Abstract
A pot culture experiment was conducted to identify carbon sequestration potential among the crops such as maize, soybean, sorghum, pearl millet, finger millet and rice through estimating carbon (C) and nitrogen (N) partition in root and shoots. Plant biomass, C and N were measured and C:N, C:C, N:N, C:N ratio were calculated at 30, 50, 75, 90 DAS (days after sowing) and at crop maturity from each crop. Among the crops grown, total dry biomass was decreasing in the order of maize > pearl millet > sorghum > soybean > rice > finger millet. The highest plant biomass was recorded in maize crop (15.82 g/plant at 30 DAS and 44.28 g/plant at 90 DAS). There was a considerable variation observed in N:N, C:C and C:N ratio among the crops as well as at crop growth stages wise. The C:N ratio increased with crop growth from 30 DAS to crop maturity in all the crops. The C:N ratio among the crops at 30 DAS was varied from 27.53 (in soybean) to 69.66 (in rice). By balancing both plant biomass and C:N ratio, it was concluded that carbon sequestration potential of maize, sorghum and pearl millet was higher when compared to rice, finger millet and soybean.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carbon (C) is a main energy source for soil microorganism, which is converting residues into plant available nutrients via soil humus [1]. It is an essential component of soil ecosystem, and responsible for chemical reactions, physical events and biological process. Soil organic carbon (SOC) increases in soil mainly due to incorporation of residues, and a net loss of soil C may increase atmospheric C as a green house gas [2]. Global scientist community is interested to know the characterization of soil C dynamics which require actual crop residue C entering into soil. This includes above ground plant parts such as stem and leaf and below ground parts like roots and exudates [3]. Agricultural soils may act as a sink or source of CO2 depending on land management. They can potentially store some of the atmospheric CO2 fixed by crop plants and hence mitigate greenhouse gas emissions from the agricultural sector. The Plant root exudates contributing significant amount of C to soil. The exudates play an important role in C flow in the soil–plant system; 16–33 % of the C assimilated by plants through photosynthesis is transferred into the soil through the roots [4]. Soil organic carbon and nitrogen (N) have long been identified as factors that are important to soil fertility in both managed and natural ecosystems [5]. The rhizodeposit C constitutes 40 % of the total root-derived C [6]. The root residues account for about 50 % of the SOC pool [7]. In plants, 1.5–3.0 times more root C than shoot C is stabilized in the SOC pool, which suggests that root biomass makes a greater contribution to soil C sequestration than aboveground residues [8]. On average, a whole corn plant at physiological maturity contains 436 kg C per 1,000 kg dry matter, distributed as follows: 26.6 % in the leaves, 24.5 % in the stem, 32 % in the grain, 7 % in the roots, and 9.8 % in the cob [8]. About 7.7–20 % of the corn shoot residues are retained in the SOC pool in long-term field experiments [9]. The amount of SOC that exists in any given soil is determined by the balance between the rates of organic carbon input (vegetation, roots) and output (CO2 from microbial decomposition).
Indian soils are deficient in nitrogen and carbon (nearly 62 %). These can be increased by two ways; (1) addition of fertilizers (2) use of crop residues. The first way may increase cost of production and deteriorate soil quality in long run, but in the second case plant biomass of various crops can be used as a crop residues and proper management practices of them maintain the sustainability of soil for longer duration. If nitrogen content in plant part is known, predict nutrient supply through plant biomass at various time intervals can be predicted by using different models which are used to predict the biomass availability and nutrient supply. Therefore, a study was undertaken to provide quantitative estimates of C and N mass in plant parts and the ratios of N:N, C:C and C:N ratios in root and shoots of kharif crops viz. maize, soybean, sorghum, pearl millet, finger millet and rice at various time intervals for accounting carbon sequestration potential of these crops.
Materials and Method
The pot culture experiment was conducted using six crops; maize, soybean, sorghum, pearl millet, finger millet and rice during kharif season of 2011-12 in Glass Screen House of Division of Environmental Soil Science, Indian Institute of Soil Science (IISS), Bhopal, India. The surface soil (0–15 cm) was collected from IISS field for experimentation. The soil was clay loam in texture with pH(1:2.5) 8.06; EC (dSm−1) 0.57; organic carbon 0.44 %; available N 175 kg ha−1; available phosphorus (P) 7.53 kg ha−1, available potassium (K) 185 kg ha−1, available sulphur 9.1 kg ha−1 and diethylene triamine penta acetic acid (DTPA) extractable zinc (Zn) 0.46 ppm. Initial soil was analyzed by following standard methods of analysis [10]. The processed soil was filled in 90 pots @ 10 kg/pot. Five crops (maize, soybean, sorghum, pearl millet, finger millet) were grown and rice seedlings were transplanted in each pot in a manner that each treatment should be replicated for three times. Fertilizers were applied @ 100:60:60 of N, P2O5 and K2O (kg/ha) for sorghum, rice, maize and pearl millet; 30:60:40 of N, P2O5 and K2O (kg/ha) for soybean and 60:40:40 of N, P2O5 and K2O (kg/ha) for finger millet. These fertilizers were applied through urea, di-ammonium phosphate (DAP) and muriate of potash (MOP). Half dose of N and full dose of P and K were applied at the time of sowing in rice, maize, sorghum, pearl millet and finger millet as basal dose and the remaining dose of nitrogen was applied as two equal splits at 30 and 60 DAS. In soybean, all the nutrients were applied at the time of sowing as basal dose. Zinc sulphate (10 kg/ha for rice and 7 kg/ha for other crops) was applied at the time of sowing.
Plant samples were collected at different time interval viz. 30, 50, 75, 90 DAS and at crop maturity for analysis. At every sampling stage one plant uprooted from each pot and washed with distilled water and collected in paper bags. The plant (root and shoot) parts were separated with the help of scissors and stored in separate paper bags. The plants in paper bags were further allowed to air dry for 2–3 days and then placed in hot air oven at 60 °C for 24 h. Dry matter of shoot and root were recorded and total nitrogen and, total carbon were estimated with the help of CN analyzer model FLASH 2000 organic elemental Statistical analysis was done for complete randomized design [11].
Results and Discussion
Plant Biomass
Data pertaining to plant biomass (root and shoot) was presented in Table 1. It was clear from the results that total plant biomass was increasing with plant growth period from initial crop growth to 90 DAS and thereafter it declined at crop maturity stage in all the crops but the amount and rate of accumulation of biomass varied among the crops at various growth stages. During the course of investigation total plant biomass of maize, pearl millet, sorghum, rice, finger millet and soybean varied between 15.82–39.91, 10.75–33.86, 7.46–28.6, 4.14–21.8, 3.53–21.5 and 4.56–17.29 g, respectively.
Total plant biomass was recorded highest at 90 DAS in all crops. Root and shoot biomass varied widely among the crops at different growth stages. The maximum per cent increase in biomass was recorded at 90 DAS. After 90 DAS shoot biomass was slightly declined but root biomass increased from initial to final crop growth stages for all the crops. The root biomass varied in maize 6.40–10.64 g, pearl millet 4.00–8.24 g, sorghum 1.27–5.18 g, rice 1.72–6.14 g, finger millet 1.41–6.08 g and soybean 1.27–3.51 g. Several studies indicated that soil N availability, although strongly altering shoot growth, does not significantly affect the dynamics of root growth at depth [7]. Among all crops, maize crop contributed maximum root biomass (10.64 g) followed by pearl millet, rice, finger millet, sorghum and soybean crops. The balance fertilizer enhanced the carbon sequestration rate, and was more in maize-wheat cropping system [12]. The above result also fulfill the hypothesis of growth curve, in which plant attained maximum biomass and after it slightly decline [13, 14].
N and C:C Ratio in Root and Shoot
Nitrogen and carbon ratio between root and shoot were enlisted in the Table 2. The nitrogen ratio between root and shoot was increased from 30 DAS to 75 DAS and then after decreases in all the crops. In finger millet crop recorded maximum (0.46) N/N ratio followed by rice (0.44), pearl millet (0.42), sorghum (0.40), soybean (0.37) and maize (0.31) at 30 DAS. In initial stages plant take more amounts of nitrogenous substances to increasing plant biomass. In several species it has been observed that localized NO3 − application induces root proliferation due to increased growth of laterals [15–17]. Carbon:carbon (C:C) ratio between root and shoot also varied widely. It was almost equal in all the crops at 30 DAS and increasing with increasing time interval. It was maximum in pearl millet crop (1.04) followed, sorghum (0.99), finger millet (0.98), maize (0.94), rice (0.93) and soybean (0.92) at crop maturity. Carbon content is more in non-legume crops in comparison to legumes. Carbon content increased with the crop growth. It may be due to cumulative increase in the accumulation of biomass through photosynthesis. Carbon is a structural element; it didn’t show higher variation in different crop. It varied between 42 and 46 % in major crops [18].
C:N Ratio
The C:N ratio was considered as important parameter that controls the carbon sequestration potential of the crops. The C:N ratio was lower at initial stages and increased with growth period of the crops. The lower per cent increment in C:N ratio was observed between 30 and 75 DAS but it shot up at 90 DAS. It might be due to higher nitrogen uptake by crops at initially stages and low uptake at later stages and also total C accumulation was higher in the later stages. Different crops showed different C:N, it was varied between 65.7–85.6, 69.7–84.5, 5.4–82.1, 50.4–65.9, 51–57.3 and 27.5–33.9, in sorghum, rice, finger millet, pearl millet, maize and soybean, respectively (Table 2). Nitrogen and C assimilation and allocation in plant parts and between plants play a particular role in crop productivity. Therefore it is affected by nitrogen concentration in soil solution, plant species (C3 or C4) and climatic conditions [19]. Researchers suggested that, the impact of root proliferation on N uptake may be limited [20, 21] and more critical for plant-to-plant competition in N uptake than for N uptake of a whole plant population such as a crop [22]. Carbon nitrogen ratios in plants are also affected by concentration of N in labile pool, crop growth pattern and plant species [23].
Carbon Sequestration Potential
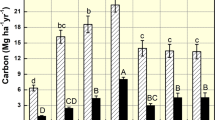
Based on the C:N ratio and yield of the crop biomass, the carbon sequestration potential of a particular crop can be calculated [24]. The biomass with wider C:N ratio takes time for decomposition and slowly releases CO2 to the atmosphere. It leads to increase in storage time of the C in soil. These data can also be used in many simulation models to compute the carbon sequestration potential of crops at particular crop growth stage [25]. The crops such as sorghum, rice and finger millet have the wider C:N ratio and lower crop biomass particularly below ground biomass. But in maize and pearl millet, it was vice versa (Fig. 1).
Balancing both plant biomass and C:N ratio, the crop having higher carbon sequestration potential was identified. By assuming, the crops which had C:N ratio more than the threshold C:N ratio (50) and plant biomass higher than the threshold biomass (25 g/plant) were considered as having higher carbon sequestration potential. It is clear from the study, the carbon sequestration potential of maize, sorghum and pearl millet was higher as compared to rice, finger millet and soybean.
References
Coleman DC, Reid CPP, Cole CV (1983) Biological strategies of nutrient cycling in soil systems. Adv Ecol Res 13:1–57
Lamke RL, Zhong Z, Campbell CA, Zenter RP (2007) Can pulse crops play a role in mitigating greenhouse gases from North American agriculture. Agron J 99:1719–1725
Bolinder MA, Janzen HH, Gregorich EG, Angers DA, Vanden Bygaart AJ (2007) An approach for estimating net primary productivity and annual carbon inputs to soil for common agricultural crops in Canada. Agric Ecosyst Environ 118:29–42
Heal OW, Anderson JM, Swift MJ (1997) Plant litter quality and decomposition: an historical overview. In: Cadish G, Giller KE (eds) Driven by nature: plant litter quality and decomposition. CAB International, Oxford
Kucharik CJ, Brye KR, Norman JM, Foley JA, Gower ST, Bundy LG (2001) Measurements and modeling of carbon and nitrogen cycling in agroecosystems of Southern Wisconsin: potential for SOC sequestration during the next 50 years. Ecosystem 4:237–258
Buyanovsky GA, Wagner GH (1986) Post-harvest residue input to cropland. Plant Soil 93:57–65
Molina JAE, Clapp CE, Linden DR, Allmaras RR, Layese MF, Dowdy RH, Cheng HH (2001) Modeling the incorporation of corn (Zea mays L.) carbon from roots and rhizodeposition into soil organic matter. Soil Biol Biochem 33:83–92
Johnson JMF, Allmaras RR, Reicosky DC (2006) Estimating source carbon from crop residues, roots, and rhizodeposits using the national grain yield database. Agron J 98:622–636
Bolinder MA, Angers DA, Giroux M, Laverdiere MR (1999) Estimating C inputs retained as soil organic matter from corn (Zea mays L.). Plant Soil 215:85–91
Singh D, Chhonkar PK, Pandey RN (1999) Soil plant water analysis: a methods manual. Westville Publishing House, New Delhi
Gomez KA, Gomez A (1983) Statistical procedures for agricultural research. Wiley, New York
Kukal SS, Rasool R, Benbi DK (2009) Soil organic carbon sequestration in relation to organic and inorganic fertilization in rice-wheat and maize-wheat systems. Soil Till Res 102:87–92
Lal R, Kimble JM, Follett RF, Stewart BA (1997) Soil processes and carbon cycles. CRC Press, Boca Raton
Benbi DK (2010) Opportunities for carbon sequestration in Indian agro-ecosystems. In: 75th Annual Convention, IISS, Bhopal, 14–17 Nov 2010, pp 97–104
Samuelson ME, Eliasson L, Larsson CM (1992) Nitrate regulated growth and cytokinin responses in seminal roots of barley. Plant Physiol 98:309–315
Robinson D (1994) The responses of plants to non-uniform supplies of nutrients. New Phytol 127:635–674
Zhang H, Forde BG (2000) Regulation of Arabidopsis root development by nitrate availability. J Exp Bot 51:51–59
Brady NC, Weil RR (2002) The nature and properties of soils. Prentice Hall, Upper Saddle River
Greenwood DJ, Lemaire G, Gosse G, Cruz P, Draycott A, Neeteson JJ (1990) Decline in percentage N of C3 and C4 crops with increasing plant mass. Annu Bot 66:425–436
Dotaniya ML, Datta SC, Biswas DR, Meena BP (2013) Effect of solution phosphorus concentration on the exudation of oxalate ions by wheat (Triticum aestivum L.). Proc Natl Acad Sci India Sect B 83(3):305–309
Dotaniya ML (2012) Crop residue management in rice–wheat cropping system. Lap Lambert Academic Publisher, Germany
Hodge A, Robinson D, Griffiths BS, Fitter AH (1999) Why plants bother: root proliferation results in increased nitrogen capture from an organic patch when two grasses compete. Plant Cell Environ 22:811–820
Lemaire G, Cruz P, Gosse G, Chartier M (1985) Study of the relationship between the dynamics of levy nitrogen dynamics and dry matter growth of a stand of alfalfa (Medicago sativa L.). Agron 5:685–692
Lakaria BL, Singh M, Reddy KS, Biswas AK, Jha P, Choudhary RS, Singh AB, Rao AS (2012) Carbon addition and storage under integrated nutrient management in soybean-wheat cropping sequence in a Vertisol of central India. Natl Acad Sci Lett 35(3):131–138
Jha P, De A, Lakaria BL, Biswas AK, Singh M, Reddy KS, Rao AS (2012) Soil carbon pools, mineralization and fluxes associated with land use change in Vertisols of central India. Natl Acad Sci Lett 35(6):475–483
Acknowledgments
The authors are grateful to Dr. S. Ramana, Principal Scientist, Indian Institute of Soil Science, Bhopal for proving guidance during preparation of manuscript and highly thankful to supporting staff of IISS for the valuable help during experiment.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kushwah, S.K., Dotaniya, M.L., Upadhyay, A.K. et al. Assessing Carbon and Nitrogen Partition in Kharif Crops for Their Carbon Sequestration Potential. Natl. Acad. Sci. Lett. 37, 213–217 (2014). https://doi.org/10.1007/s40009-014-0230-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40009-014-0230-y