Abstract
Purpose
Cerebrospinal fluid (CSF) granulocytes are associated with bacterial meningitis, but information on its diagnostic value is limited and primarily based on retrospective studies. Therefore, we assessed the diagnostic accuracy of CSF granulocytes.
Methods
We analyzed CSF granulocytes (index test) from all consecutive patients in two prospective cohort studies in the Netherlands. Both studies included patients ≥ 16 years, suspected of a central nervous system (CNS) infection, who underwent a diagnostic lumbar puncture. All episodes with elevated CSF leukocytes (≥ 5 cells per mm3) were selected and categorized by clinical diagnosis (reference standard).
Results
Of 1261 episodes, 625 (50%) had elevated CSF leukocytes and 541 (87%) were included. 117 of 541 (22%) were diagnosed with bacterial meningitis, 144 (27%) with viral meningoencephalitis, 49 (9%) with other CNS infections, 76 (14%) with CNS autoimmune disorders, 93 (17%) with other neurological diseases and 62 (11%) with systemic diseases. The area under the curve to discriminate bacterial meningitis from other diagnoses was 0.97 (95% confidence interval [CI] 0.95–0.98) for CSF granulocyte count and 0.93 (95% CI 0.91–0.96) for CSF granulocyte percentage. CSF granulocyte predominance occurred in all diagnostic categories. A cutoff at 50% CSF granulocytes gave a sensitivity of 94% (95% CI 90–98), specificity of 80% (95% CI 76–84), negative predictive value of 98% (95% CI 97–99) and positive predictive value of 57% (95% CI 52–62).
Conclusion
CSF granulocytes have a high diagnostic accuracy for bacterial meningitis in patients suspected of a CNS infection. CSF granulocyte predominance occurred in all diagnostic categories, limiting its value in clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Central nervous system (CNS) infections are a global health problem with high mortality and morbidity rates [1]. Diagnosing CNS infections can pose a challenge as clinical features, such as the classic triad of bacterial meningitis (fever, headache and neck stiffness), lack adequate sensitivity and correspond to a broad differential diagnosis ranging from benign to life-threatening conditions [2]. As clinical features lack diagnostic accuracy, cerebrospinal fluid (CSF) examination is required in the diagnostic work-up of patients suspected of a CNS infection. Conventional CSF examination consists of determining the number of leukocytes and their differentiation, glucose and total protein level, after which microbiological testing is done to identify a pathogen. However, in acute care settings, physicians frequently have to establish a treatment strategy based on the initial CSF results as microbiological testing requires more time. Several prediction models have been established to aid the probability assessment of bacterial meningitis in patients suspected of a CNS infection, often taking into account the relative amount or absolute number of granulocytes [3]. Granulocytes may give information on the type of immune response and an abundance relative to the number of mononuclear cells has been associated with bacterial meningitis [4]. However, little is known about the diagnostic accuracy of CSF granulocytes as most of the current knowledge on CSF leukocyte subsets is based on studies that are limited by a retrospective study design, comparing cases of bacterial meningitis to other pre-defined diagnoses instead of evaluating them in the context of all cases suspected of a CNS infection [5]. Therefore, we evaluated the diagnostic accuracy of CSF granulocytes in patients with a suspected CNS infection.
Methods
Study population and data collection
We analyzed CSF granulocyte counts and proportions (= index tests) from all consecutive patients in two prospective cohort studies in the Netherlands. One single-center pilot study (pediatric and adult causes of encephalitis and meningitis [PACEM]) [2] and one ongoing multicenter cohort study (improving prognosis using innovative methods to diagnose causes of encephalitis [I-PACE]) [6], respectively, from the periods 2012 to 2015 and 2017 to now. Both studies included patients ≥ 16 years that were suspected of a CNS infection and who underwent a diagnostic lumbar puncture. Patients were included either at presentation at the emergency department or during admission in case the lumbar puncture was performed at a later time point. Exclusion criteria were a suspected CNS infection within 3 months after neurosurgery and/or a traumatic brain injury or the presence of a neurosurgical or neurostimulatory device within the central nervous system. Eligible patients were reported to the investigators by the treating physician or identified during morning rounds and in laboratory records. Written informed consent was obtained from all participants, or their legal representative when the patient was incapacitated at the time of inclusion. Repeated inclusions of the same patient during the study period were considered only in those who underwent a lumbar puncture for a new suspicion of a CNS infection. Follow-up lumbar punctures of an initial suspicion were not included in the current study. Clinical data were obtained from the patients’ electronic health records and stored in secured online case record forms. All patient data were anonymized in accordance with Dutch privacy legislation.
We selected episodes with abnormal CSF leukocyte counts (i.e., ≥ 5 cells per mm3) in which CSF granulocytes were determined as part of routine clinical care and granulocyte predominance was defined as a granulocyte percentage of > 50%. Cell counts and differentiation were conducted using an automated cell counter. Episodes in which CSF granulocytes were not determined were excluded from the analysis. The diagnostic accuracy of CSF granulocytes, as well as CSF leukocytes, was determined. The following measures of diagnostic accuracy were calculated: sensitivity, specificity, predictive values and the area under the curve (AUC). In addition, the Youden’s index, a cutoff value that maximizes the sensitivity and specificity, was determined and evaluated.
Episodes were considered hospital-acquired if they occurred during admission (> 48 h after presentation) or within 1 week after discharge from a previous hospital admission. All other episodes were considered community-acquired. An immunocompromised state was defined as the use of immunosuppressive drugs or a medical history of human immunodeficiency virus (HIV) infection, cancer, diabetes mellitus or alcoholism. The Glasgow Coma Scale (GCS) score was used to describe the level of consciousness at presentation [7]. A GCS-score of ≤ 14 was considered as an altered mental status and a GCS-score of ≤ 8 indicated a coma.
Diagnostic categories
The final clinical diagnoses (= reference standard) were classified into the following categories, as previously described [2]: CNS infection, CNS autoimmune disorder, other neurological disease and systemic disease; CNS infections were subdivided into bacterial meningitis, viral meningoencephalitis or other CNS infection. All episodes were independently evaluated and classified by two clinicians (SS, SO) and disagreements were resolved in consensus with a third clinician (MB). Cohen’s Kappa coefficient (κ), a measure of inter-observer agreement, was calculated with a κ = 0.76 for the first cohort and κ = 0.58 for the second cohort.
Statistical analysis
Data analyses and visualization were performed using RStudio version 4.2.1 (RStudio Inc. Boston, USA) and GraphPad Prism version 9.5.1 (GraphPad Software Inc. California, USA). The following additional R packages were used: tidyverse, ggplot2 and pROC. Tidyverse was used for data handling, ggplot2 for data visualization and pROC to produce receiver operator characteristics (ROC) curves and to calculate the area under the curve (AUC), sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV). P-values ≤ 0.05 were considered statistically significant.
Results
Patient characteristics
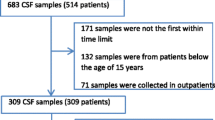
Overall, 1261 episodes of suspected CNS infections were included in this study of which 636 episodes (50%) were excluded because of a CSF leukocyte count < 5 cells per mm3. Of the remaining episodes, 84 were excluded due to the absence of CSF granulocyte counts. In total, 541 of 1261 episodes were eligible for analysis. The median age of all episodes was 53 years (interquartile range [IQR] 37–66) and 237 of 541 (44%) episodes occurred in females (Table 1). In 38 of 380 (10%) episodes, a previous CNS infection was reported and in 37 of 538 (7%) the episode was defined as hospital-acquired. An immunocompromised state was present in 241 of 541 episodes (45%) and was due to immunosuppressive therapy in 84 of 539 episodes (16%), diabetes mellitus in 84 of 541 episodes (16%), cancer in 75 of 539 episodes (14%) and HIV in 44 of 541 episodes (8%). Upon presentation, headache was the most common symptom occurring in 327 of 472 episodes (69%), followed by nausea or vomiting in 199 of 469 episodes (42%) and seizures in 62 of 513 episodes (12%). Fever was present in 216 of 532 episodes (41%), neck stiffness occurred in 126 of 533 episodes (24%) and an altered mental status in 256 of 538 episodes (48%).
Final diagnoses
A CNS infection was diagnosed in 310 of 541 episodes (57%; Fig. 1). Of those, viral meningoencephalitis was the most common cause and occurred in 144 of 541 episodes (27%). Bacterial meningitis and other CNS infections occurred in, respectively, 117 (22%) and 49 (9%) of 541 episodes. The remaining episodes were diagnosed as CNS autoimmune disorder in 76 of 541 episodes (14%), other neurological disease in 93 of 541 episodes (17%) or systemic disease in 62 of 541 episodes (11%).
Cerebrospinal fluid characteristics
The median CSF leukocyte count of all episodes suspected of a CNS infection was 60 per mm3 (IQR 11–389), with a median CSF granulocyte count of 5 per mm3 (IQR 1–123). The median granulocyte percentage was 20% (IQR 1–73) and 191 of 541 episodes (35%) had granulocyte predominance (Table 2). Episodes diagnosed with bacterial meningitis had a median CSF leukocyte count of 2189 cells per mm3 (IQR 876–6670) with a median granulocyte count of 1571 cells per mm3 (IQR 658–6211). The median granulocyte percentage was 89% (IQR 78–95) and 109 of 117 episodes (93%) demonstrated granulocyte predominance (Table 2 and Fig. 2). The median CSF leukocyte count of episodes diagnosed with viral meningoencephalitis was 101 cells per mm3 (IQR 34–237) with a median granulocyte count of 5 cells per mm3 (IQR 1–23). The median granulocyte percentage was 6% (IQR 1–28) with a granulocyte predominance in 20 of 144 episodes (14%). Episodes with other CNS infections had a median CSF leukocyte count of 95 (IQR 30–319) and a median granulocyte count and percentage of 4 cells per mm3 (IQR 1–106) and 10% (IQR 0–50), respectively. In 11 of the 49 episodes (22%) with other CNS infections, the CSF demonstrated granulocyte predominance. Granulocyte predominance occurred in all diagnostic categories (Fig. 2). Eight of 117 episodes (7%) diagnosed with bacterial meningitis had a granulocyte percentage of 50% or lower. Of those, four had a concomitant positive culture of either CSF (Streptococcus pneumoniae, Listeria monocytogenes or Rhodotorula mucilaginosa) or blood (Streptococcus bovis). The remaining four episodes were CSF or blood culture negative.
Diagnostic accuracy
The majority of episodes diagnosed with bacterial meningitis were characterized by high leukocyte counts and a high granulocyte percentage in the CSF (Fig. 3). A specificity of 100% for the diagnosis of bacterial meningitis was established at a cutoff point of 2967 CSF leukocytes per mm3 and 2754 CSF granulocytes per mm3 with a similar sensitivity of, respectively, 44% (95% confidence interval [CI] 34–52) and 43% (95% CI 34–51) (Table 3). A cutoff at 50% CSF granulocytes showed a sensitivity of 94% (95% CI 90–98) and specificity of 80% (95% CI 76–84). The Youden index for granulocyte percentages (70% CSF granulocytes) showed a sensitivity and specificity of, respectively, 86% (95% CI 80–92) and 90% (95% CI 87–92). The AUC for predicting bacterial meningitis using a single predictor was 0.94 (95% CI 0.91–0.96) for CSF leukocytes, 0.97 (95% CI 0.95–0.98) for CSF granulocytes per mm3 and 0.93 (95% CI 0.91–0.96) for CSF granulocyte percentage (Fig. 4).
a ROC curves of leukocytes and granulocytes in the CSF of all episodes. b ROC curves of leukocytes and granulocytes in the CSF of episodes with 5–3000/mm3 CSF leukocytes. The AUCs of leukocytes, granulocytes and granulocyte percentage in the CSF as a single predictor for bacterial meningitis are depicted in, respectively, black, yellow and blue. The AUCs of a logistic regression model using bacterial meningitis as the dependent variable and leukocytes together with a cutoff point for granulocyte percentage at either granulocyte predominance (> 50%) or the Youden index as the independent variables are depicted in, respectively, in grey and green. Receiver operator characteristics (ROC), cerebrospinal fluid (CSF), area under the curve (AUC).
A logistic regression model was fitted to predict the probability of being diagnosed with bacterial meningitis using a combination of CSF leukocytes with a cutoff at either 50% or 70% (Youden index) CSF granulocytes. The model using a cutoff of 50% CSF granulocytes yielded an AUC of 0.96 (CI 0.94–0.98) and the model using a cutoff of 70% CSF granulocytes yielded an AUC of 0.96 (CI 0.94–0.98). Since a CSF leukocyte count of 2967 cells per mm3 ruled out other diagnoses (positive predictive value, PPV 100%), the same test characteristics were calculated for a subgroup of episodes with leukocyte counts between 5 and 3000 cells per mm3 (approximation of 2967 mm3) which showed a similar outcome (Table 3 and Fig. 4).
Discussion
In this prospective study of diagnostic accuracy, we showed that CSF granulocytes have a high diagnostic accuracy for diagnosing bacterial meningitis among patients suspected of a CNS infection. The association between bacterial meningitis and elevated CSF granulocytes has been a consistent finding [4]. CSF granulocyte predominance was found mostly in episodes with bacterial meningitis as well; however, its occurrence was evident across all diagnostic categories limiting its utility in providing guidance for therapeutic considerations. These results correspond with the findings on CSF granulocyte percentage in two large retrospective cohort studies [8, 9]. Both studies, containing CSF samples of > 1000 patients with elevated CSF leukocytes, described the causes of CSF pleocytosis, their corresponding CSF features and the predictive value of CSF leukocytes (alone or as part of the CHANCE score) for the diagnosis of bacterial meningitis. Although the predictive value of CSF leukocytes alone was similar to the one found in our study, none of the studies mentioned the predictive value of CSF granulocytes. In addition, both studies were limited by a retrospective study design in which cases of bacterial meningitis were compared to other cases with a pre-defined diagnosis instead of all cases suspected of a CNS infection. Comparisons as such do not render reliable predictive values for the diagnosis of bacterial meningitis, as part of the study population may not have had an initial suspicion of a CNS infection. Whereas the sensitivity of CSF features derived from those studies might be similar in the context of patients suspected of a CNS infection, its specificity will likely be lower.
CSF granulocyte predominance in viral CNS infections has been described before, for instance a prospective cohort study of 418 patients with enterovirus meningitis found that 28% presented with granulocyte predominance, especially in the early days of the disease [10]. Conversely, in bacterial meningitis, CSF granulocyte predominance may be absent. The occurrence of Listeria monocytogenes in those episodes was expected as lower CSF granulocyte proportions have already been described [11] and granulocyte predominance is found in only 65–81% of cases [12]. However, also for Streptococcus pneumoniae, the most common cause of bacterial meningitis [13] which typically manifests with a high leukocyte count in CSF [14], we found a case with monocyte predominance. Lower CSF leukocyte counts in pneumococcal meningitis (less than 1000 cells per mm3) have been described to occur in 33% of cases [14, 15] and even cases with normal CSF leukocyte counts have been described in 2% [16]. However, in those instances, the granulocyte proportions are often not determined and CSF leukocyte differentiation is not performed when the leukocyte count is normal. Despite its high diagnostic accuracy in bacterial meningitis, CSF granulocytes do not discriminate perfectly. This finding stresses the need for new diagnostics to differentiate CNS infections or discriminate CNS infections from non-CNS infections, especially in the acute care setting when microbiological examination is still pending. Measurements of inflammatory proteins in CSF, such as C-reactive protein (CRP) and cytokines, may be an interesting option to explore as their measuring techniques are already implemented in many clinical laboratories and results are rapidly available. Several studies have evaluated the diagnostic accuracy of various inflammatory markers in CSF to identify patients with bacterial meningitis [17,18,19,20]. Some CSF markers, such as interleukin (IL-)6, IL-10, CRP and procalcitonin, show considerably discriminatory capabilities but do not outperform routinely measured CSF parameters [17,18,19,20]. These findings are derived from small study populations that do not accurately represent clinical practice. Larger studies on diagnostic accuracy that include all patients suspected of a CNS infection are needed to evaluate the added diagnostic value of either individual or combinations of inflammatory markers in CSF.
Our study is subject to certain limitations. First, 636 of 1261 episodes (50%) had a normal CSF leukocyte count (< 5 leukocytes per mm3) and were filtered out due to the absence of information on leukocyte determination in the majority of cases as this was frequently omitted in the diagnostic work-up. Although this comprises half of the potentially eligible episodes, it does not reduce the validity of this study as those episodes would no longer be suspected of a CNS infection. Second, the Cohen’s Kappa coefficient ranges from moderate to substantial, demonstrating a considerable amount of disagreement between the observers when evaluating the episodes. Even though this may have led to the misclassification of some episodes, a certain amount of inter-observer disagreement was to be expected in a study population of this size and complexity. Furthermore, it underlines the importance of novel diagnostic strategies to differentiate patients with a higher degree of certainty. Finally, a substantial number of episodes were included in a tertiary referral center with specific expertise in CNS infections. This probably introduced selection bias as episodes may have been included, for instance, during a hospital admission in the context of a second opinion. However, the majority of patients came from the region in which the hospital was located and was thus an approximation of the average population. Although this might have a slight effect on the external validity of the results, it does provide relevant information about atypical or complex cases.
Conclusion
CSF granulocytes have a high diagnostic accuracy to diagnose bacterial meningitis among patients suspected of a CNS infection. However, CSF granulocyte predominance occurred in all diagnostic categories, limiting its use in clinical decision making. Instead, it is safer to base treatment decisions on microbiological examination, which is indicated regardless of the CSF leukocyte count or its differentiation.
Data availability
Data protection regulations in the Netherlands do not allow for sharing of individual participant data. Datasets with selected aggregated data is available upon request. Proposals can be directed to ipace@amsterdamumc.nl.
References
van de Beek D, et al. Community-acquired bacterial meningitis. Nat Rev Dis Primers. 2016;2:16074.
Khatib U, van de Beek D, Lees JA, Brouwer MC. Adults with suspected central nervous system infection: A prospective study of diagnostic accuracy. J Infect. 2017;74:1–9.
van Zeggeren IE, et al. Systematic review and validation of diagnostic prediction models in patients suspected of meningitis. J Infect. 2020;80:143–51.
Spanos A, Harrell FE Jr, Durack DT. Differential diagnosis of acute meningitis. An analysis of the predictive value of initial observations. JAMA. 1989;262:2700–7.
Brouwer MC, Thwaites GE, Tunkel AR, van de Beek D. Dilemmas in the diagnosis of acute community-acquired bacterial meningitis. Lancet. 2012;380:1684–92.
Olie SE, et al. Seizures in adults with suspected central nervous system infection. BMC Neurol. 2022;22:426.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2:81–4.
Dyckhoff-Shen S. et al. (2023) Characterization and diagnosis spectrum of patients with cerebrospinal fluid pleocytosis. Infection.
Baunbaek Egelund G, et al. Cerebrospinal fluid pleocytosis in infectious and noninfectious central nervous system disease: A retrospective cohort study. Medicine (Baltimore). 2017;96: e6686.
Bodilsen J, et al. Enterovirus Meningitis in Adults: A Prospective Nationwide Population-Based Cohort Study. Neurology. 2021;97:e454–63.
Charlier C, et al. Clinical features and prognostic factors of listeriosis: the MONALISA national prospective cohort study. Lancet Infect Dis. 2017;17:510–9.
Koopmans MM, et al. Listeria monocytogenes sequence type 6 and increased rate of unfavorable outcome in meningitis: epidemiologic cohort study. Clin Infect Dis. 2013;57:247–53.
Koelman DLH, et al. Changing Epidemiology of Bacterial Meningitis Since Introduction of Conjugate Vaccines: 3 Decades of National Meningitis Surveillance in The Netherlands. Clin Infect Dis. 2021;73:e1099–107.
Koelman DLH, et al. Pneumococcal Meningitis in Adults: A Prospective Nationwide Cohort Study Over a 20-year Period. Clin Infect Dis. 2022;74:657–67.
Weisfelt M, et al. Attenuated cerebrospinal fluid leukocyte count and sepsis in adults with pneumococcal meningitis: a prospective cohort study. BMC Infect Dis. 2006;6:149.
van Soest TM, et al. Bacterial meningitis presenting with a normal cerebrospinal fluid leukocyte count. J Infect. 2022;84:615–20.
Srinivasan L, et al. Cerebrospinal fluid cytokines in the diagnosis of bacterial meningitis in infants. Pediatr Res. 2016;80:566–72.
de Almeida SM, Castoldi JR, Riechi SC. Comparison between cerebrospinal fluid biomarkers for differential diagnosis of acute meningitis. Diagnosis (Berl). 2023;10:298–308.
Alons IM, et al. Procalcitonin in cerebrospinal fluid in meningitis: a prospective diagnostic study. Brain Behav. 2016;6:e00545.
Reshi Z, et al. Cerebrospinal fluid procalcitonin as a biomarker of bacterial meningitis in neonates. J Perinatol. 2017;37:927–31.
Acknowledgements
I-PACE Study Group (alphabetical order): Judith Citroen (Onze Lieve Vrouwe Gasthuis), Björn M. van Geel (Noordwest Ziekenhuisgroep), Sebastiaan G.B. Heckenberg (Spaarne Gasthuis), Korné Jellema (Haaglanden Medisch Centrum), Maartje I. Kester (Flevoziekenhuis), Joep Killestein (Amsterdam Universitair Medisch Centrum, locatie VUMC), Barry B. Mook (HagaZiekenhuis), Maarten J. Titulaer (Erasmus MC), Kiril E.B. van Veen (Alrijne Ziekenhuis), Yannick Resok (Haaglanden Medisch Centrum).
Funding
Supported by grants from the European Research Council (ERC Consolidator grant 101001237 to MB), and the Netherlands Organization for Health Research and Development (Vici grant number 918.19.627 to DvdB; Vidi grant number 917.17.308 to MB).
Author information
Authors and Affiliations
Consortia
Contributions
SS: methodology, data collection, data analysis, data interpretation, and writing of the original draft of the manuscript; SO: data collection, review and editing of the report; LtH: data collection, review and editing of the report; IvZ: study design, data collection, review and editing of the report; DvdB: review, editing and supervision of the report; MB: methodology, study design, data interpretation, review, editing and supervision of the report, and funding acquisition. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The study was approved by the biobank ethics committee of the Amsterdam UMC, location AMC, Amsterdam, The Netherlands (number BTC AMC2014_290).
Consent for publication
Not applicable.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Staal, S.L., Olie, S.E., ter Horst, L. et al. Granulocytes in cerebrospinal fluid of adults suspected of a central nervous system infection: a prospective study of diagnostic accuracy. Infection 52, 1415–1423 (2024). https://doi.org/10.1007/s15010-024-02200-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-024-02200-5