Abstract
Purpose
Fracture-related infections (FRI) pose a difficult management problem, as they require numerous surgical interventions and extended antibiotic treatments, especially when a multidrug-resistant organism is involved, with a paucity of available literature that provides guidance.
Results
A 42 year-old male presents an open diaphyseal tibia and fibula fracture, complicated by soft tissue necrosis and infections caused by extensively drug-resistant Acinetobacter baumannii (XDR-Ab). Initially treated with a damage control external fixator, the patient underwent multiple surgical procedures, including radical debridement, negative pressure wound therapy, external fixator revisions and reconstructive surgery using a latissimus dorsi free flap. The emergence of colistin resistance in the Acinetobacter baumannii strain led to the compassionate use of cefiderocol, finally achieving clinical cure.
Conclusions
This case report is one of the firsts that highlights the potential efficacy of cefiderocol in treating challenging bone and joint infections sustained by XDR-Ab. The successful outcome also emphasizes the importance of a comprehensive, multidisciplinary approach in achieving favorable results in complex FRI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fracture-related infection (FRI) is a severe complication following bone injury that requires multiple operations or even amputation to save the patient’s life, causing a major problem for orthopedic surgeons as well as a serious social and economic burden [1]; in order to overcome this issues, multidisciplinary approaches are needed with orthopedic, infectious disease specialist and plastic surgeon combined and timely treatment. Acinetobacter baumannii (Ab) is a Gram-negative, aerobic, pleomorphic and non-motile bacillus, and it is an opportunistic pathogen primarily associated with hospital-acquired infections, representing a major clinical challenge due to its mechanisms of antimicrobial resistance [2, 3]. When a FRI is sustained by a germ of this kind, with few therapeutic options, poses an even greater challenge. Cefiderocol (FDC) is a new siderophore cephalosporin, authorized for infections caused by carbapenem-resistant gram-negative bacteria, and here we present the case of a fracture-related infection successfully treated with FDC requested under compassionate use as rescue therapy since the development of colistin resistance by Acinetobacter baumannii.
Case presentation
A 42 year-old male patient with an open diaphyseal tibia and fibula fracture of the left leg (Gustilo 3b, AO 42A2,4F2B), resulting from a car accident, initially received treatment at a peripheral hospital with a damage control external fixator. His previous medical history was not relevant, except for a prior left sciatic nerve deficit due to chronic radiculopathy. Subsequently, he was transferred to our institution (Umberto I University Hospital, Rome) after 12 days, presenting with severe soft tissue necrosis. Soon after his arrival, he underwent radical debridement and the placement of negative pressure wound therapy (NPWT) (Fig.1). Empirical antibiotic treatment with meropenem (MEM) and vancomycin (VAN) was initiated (Fig. 2).
Main in-hospital events and antibiotics timeline. COL colistin, MEM meropenem, VAN vancomycin, TEC teicoplanin, NPWT negative pressure wound therapy, EF external fixator, CRAB carbapenem-resistant Acinetobacter baumannii, MRSA methicillin-resistant Staphylococcus aureus, MRSE methicillin-resistant Staphylococcus epidermidis, PDR-AB pandrug-resistant Acinetobacter baumannii, LZD linezolid, DAP daptomycin, TG tigecycline, FDC cefiderocol. *Only treatments lasting more than 72 h will be shown
In the month following the initial surgery, the patient underwent serial debridement and NPWT renewal. Almost one month after trauma, a new fracture debridement and reduction with external fixation revision were performed, transitioning from a damage control construct to a monoplanar definitive one (Procallus—Orthofix).
Two months after trauma, the plastic surgery team conducted reconstructive surgery for soft tissue loss using a latissimus dorsi free flap. Intraoperative samples collected were positive for methicillin-resistant Staphylococcus aureus (MRSA), Enterococcus faecalis (susceptible to ampicillin), and carbapenem-resistant Acinetobacter baumannii (CRAB), susceptible only to colistin (MIC = 0.5 mcg/mL). Given the antibiotic options available at our hospital at that time, therapy with MEM, teicoplanin (TEC), and colistin (COL) was initiated. After four weeks of antibiotics, with observed improvement, the patient was discharged.
During the outpatient follow-up four months after the trauma, the patient exhibited flap swelling, decubitus on the fixator body, and modest leakage of proximal pins. Consequently, the external fixator was elevated, and empiric therapy with levofloxacin (LVX) and minocycline (MIN) was prescribed. However, after two days, the patient was readmitted to the peripheral hospital in a confused and febrile state, with severe dyspnea, and diagnosed with septic thrombo-embolism.
The patient initiated antimicrobial therapy with daptomycin (DAP), linezolid (LZD), and COL, as the blood cultures were positive for MRSA, and the skin swabs collected were positive for methicillin-resistant Staphylococcus epidermidis (MRSE) and CRAB. One week after admission, he was transferred to the Department of Infectious and Tropical Diseases of our hospital. Physical examination revealed edema in the left lower limb, purulent discharge from the proximal pins, and positive thermotactile response over the entire knee and leg region (Fig. 3).
Magnetic resonance imaging revealed corpuscular fluid collections at the anterolateral compartment of the left leg. Subsequently, source control surgery was performed, involving debridement, drainage of the collections, NPWT placement, and progressive skin closure using a “shoelace technique”. Simultaneously, MRSA and CRAB were isolated from biopsy. Two weeks after source control surgery, following the isolation of an Ab strain resistant to colistin (MIC > 4 mcg/mL) from biopsies during NPWT renewal, LZD and DAP were discontinued, and therapy continued with COL, MEM, and tigecycline (TG).
The patient’s soft tissue condition progressively improved until five weeks after source control surgery when NPWT was removed, and a dermal substitute was placed until the final combined ortho-plastic surgery eight weeks after source control surgery (approximately seven months after the initial trauma). During this surgery, a partial-thickness epidermal graft was placed, and the external fixator was stabilized by converting it to a multiplanar system (Galaxy System—Orthofix) (Fig. 4). No antibiotic delivery systems were employed, in line with our hospital’s policy, which suggests their utilization in the presence of internal fixation devices [4] or in cases involving substantial joint or endosteal collections.
On the same day, since the susceptibility of the isolated strain to cefiderocol (FDC) was determined in the meantime, the patient discontinued the previous antibiotic therapy and initiated FDC on a compassionate basis as rescue therapy. He received 2 g every 8 h via a 3 h infusion for six weeks without experiencing side effects.
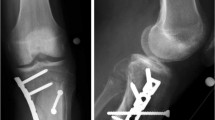
After six weeks, he was discharged from the Department of Infectious and Tropical Diseases, continuing his treatment course with clinical and radiographic follow-ups at our bone infection outpatient clinic at Umberto I University Hospital on a monthly basis. Serial X-ray examinations showed very slow progressive bone callus formation. Finally, two years after the initial trauma, the external fixator was removed (Fig. 5). Concurrently, the skin condition continued to improve, leading to flap debulking surgery three years after the flap surgery.
At the last available checkup conducted three years after patient discharge, the patient reported having returned to his daily activities, walking independently without the need for aids, pain-free, and with a healing skin condition.
Discussion
When a FRI occurs, occasionally, soft tissue defects are of significant size, necessitating multiple surgical debridements and the provisional application of NPWT until the feasibility of definitive soft tissue coverage. Throughout this hospitalization period, pathogens known to cause hospital-acquired infections can become prominent players in FRI development. In this context, this case report underscores several critical antimicrobial aspects in FRI management. One of the primary challenges encountered was the presence of multiple antibiotic-resistant bacteria, including CRAB, MRSA and MRSE, necessitating careful selection of antimicrobial agents guided by culture and susceptibility testing.
Ab is often associated with healthcare settings, especially Intensive Care Units and surgical wards given its ability to persist on surfaces and to aerosolize. Therefore, patients who have undergone invasive procedures, surgeries, or prolonged hospitalizations are at higher risk of acquiring Ab infections, including osteomyelitis. Studies on American soldiers have shown an increase in this type of infection in war wounds, with a progressive shift towards nosocomial acquisition [5]. More recent Chinese studies [6, 7] have shown a progressive increase in FRI sustained by Gram-negative multidrug resistant organism (MDROs). Unfortunately consistent data about bone infections sustained by Gram-negative MDROs in Europe are lacking, but according to the latest ECDC data in Italy, Ab isolates are carbapenem-resistant in 84% of cases [8]. Hence, we can easily expect an increasing number of FRI sustained by Gram-negative MDROs, especially CRAB.
Until now colistin has always been considered the backbone therapy for CRAB, and the available guidelines, both recent and past, suggest the central role of this drug, underlining the complexity of managing such infections [9, 10]. The eradication of MDROs from bone tissue presents a significant management challenge, given the limited availability of active antimicrobials, the difficulty in penetrating bone tissue and the potential drug-related toxicity. Only clinical case reports discuss the management of bone infections caused by MDROs [11,12,13]: Siebenbürger et al. [14], describe the microbiological eradication of Ab and E. cloacae with a regimen of both intravenous and local colistin, and Pasticci et al. [15] reports the tolerability of a long regimen with colistin while treating a polymicrobial prosthetic joint infection.
Furthermore, the development of colistin resistance by Ab, besides being a negative prognostic factor, highlights the challenges associated with managing such infections. Consequently, after weeks of colistin based therapy, we requested cefiderocol on a compassionate basis as a rescue therapy for our patient.
Cefiderocol is a new siderophore cephalosporin approved for the treatment of carbapenem-resistant gram-negative bacterial infections [16]. Even if CREDIBLE-CR [17] study has raised doubts about its effectiveness in treating extensively resistant (XDR)-Ab infections, real life experiences of complex infections successfully treated with FDC are steadily rising. We found five other similar cases (table 1): among these, Dagher at al. [19], describe XDR-Ab osteomyelitis treated with FDC for 109 days, achieving both clinical and microbiological cure. It is interesting to note that all cases were supported by colistin-sensitive Ab, unlike ours, which had developed resistance.
Alamarat et al., [22] also describe long-term (14 weeks) compassionate use of FDC to treat chronic osteomyelitis caused by XDR Pseudomonas aeruginosa and ESBL Klebsiella pneumoniae in a pediatric patient, resulting in apparent cure and avoided amputation. All these cases, along with the one described here, seem to support the role of FDC in treating complex XDR-Ab infections, especially when biofilm is involved. This is probably due to its action on the bacterial iron-transport system because iron is crucial for biofilm formation, and siderophore production is upregulated in biofilms [23].
One of the key points highlighted in this case is the complexity and challenges associated with managing open fractures, especially those complicated by soft tissue necrosis and subsequent infections. The initial presentation of soft tissue necrosis necessitated aggressive multiple debridements and NPWT placement to promote wound healing and control of the infection. The multidisciplinary approach involving plastic surgery, infectious diseases, and orthopedic surgery was crucial in managing the various aspects of the patient's condition, including wound care, infection control, and surgical interventions. Source control surgeries, including debridement and drainage of fluid collections, were essential in managing persistent infections and preventing further complications.
The patient's return to normal activities with a healing skin condition signifies a favorable prognosis and underscores the importance of comprehensive management strategies in achieving successful outcomes in complex cases of FRI.
Conclusions
This case report illustrates the challenges and complexities involved in the management of FRI especially when a multidrug-resistant organism is involved. A multidisciplinary approach incorporating surgical intervention, antibiotic therapy, wound care, and long-term follow-up is essential for achieving optimal outcomes in such cases. The good outcome achieved, although we cannot attribute it to FDC only, supports, in line with the literature, the use of this antibiotic in managing these very complex infections.
Data availability
No datasets were generated or analysed during the current study.
Change history
07 June 2024
A Correction to this paper has been published: https://doi.org/10.1007/s15010-024-02306-w
References
Metsemakers WJ, Morgenstern M, Senneville E, et al. Fracture-related infection (FRI) group general treatment principles for fracture-related infection: recommendations from an international expert group. Arch Orthop Trauma Surg. 2020. https://doi.org/10.1007/s00402-019-03287-4.
Lee CR, et al. ‘Biology of Acinetobacter baumannii: pathogenesis, antibiotic resistance mechanisms, and prospective treatment options’. Front Cell Infect. 2017. https://doi.org/10.3389/fcimb.2017.00055.
Harding CM, Hennon SW, Feldman MF. Uncovering the mechanisms of Acinetobacter baumannii virulence. Nat Rev Microbiol. 2018;16(2):91–102. https://doi.org/10.1038/nrmicro.2017.148.
De Meo D, Ceccarelli G, Iaiani G, Lo Torto F, Ribuffo D, Persiani P, Villani C. Clinical application of antibacterial hydrogel and coating in orthopaedic and traumatology surgery. Gels. 2021;7(3):126. https://doi.org/10.3390/gels7030126.
Fily F, Ronat JB, Malou N, et al. Post-traumatic osteomyelitis in middle east war-wounded civilians: resistance to first-line antibiotics in selected bacteria over the decade 2006–2016. BMC Infect Dis. 2019;19(1):103. https://doi.org/10.1186/s12879-019-3741-9.
Ren Y, Liu L, Sun D, et al. Epidemiological updates of post-traumatic related limb osteomyelitis in china: a 10 years multicentre cohort study. Int J Surg. 2023;109(9):2721–31. https://doi.org/10.1097/JS9.0000000000000502.
Zhang Z, Liu P, Wang W, et al. Epidemiology and drug resistance of fracture-related infection of the long bones of the extremities: a retrospective study at the largest trauma center in Southwest China. Front Microbiol. 2022;12(13):923735. https://doi.org/10.3389/fmicb.2022.923735.
European Centre for Disease Prevention and Control (ECDC). Antimicrobial resistance in the EU/EEA (EARS-Net). http://atlas.ecdc.europa.eu/public/index.aspx?Dataset=27&HealthTopic=4&Indicator=802101&GeoResolution=2&TimeResolution=Year&StartTime=2012&EndTime=2022&CurrentTime=2022&Distribution=802106&DistributionRepresentation=B&TimeSeries=region&TimeSeriesRepresentation=T. (Accessed 26 Feb 2024).
Tiseo G, Brigante G, Giacobbe DR, et al. Diagnosis and management of infections caused by multidrug-resistant bacteria: guideline endorsed by the Italian (society of infection and tropical diseases SIMIT), the Italian society of anti-infective therapy (SITA), the Italian group for antimicrobial stewardship (GISA), the Italian association of clinical microbiologists (AMCLI) and the Italian society of microbiology (SIM). Int J Antimicrob Agents. 2022;60(2):106611. https://doi.org/10.1016/j.ijantimicag.2022.106611.
Depypere M, Kuehl R, Metsemakers WJ, et al. Fracture-related infection (FRI) consensus group recommendations for systemic antimicrobial therapy in fracture-related infection: a consensus from an international expert group. J Orthop Trauma. 2020;34(1):30–41. https://doi.org/10.1097/BOT.0000000000001626.
Krajewski J, Bode-Böger SM, Tröger U, Martens-Lobenhoffer J, et al. Successful treatment of extensively drug-resistant Pseudomonas aeruginosa osteomyelitis using a colistin- and tobramycin-impregnated PMMA spacer. Int J Antimicrob Agents. 2014;44(4):363–6. https://doi.org/10.1016/j.ijantimicag.2014.05.023.
Papagelopoulos PJ, Mavrogenis AF, Giannitsioti E, et al. Management of a multidrug-resistant Pseudomonas aeruginosa infected total knee arthroplasty using colistin. A case report and review of the literature. J Arthroplasty. 2007;22(3):457–63. https://doi.org/10.1016/j.arth.2006.05.006.
Beieler AM, Belknap RW, Dayton MR, et al. Eradication of multidrug-resistant Acinetobacter baumannii in a female patient with total hip arthroplasty, with debridement and retention: a case report. J Med Case Rep. 2009;3(3):45. https://doi.org/10.1186/1752-1947-3-45.
Siebenbürger G, Grabein B, Schenck T, et al. Eradication of Acinetobacter baumannii/Enterobacter cloacae complex in an open proximal tibial fracture and closed drop foot correction with a multidisciplinary approach using the Taylor Spatial Frame®: a case report. Eur J Med Res. 2019;24(1):2. https://doi.org/10.1186/s40001-019-0360-2.
Pasticci MB, Di Filippo P, Pasqualini L, et al. Tolerability and efficacy of long-term treatment with daptomycin, ceftazidime and colistin in a patient with a polymicrobial, multidrug-resistant prosthetic joint reinfection: a case report. J Med Case Rep. 2014;12(8):186. https://doi.org/10.1186/1752-1947-8-186.
Zhanel GG, Golden AR, Zelenitsky S, et al. Cefiderocol: a siderophore cephalosporin with activity against carbapenem-resistant and multidrug-resistant gram-negative bacilli. Drugs. 2019;79(3):271–89. https://doi.org/10.1007/s40265-019-1055-2.
Bassetti M, Echols R, et al. Efficacy and safety of cefiderocol or best available therapy for the treatment of serious infections caused by carbapenem-resistant gram-negative bacteria (CREDIBLE-CR): a randomised, open-label, multicentre, pathogen-focused, descriptive, phase 3 trial. Lancet Infect Dis. 2021;21(2):226–40. https://doi.org/10.1016/S1473-3099(20)30796-9.
Oliva A, Ceccarelli G, De Angelis M, Sacco F, Miele MC, Mastroianni CM, Venditti M. Cefiderocol for compassionate use in the treatment of complicated infections caused by extensively and pan-resistant Acinetobacter baumannii. J Glob Antimicrob Resist. 2020;23:292–6. https://doi.org/10.1016/j.jgar.2020.09.019.
Dagher M, Ruffin F, Marshall S, et al. Case report: successful rescue therapy of extensively drug-resistant Acinetobacter baumannii osteomyelitis with cefiderocol. Open Forum Infect Dis. 2020. https://doi.org/10.1093/ofid/ofaa150.
Zingg S, Nicoletti GJ, Kuster S, et al. Cefiderocol for extensively drug-resistant gram-negative bacterial infections: real-world experience from a case series and review of the literature. Open Forum Infect Dis. 2020. https://doi.org/10.1093/ofid/ofaa185.
Mabayoje DA, NicFhogartaigh C, Cherian BP, Tan MGM, Wareham DW. Compassionate use of cefiderocol for carbapenem-resistant Acinetobacter baumannii prosthetic joint infection. JAC Antimicrob Resist. 2021;3(Suppl 1):i21–4. https://doi.org/10.1093/jacamr/dlab055.
Alamarat ZI, Babic J, Tran TT, et al. Long-term compassionate use of cefiderocol to treat chronic osteomyelitis caused by extensively drug-resistant Pseudomonas aeruginosa and extended-spectrum-β-lactamase-producing Klebsiella pneumoniae in a pediatric patient. Antimicrob Agents Chemother. 2020;64(4):e01872-19. https://doi.org/10.1128/AAC.01872-19.
Kang D, Kirienko NV. Interdependence between iron acquisition and biofilm formation in Pseudomonas aeruginosa. J Microbiol. 2018;56(7):449–57. https://doi.org/10.1007/s12275-018-8114-3.
Funding
Open access funding provided by Università degli Studi di Roma La Sapienza within the CRUI-CARE Agreement. The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
Flavia Petrucci, Beatrice Perciballi, Giancarlo Iaiani and Daniele De Meo have created the study design. Marco Rivano Capparuccia and Federico Lo Torto have recruited the patient. Marco Rivano Capparuccia, Giancarlo Iaiani, Federico Lo Torto, Diego Ribuffo, Stefano Gumina and Daniele De Meo have performed the treatment. Flavia Petrucci, Beatrice Perciballi, Stefano Gumina and Diego Ribuffo have carried out the data analysis and interpretation and prepared the figures. Flavia Petrucci, Beatrice Perciballi and Daniele De Meo have written the main manuscript text, which has been revised critically for important intellectual content by Marco Rivano Capparuccia, Giancarlo Iaiani, Federico Lo Torto, Diego Ribuffo, Stefano Gumina. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
The patient has given both verbal and written consent to the examination.
Consent to publish
The patient has given his consent to the publication of the results of the treatment in an anonymized form.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Petrucci, F., Perciballi, B., Rivano Capparuccia, M. et al. Compassionate use of cefiderocol in a complex case of extensively drug-resistant Acinetobacter baumannii fracture-related infection: a comprehensive approach and multidisciplinary management. Infection (2024). https://doi.org/10.1007/s15010-024-02294-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s15010-024-02294-x