 77:369–375
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Zhu JK (2007) Plant salt stress. In: Encyclopedia of Life Science, Wiley, Chichester
Ahmad P, Jallel CA, Arooz MM, Nabi G (2009) Generation of ROS and non enzymatic antioxidants during abiotic stress in plants. Bot Res Int 21:11–20
Scandalios JG (1993) Oxygen stress and superoxide dismutase. Plant Physiol 101:7–12
Chai YY, Jiang CD, Shi L, Shi TS, Gu WB (2010) Effect of exogenous spermine on sweet sorghum during germination under salinity. Biol Planta 54:145–148
Maity U, Bera AK (2009) Effect of exogenous application of brassinolide and salicylic acid on certain physiological and biochemical aspects of green gram (Vigna radita L. wilczek). Indian J Agric Res 43:194–199
Ozdemir FO, Bor M, Demiral, T Tuurkan (2004) Effects of 24-epibrassinolide on seed germination, seedling growth, lipid peroxidation, proline content and antioxidative system of rice (Oryza sativa L.) under salinity stress. Plant Growth Regul 42:203–211
Arora N, Bhardwaj SharmaP, Arora HK (2008) Effect of 28-homobrassinolide on growth, lipid peroxidation and antioxidative enzyme activities in seedlings of Zea mays L. under salinity stress. Acta Physiol Planta 30:833–839
Chen S, Schopfer P (1999) Hydroxyl-radical production in physiological reactions. Eur J Biochem 260:726–745
Scandalios JG (1997) Molecular genetics of SOD in plants. In: Scandalios JG (ed) Oxidative stress and the molecular biology ofantioxidant defense. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, pp 527–568
Sheokand S, Kumari A, Sawhney V (2008) Effect of nitric oxide and putrescine on antioxidative responses under NaCl stress in chickpea plants. Physiol Mol Biol Plants 14:355–362
Sairam RK, Tyagi A (2004) Physiology and molecular biology of salinity stress tolerance in plants. Curr Sci 86:407–421
Anuradha S, Rao SSR (2007) The effect of brassinosteroids on radish (Raphanus sativus) seedlings under cadmium stress. Plant Soil Environ 53:465–472
Ozturk L, Demir Y (2003) Effects of putrescine and ethephon on some oxidative stress enzyme activities and proline content in salt stressed spinach leaves. Plant Growth Regul 40:89–95
Abd El-Wahed MS, Gamal El din KM (2004) Stimulation effect of spermidine and stigmasterol on growth, flowering, chemical constituents and essential oil in chamomile plants (Chamomila recutital L. Rausch) Bull. J Plant Physiol 30:48–60
Celik O, Atak C (2012) The effect of salt stress on antioxidative enzymes and proline content of two Turkish tobacco varieties. Turk J Biol 36:339–356
Anuradha S, Rao SSR (2003) Application of brassinosteroids to rice seed (Oryza sativa) reduce the impact of salt stress on growth, prevented photosynthetic pigment loss and increased nitrate reductase activity. Plant Growth Regul 40:29–32
Houimli SIM, Denden M, Mouhandes BD (2010) Effect of 24-epibrassinolide on the growth, chlorophyll, electrolyte leakage and proline by pepper plants under NaCl stress. Eur Asian J Biosci 4:96–104
Chattopadhayay MK, Tiwari BS, Chattopadhayay G, Sengupta DN, Ghosh B (2002) Protective role of exogenous polyamines on the salinity stressed rice (Oryza sativa) plants. Physiol Planta 116:192–199
Anjum MA (2011) Effect of exogenously applied spermidine on growth and physiology of citrus rootstock Troyer citrange under saline conditions. Turk J Agric For 35:43–53
Acknowledgments
The authors are indebted to Prof. Geeta Sumbali Head, Department of Botany, University of Jammu, for providing necessary facilities in the lab. They wish to express their sincere thanks to Assistant Prof. Vijay Shivgotra from Department of Statistics for his efficient support in the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, A., Slathia, S., Choudhary, S.P. et al. Role of 24-Epibrassinolide, Putrescine and Spermine in Salinity Stressed Adiantum capillus-veneris Leaves. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 84, 183–192 (2014). https://doi.org/10.1007/s40011-013-0195-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-013-0195-5