Abstract
Purpose
In late 2022, a surge of severe S. pyogenes infections was reported in several European countries. This study assessed hospitalizations and disease severity of community-acquired bacterial infections with S. pyogenes, S. pneumoniae, N. meningitidis, and H. influenzae among children in North Rhine-Westphalia (NRW), Germany, during the last quarter of 2022 compared to long-term incidences.
Methods
Hospital cases due to bacterial infections between October and December 2022 were collected in a multicenter study (MC) from 59/62 (95%) children's hospitals in NRW and combined with surveillance data (2016–2023) from the national reference laboratories for streptococci, N. meningitidis, and H. influenzae. Overall and pathogen-specific incidence rates (IR) from January 2016 to March 2023 were estimated via capture–recapture analyses. Expected annual deaths from the studied pathogens were calculated from national death cause statistics.
Results
In the MC study, 153 cases with high overall disease severity were reported with pneumonia being most common (59%, n = 91). IRs of bacterial infections declined at the beginning of the COVID-19 pandemic and massively surged to unprecedented levels in late 2022 and early 2023 (overall hospitalizations 3.5-fold), with S. pyogenes and S. pneumoniae as main drivers (18-fold and threefold). Observed deaths during the study period exceeded the expected number for the entire year in NRW by far (7 vs. 0.9).
Discussion
The unprecedented peak of bacterial infections and deaths in late 2022 and early 2023 was caused mainly by S. pyogenes and S. pneumoniae. Improved precautionary measures are needed to attenuate future outbreaks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the outbreak of the COVID-19 pandemic in 2020, children’s and adolescents’ health has been challenged in numerous ways. Among others, a decline of bacterial infections and distorted periodicity of seasonal infection waves were observed across the world and affected all age groups [1,2,3,4,5]. An unintended result of reduced infections is the lack of natural immunity, especially in the youngest.
Beginning in late fall 2022, rising numbers of severe infections by group A streptococci were reported from Spain, France, Great Britain, and other countries [6,7,8,9]. At the same time, a massive wave of acute viral and bacterial infections caused a near-collapse of the German paediatric health care system including out- and inpatient sectors and overwhelming pediatric intensive care capacities [10,11,12,13]. Pediatric health care providers in Germany claimed this was an unprecedented public health emergency [10,11,12].
To verify the self-declared public health emergency from bacterial infections, we conducted this study. It was designed to assess hospitalizations due to community-acquired bacterial infections by Streptococcus pyogenes (S. pyogenes), Streptococcus pneumoniae (S. pneumoniae), Neisseria meningitidis (N. meningitidis), and Haemophilus influenzae (H. influenzae) in the last quarter of 2022 compared to seasonally expected infection waves since 2016.
Methods
Study design
This is a retrospective multicenter study (MC) on community-acquired bacterial infections requiring hospitalization in children and adolescents in North Rhine-Westphalia/Germany (NRW) between October 1st and December 31st of 2022. The results were combined with four data sources to deduce long-term trends.
(1) For the MC study, clinical data were collected from 59 of 62 (95%) children’s hospitals in NRW. (2) Surveillance data from the German reference laboratory for streptococci (GRLS) and the national reference laboratory for N. meningitidis and H. influenzae (NRLMHi) were matched with cases from the MC study for capture–recapture (CRC) analyses. (3) Monthly reference laboratory-reported incidences from January 2016 to March 2023 were combined with population statistics. (4) The expected number of deaths from bacterial infections per year in children < 15 years was calculated from national death cause statistics.
Multicenter study
Study population
Children aged > 27 days and < 18 years admitted to a children’s hospital in NRW during the study period due to a community-acquired infection with S. pyogenes, S. pneumoniae, N. meningitidis, or H. influenzae were eligible, including invasive and non-invasive infections but no catheter-associated or nosocomial infections.
Clinical data collection
Eligible patients were identified via positive test results for the pathogen of interest by the local microbiology departments or derived from diagnose related group codes (International Classification of Diseases, 10th revision, German modification, ICD-10-GM). Eligibility was confirmed via retrospective chart review by the local investigators and de-identified clinical data entered into web-based case report forms at www.limesurvey.org.
Central nervous system (CNS) infections were defined as infections limited to the CNS (e.g., cerebrospinal fluid, intracranial abscess/empyema). Eye and ear–nose–throat (E+ENT) infections were defined as infections primarily located in the upper respiratory tract down to the larynx, including paranasal sinuses, Eustachian tubes, middle ear, mastoid cavities, and orbita. E+ENT with cerebral invasion were defined as primary E+ENT infections with penetration into the CNS.
Concomitant viral infections were diagnosed by the local laboratories via swabs from the respiratory tract by polymerase chain reaction or antigen testing. No information was available on viral infections preceding admission and vaccination status.
In cases with detection of multiple pathogens or non-invasive infection, the relevant pathogen was determined by a pediatric infectious disease specialist (S.G.) and a pediatric intensivist with additional qualification as infectious disease specialist (C.D.S.) according to clinical and laboratory findings. Pneumonia was confirmed either from blood culture, pleural empyema or tracheal secretion/bronchoalveolar lavage fluid. In cases with adequate antibiotic treatment prior to hospitalization, pneumonia was also accepted if radiologic signs were present and another mode of detection was positive for the respective pathogen, e.g., pneumococcal antigen in urine or S. pyogenes antigen in throat swabs.
Duplicate reports in the dataset originating from inter-center referrals were removed after updating the total duration of hospital stay.
Children’s hospital capacities of NRW
Each participating hospital reported the number of non-surgical beds on pediatric wards (including neonatal and pediatric intensive care units (ICU)). Surgical beds that are not strictly designated for surgical patients but can be used for either surgical or non-surgical patients as needed were counted as pediatric. The capacities of the three non-participating centers were obtained by inquiry to the respective children’s hospital.
Surveillance of invasive bacterial infections
Reporting of invasive infections by the studied pathogens is mandatory except for S. pyogenes (Supplementary Table S1). Incidences used in this study are from NRW cases only. Serotypes of S. pneumoniae and H. influenzae and serogroups of N. meningitidis were provided by the nationwide reference laboratories (NRL) to assess vaccine-preventability of invasive infections in two time periods: 2016–2021 and 2022–03/2023.
Population and death cause statistics
Nationwide and NRW mid-year populations for cases < 18 years of age from 2016 to 2022 were derived from the Federal Statistical Office (FSO). Annual deaths from streptococci, N. meningitidis, and H. influenzae infections from 2016 to 2021 were extracted via ICD-10-GM codes from national death cause statistics provided by the FSO (Supplementary Table S2). Because age grouping at the FSO is available in 5-year categories, expected deaths were calculated only for children < 15 years of age. The number of expected deaths per year in NRW was extrapolated from the proportion of children living in NRW compared to the total pediatric population of Germany (22%).
Linkage of clinical and surveillance data
Clinical MC and NRL surveillance data of S. pyogenes, S. pneumoniae, N. meningitidis and H. influenzae were matched by patient’s age, sex, sample collection site and date, identified pathogen, and serotype if applicable (full agreement of all items was considered a match). Direct record linkage by name or date of birth was inapplicable due to de-identification and prohibited by general data protection regulations.
Statistical analyses
Continuous variables are presented as means with 95% confidence intervals (CI) if evenly distributed and as median with interquartile range (IQR) if skewed. Discrete variables are presented as counts and percent. The dataset of the MC study contained no missing data.
Capture–recapture analyses (CRC) were conducted using a generalized log-linear model. CRC analysis is a statistical method used to estimate the size of a population by capturing, marking, and releasing individuals, and then recapturing a portion of them later. It involves comparing the marked and unmarked individuals in the recaptured sample to make inferences about the total population size. In this study, we matched the cases from MC study and the cases reported to the NRLs to identify the overlap (= recaptured cases) and estimate the total number of severe infections for each respective pathogen.
Monthly overall and pathogen-specific incidence rates per 100.000 child years (CY) were calculated from total case numbers estimated by CRC analyses assuming a consistent reporting rate over time. Monthly incidence rate ratios (IRR) were calculated for the period from January 2020 to March 2023, with the corresponding months of 2016–2019 as references. Besides pathogen-specific incidence rates and IRRs, cumulative monthly incidence rates and IRRs were calculated.
The expected number of deaths from the studied pathogens in cases < 15 years of age was calculated from the average number of annual deaths in Germany and the fraction of children living in NRW.
SAS Enterprise Guide 8.3 (SAS Institute Inc., Cary, NC, USA) was used to perform statistical analyses and produce figures.
Ethics approval
The study was approved by the Ethics Committee of the Medical Faculty of the University of Duisburg-Essen (22-11045-BO).
Results
Fifty-nine of 62 (95%) children’s hospitals in NRW participated in the MC study, comprising 4066 of 4323 (94%) pediatric hospital beds. At the end of 2022, 22% of Germany’s children lived in NRW. Subtracting the 6% capacities from non-participating hospitals, the participating centers supplied hospital care for approximately 20.7% of German children.
A total of 153 cases were reported with a median age of 4 years (IQR 1–7). Fifty-eight (38%) cases were from S. pyogenes, 62 (41%) from S. pneumoniae, 2 (1%) from N. meningitidis, and 19 (12%) from H. influenzae (Table 1). The most frequent presentation was pneumonia (n = 91, 59%), followed by sepsis/systemic inflammatory response syndrome/toxic shock syndrome (n = 28, 18%), skin or soft tissue infections (n = 22, 14%), and E+ENT infection without cerebral invasion (n = 22, 14%). Viral co-infections were observed in 71 (46%) cases. ICU admissions were frequent (n = 90, 59%), as was mechanical ventilation (n = 67, 44%), surgical source control (n = 41, 27%), and use of vasopressors or inotropes (n = 26, 17%) (Table 1, Fig. 1). Eight children died (5%), and five (3%) were discharged to a rehabilitation or care facility. Overall functional neurological outcome at hospital discharge in survivors was good (median pediatric cerebral performance category (PCPC) = 1, IQR 1–2). The sites of pathogen detection in the MC study and the proportion of invasive infections varied between the pathogens, with the highest absolute burden of invasive infections caused by S. pyogenes and S. pneumoniae (Table 2).
Hospital-treated community-acquired bacterial infections in children in North Rhine-Westphalia in the last quarter of 2022. a Clinical presentation. b Treatment and outcomes. SIRS systemic inflammatory response syndrome, TSS toxic shock syndrome, ENT ear–nose–throat, CNS central nervous system, ICU intensive care unit, CPR cardiopulmonary resuscitation
Among NRL-reported cases of invasive infections, the degree of vaccine-preventability varied depending on the pathogen and changed over time (Table 3). In S. pneumoniae, the proportion of vaccine-preventable cases increased, whereas they decreased in H. influenzae and N. meningitidis.
CRC analyses showed different degrees of overlap between the MC study and NRL data. For N. meningitidis, capture by the NRL was complete, but the total incidence (n = 6, sporadic infections—no outbreak) was low compared to the estimated total numbers of S. pneumoniae (nestimated = 232), S. pyogenes (nestimated = 214), and H. influenzae (nestimated = 95) (Table 4).
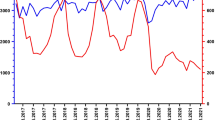
With onset of the COVID-19 pandemic, incidence rates and IRRs exhibited a decline, followed by a pathogen-specific degree of recovery up to or above pre-pandemic levels (Figs. 2 and S1 and Supplementary Table S2). Alike, cumulative monthly incidence rates and IRRs declined at the beginning of the pandemic, showed small peaks during the course of the pandemic, and peaked in December 2022 and February 2023 (Fig. 3 and Supplementary Tables S2 and S3).
Hospitalizations for community-acquired bacterial infections. a S. pyogenes. b S. pneumoniae. c N. meningitidis. d H. influenzae. Red solid line: incidence rates January 2020–March 2023; black dashed line and grey band: average monthly incidence rates 2016–2019 with 95% CI; green band: period of MC study
Hospitalizations due to acquired-bacterial infections from S. pyogenes, S. pneumoniae, N. meningitidis, and H. influenzae, January 2020–March 2023. a Cumulative hospitalization rates of S. pneumoniae (dark grey), S. pyogenes (medium grey), and N. meningitidis + H. influenzae (light grey). b IRRs of infections, reference period 2016–2019. Green band: period of MC study
The reported number of deaths in children < 15 years in the MC study exceeded the expected number of annual deaths from S. pyogenes and S. pneumoniae (7 observed; 0.9 expected). No deaths were caused by N. meningitidis (1.8 expected) and H. influenzae (0 expected) (Table 5). The death from H. influenzae in the MC study occurred in a patient aged 17 years and is, therefore, not included in this statistic.
Discussion
This multi-source study in Germany’s most populous federal state detected high incidences of pediatric general ward and pediatric ICU admissions for bacterial infections associated with S. pyogenes, S. pneumoniae, N. meningitidis, and H. influenzae in the last quarter of 2022. Disease severity was high with more than half of the cases admitted to an ICU and frequent requirement of invasive mechanical ventilation and vasoactive agents. Longitudinal assessment since 2016 revealed an unprecedented peak of bacterial infection-related hospitalizations in late 2022 and early 2023, primarily driven by S. pyogenes and S. pneumoniae. Deaths during the 3-month MC study period were seven times higher than expected for the entire year.
A major finding that has already been reported in adults and children [14, 15] was the marked overall decline of severe and invasive bacterial infections at the beginning of the COVID-19 pandemic. In our study, this overall decline was shortly interrupted in fall 2021 (when schools and day care facilities re-opened) but otherwise lasted until early summer 2022, coinciding with the timepoint when compulsory wearing of protective face masks was abrogated.
A decline of S. pyogenes infections from the onset of the pandemic until spring 2022 was also reported in France, followed by a sudden increase of non-invasive S. pyogenes infections [16]. Like Germany, France and England also witnessed strong increases of severe and invasive S. pyogenes infections during fall and winter of 2022 [7, 17]. These surges were most likely induced by an overall rise in infection numbers with the proportion of severe cases remaining constant, rather than changes in types, virulence or toxicity [7, 18,19,20]. A possible explanation is the discontinuation of face mask wearing among children, which re-enabled droplet transmission of S. pyogenes, an essential mechanism of infection [21]. High prevalence of acute respiratory virus infections as reported by the Robert Koch-Institute at the height of the S. pyogenes wave [22] may have paved the way for bacterial superinfections. Superinfections are described for influenza virus in combination with S. pyogenes and S. pneumoniae [23] and were also observed in our study.
Our study found viral co-infections in 60% of the S. pneumoniae cases, most commonly by influenza and respiratory syncytial virus. In line with this finding, asymptomatic S. pneumoniae carriage and respiratory virus infection as starting point for invasive pneumococcal disease is well-acknowledged [24]. During the pandemic, the decline in pneumococcal infections in Germany was less prominent compared to S. pyogenes infections, possibly due to viral infection waves among children in spite of pandemic measures [22]. In contrast to S. pyogenes infections, invasive pneumococcal disease can be effectively prevented by vaccination [25], necessitating high levels of vaccination rates to attenuate virus-triggered outbreaks. We were not able to assess the vaccination status of the included cases in the MC study but the cases registered by the NRL show an increase of invasive pneumococcal infections by vaccine-preventable serotypes. This finding aligns with reports that the German population is incompletely vaccinated against pneumococci: Pneumococcal vaccination coverage for children entering primary school increased from 15% in 2010 to 83% in 2017–2019 but is only 2% in adolescents and adults [26, 27]. Of infants born in 2016, only 73% received a full series of the recommended pneumococcal vaccination scheme and vaccinations were frequently delayed [28]. This low vaccination coverage and the observed increase of invasive infections from the serotypes recommended for basic immunization in infants by the German Standing Committee on Vaccination (STIKO) calls for urgent improvement to enhance protection against opportunistic pneumococcal infections. Additionally, vaccine coverage against influenza, SARS-CoV-2, and respiratory syncytial virus should be optimized to reduce the susceptibility to bacterial and superinfections.
Vaccines are also responsible for decreasing incidences of meningococcal infections in several countries [29]. In Germany, vaccination is recommended for serogroup C with a subsequent decline of serogroup C infections since its introduction [30]. The trend of decreasing meningococcal disease was further enhanced during the COVID-19 pandemic [14] and also noticeable in our study with incidence rates below pre-pandemic average. Yet, in March 2023, an increase in meningococcal infection incidence above average was observed but characterized by very low absolute numbers. In contrast to S. pneumoniae, vaccine coverage against meningococcal serogroup C in school starters in Germany is above 90% [27]. Alike, infections reported to the NRLMHi by serogroup C were rare. The majority was caused by serogroup B, which is also vaccine-preventable but not officially recommended by the STIKO and thus not covered by health insurances. Thus, coverage of serogroup B vaccination by health insurances represents a possible lever to reduce meningococcal infections in Germany even further.
For H. influenzae, a decline of invasive infections during the COVID-19 pandemic was observed in the Netherlands but associated with an increase of vaccine-preventable serotype b [31]. In our study, H. influenzae hospitalizations during the pandemic remained within seasonally expected ranges, showing only two peaks in December of 2021 and 2022, respectively. The NRLMHi recorded only 2 cases by serotype b, in line with the high vaccine coverage of 90% in German children against H. influenzae b [27]. Yet, IRRs remained almost constantly above one since June 2021, indicating a slight increase of non-vaccine H. influenzae infections, which contribute to the overall burden of pediatric hospitalizations.
Limitations of the study mainly derive from missing cases and information: Due to the incomplete coverage of children’s hospitals in NRW, the MC study missed 6% of pediatric hospital capacities. Cases treated outside of pediatric units, such as otorhinolaryngology or adult units, and pre-hospital deaths could not be captured, potentially leading to underestimation of incidences and disease severity. Variable pathogen-specific underreporting to the NRLs occurred, as commonly observed in infectious disease surveillance [32,33,34]. However, awareness of streptococcal infections due to reports from other countries may have increased reporting rates, causing overestimation of the incidence peak. Another limitation is the lack of information on viral infections preceding admission and vaccination status. Because it was not recorded if and which viral swabs patients received, we may have underestimated the proportion of viral co-infections.
In summary, the acute overload of the German pediatric health care system was caused by a rebound of acute viral and bacterial respiratory and invasive diseases that peaked at the same time, creating an unprecedented incidence of hospitalizations in children. Shortages of essential drugs, such as antibiotics and antipyretics, in the outpatient sector [35, 36] and a decrease in numbers of operable beds in children’s hospitals due to personnel shortages [37, 38] aggravated the pediatric health care crisis.
The results of this study indicate that unintended effects from preventive measures against the COVID-19 pandemic potentially continue to burden children’s health. While this is well-acknowledged for mental health and obesity, which directly increase morbidity but not mortality [39,40,41,42], the drastic increase of severe bacterial infections is new and its origin deserves deeper investigation. Further research should untangle this amalgam to quantify excess morbidity and mortality from acute bacterial infections themselves and the lack of adequate pediatric care capacities in Germany. However, even more urgent than scientific workup, is the implementation of effective measures against future outbreaks among children to prevent morbidity and deaths. These include high vaccination coverage and acquisition of natural immunity against both viral and bacterial disease, sufficient pediatric drug supply, and pediatric health care capacities adapted to seasonal demands.
Data availability
The data generated for this study are available without undue reservation to any qualified researcher upon reasonable request.
References
Eden JS, Sikazwe C, Xie R, Deng YM, Sullivan SG, Michie A, et al. Off-season RSV epidemics in Australia after easing of COVID-19 restrictions. Nat Commun. 2022;13(1):2884. https://doi.org/10.1038/s41467-022-30485-3.
Rao S, Armistead I, Messacar K, Alden NB, Schmoll E, Austin E, et al. Shifting epidemiology and severity of respiratory syncytial virus in children during the COVID-19 pandemic. JAMA Pediatr. 2023. https://doi.org/10.1001/jamapediatrics.2023.1088.
Farfour E, Clichet V, Péan de Ponfilly G, Carbonnelle E, Vasse M. Impact of COVID-19 pandemic on blood culture practices and bacteremia epidemiology. Diagn Microbiol Infect Dis. 2023;107(1):116002. https://doi.org/10.1016/j.diagmicrobio.2023.116002.
Ricketson LJ, Kellner JD. Changes in the Incidence of Invasive Pneumococcal Disease in Calgary, Canada, during the SARS-CoV-2 Pandemic 2020–2022. Microorganisms. 2023. https://doi.org/10.3390/microorganisms11051333.
Amarsy R, Robert J, Jarlier V. Impact of the first year of the COVID-19 pandemic on the epidemiology of invasive infections (bacteremia) in the hospitals of the Assistance Publique-Hôpitaux de Paris. Bull Acad Natl Med. 2023;207(2):131–5. https://doi.org/10.1016/j.banm.2022.11.013.
Cobo-Vázquez E, Aguilera-Alonso D, Carrasco-Colom J, Calvo C, Saavedra-Lozano J. Increasing incidence and severity of invasive Group A streptococcal disease in Spanish children in 2019–2022. Lancet Reg Health Eur. 2023;27: 100597. https://doi.org/10.1016/j.lanepe.2023.100597.
Guy R, Henderson KL, Coelho J, Hughes H, Mason EL, Gerver SM, et al. Increase in invasive group A streptococcal infection notifications, England, 2022. Euro Surveill. 2023. https://doi.org/10.2807/1560-7917.Es.2023.28.1.2200942.
Lassoued Y, Assad Z, Ouldali N, Caseris M, Mariani P, Birgy A, et al. Unexpected increase in invasive group A streptococcal infections in children after respiratory viruses outbreak in France: a 15-year time-series analysis. Open Forum Infect Dis. 2023;10(5):ofad188. https://doi.org/10.1093/ofid/ofad188.
Organization WH. Increased incidence of scarlet fever and invasive Group A Streptococcus infection - multi-country. (2022). https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON429. Accessed 15 July 2022.
Recent hospital survey reveals: On average, no available intensive care bed for critically ill children - Emergency physicians demand new structures. www.divi.de2022.
https://www.dw.com/en/germany-rsv-infection-wave-overloads-childrens-hospitals/a-63963430. www.dw.com2023. Accessed 4 July 2023
Children are dying because we cannot provide them with care. www.welt.de: Welt; 2022. Accessed 4 July 2023
Heimberg E, Pratsch A, Neunhoeffer F, Olivieri M, Hoffmann F. Defizitäre Versorgungsstrukturen treffen vor allem die großen pädiatrischen Intensivstationen – eine longitudinale Auswertung. Medizinische Klinik - Intensivmedizin und Notfallmedizin. 2023. https://doi.org/10.1007/s00063-023-01094-9.
Brueggemann AB, Jansen van Rensburg MJ, Shaw D, McCarthy ND, Jolley KA, Maiden MCJ, et al. Changes in the incidence of invasive disease due to Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitidis during the COVID-19 pandemic in 26 countries and territories in the Invasive Respiratory Infection Surveillance Initiative: a prospective analysis of surveillance data. Lancet Digit Health. 2021;3(6):e360–70. https://doi.org/10.1016/s2589-7500(21)00077-7.
McNeil JC, Flores AR, Kaplan SL, Hulten KG. The indirect impact of the SARS-CoV-2 pandemic on invasive group a streptococcus, streptococcus pneumoniae and Staphylococcus aureus infections in Houston area children. Pediatr Infect Dis J. 2021;40(8):e313–6. https://doi.org/10.1097/inf.0000000000003195.
Cohen JF, Rybak A, Werner A, Kochert F, Cahn-Sellem F, Gelbert N, et al. Surveillance of noninvasive group A Streptococcus infections in French ambulatory pediatrics before and during the COVID-19 pandemic: a prospective multicenter study from 2018–2022. Int J Infect Dis. 2023;134:135–41. https://doi.org/10.1016/j.ijid.2023.06.003.
van Kempen EB, Bruijning-Verhagen PCJ, Borensztajn D, Vermont CL, Quaak MSW, Janson J-A, et al. Increase in invasive group a streptococcal infections in children in the Netherlands, a survey among 7 hospitals in 2022. Pediatric Infect Dis J. 2023;42(4):e122–4.
de Gier B, Marchal N, de Beer-Schuurman I, te Wierik M, Hooiveld M, Group I-AS, et al. Increase in invasive group A streptococcal (Streptococcus pyogenes) infections (iGAS) in young children in the Netherlands, 2022. Eurosurveillance. 2023;28(1):2200941. https://doi.org/10.2807/1560-7917.ES.2023.28.1.2200941.
Johannesen TB, Munkstrup C, Edslev SM, Baig S, Nielsen S, Funk T, et al. Increase in invasive group A streptococcal infections and emergence of novel, rapidly expanding sub-lineage of the virulent Streptococcus pyogenes M1 clone, Denmark, 2023. Eurosurveillance. 2023;28(26):2300291. https://doi.org/10.2807/1560-7917.ES.2023.28.26.2300291.
Robert-Koch-Institut. Update: Anstieg bakterieller Infektionen durch Gruppe-A- Streptokokken, Pneumokokken und Haemophilus influenzae in Deutschland seit Ende 2022. Epidemiologisches Bulletin. Berlin: Robert-Koch-Institut; 2023. p. 45.
Avire NJ, Whiley H, Ross K. A review of Streptococcus pyogenes: Public health risk factors, prevention and control. Pathogens. 2021. https://doi.org/10.3390/pathogens10020248.
Buda S, Dürrwald R, Biere B, Reiche J, Buchholz U, Tolksdorf K, et al. ARE-Wochenbericht KW 25/2023. Arbeitsgemeinschaft Influenza – Robert Koch-Institut. 2023. https://doi.org/10.25646/11563.
Oliver J, Thielemans E, McMinn A, Baker C, Britton PN, Clark JE, et al. Invasive group A Streptococcus disease in Australian children: 2016 to 2018—a descriptive cohort study. BMC Public Health. 2019;19(1):1750. https://doi.org/10.1186/s12889-019-8085-2.
Danino D, Ben-Shimol S, van der Beek BA, Givon-Lavi N, Avni YS, Greenberg D, et al. Decline in pneumococcal disease in young children during the coronavirus disease 2019 (COVID-19) pandemic in Israel associated with suppression of seasonal respiratory viruses, despite persistent pneumococcal carriage: a prospective cohort study. Clin Infect Dis. 2022;75(1):e1154–64. https://doi.org/10.1093/cid/ciab1014.
Chesdachai S, Graden AR, DeSimone DC, Weaver AL, Baddour LM, Joshi AY. Changing trends of invasive pneumococcal disease in the era of conjugate pneumococcal vaccination in Olmsted county: a population-based study. Mayo Clin Proc. 2022;97(12):2304–13. https://doi.org/10.1016/j.mayocp.2022.06.037.
Deb A, Podmore B, Barnett R, Beier D, Galetzka W, Qizilbash N, et al. Pneumococcal vaccination coverage in individuals (16–59 years) with a newly diagnosed risk condition in Germany. BMC Infect Dis. 2022;22(1):753. https://doi.org/10.1186/s12879-022-07736-1.
Vaccination coverage of children presenting their vaccination card at school entry health examinations. 2023. https://www.gbe-bund.de/gbe/pkg_isgbe5.prc_menu_olap?p_uid=gast&p_aid=34430128&p_sprache=E&p_help=0&p_indnr=831&p_indsp=27110398&p_ityp=H&p_fid=. Accessed 2 July 2023.
Laurenz M, von Eiff C, Borchert K, Jacob C, Seidel K, Schley K. Vaccination rates and adherence in pneumococcal conjugate vaccination in mature born infants before and after vaccination schedule change – A claims database analysis. Vaccine. 2021;39(24):3287–95. https://doi.org/10.1016/j.vaccine.2021.04.029.
Nuttens C, Findlow J, Balmer P, Swerdlow DL, Tin Tin Htar M. Evolution of invasive meningococcal disease epidemiology in Europe, 2008 to 2017. Euro Surveill. 2022. https://doi.org/10.2807/1560-7917.Es.2022.27.3.2002075.
Robert-Koch-Institut. Invasive Meningokokken-Erkrankungen 2012–2015. Epidemiologisches Bulletin. Berlin: Robert-Koch-Institut; 2016. p. 18.
Steens A, Knol MJ, Freudenburg-de Graaf W, de Melker HE, van der Ende A, van Sorge NM. Pathogen- and type-specific changes in invasive bacterial disease epidemiology during the first year of the COVID-19 PANDEMIC in The Netherlands. Microorganisms. 2022. https://doi.org/10.3390/microorganisms10050972.
Doyle TJ, Glynn MK, Groseclose SL. Completeness of notifiable infectious disease reporting in the United States: an analytical literature review. Am J Epidemiol. 2002;155(9):866–74. https://doi.org/10.1093/aje/155.9.866.
Meadows AJ, Oppenheim B, Guerrero J, Ash B, Badker R, Lam CK, et al. Infectious disease underreporting is predicted by country-level preparedness, politics, and pathogen severity. Health Secur. 2022;20(4):331–8. https://doi.org/10.1089/hs.2021.0197.
Gibbons CL, Mangen M-JJ, Plass D, Havelaar AH, Brooke RJ, Kramarz P, et al. Measuring underreporting and under-ascertainment in infectious disease datasets: a comparison of methods. BMC Public Health. 2014;14(1):147. https://doi.org/10.1186/1471-2458-14-147.
Shachar C, Gruppuso PA, Adashi EY. Pediatric drug and other shortages in the age of supply chain disruption. JAMA. 2023;329(24):2127–8. https://doi.org/10.1001/jama.2023.4755.
Reali L, Mendie C, Fischbach T, Werner A, Leitner AY, Karall D, et al. Open letter: The health of our children and adolescents is endangered throughout Europe due to the medication shortage. www.bvkj.de2023. Accessed 4 July 2023
Parvu V. Alarming study results: Physicians warn of supply crisis in German children's hospitals. www.divi.de2019. Accessed 4 July 2023
DIVI criticizes staff shortage in pediatric intensive care units in Germany. www.divi.de2018. Accessed 4 July 2023
Chang TH, Chen YC, Chen WY, Chen CY, Hsu WY, Chou Y, et al. Weight Gain Associated with COVID-19 Lockdown in Children and Adolescents: A Systematic Review and Meta-Analysis. Nutrients. 2021. https://doi.org/10.3390/nu13103668.
Viner R, Russell S, Saulle R, Croker H, Stansfield C, Packer J, et al. School closures during social lockdown and mental health, health behaviors, and well-being among children and adolescents during the first COVID-19 wave: a systematic review. JAMA Pediatr. 2022;176:400–9. https://doi.org/10.1001/jamapediatrics.2021.5840.
Bruns N, Willemsen L, Stang A, Kowall B, Holtkamp K, Kamp O, et al. Pediatric intensive care unit admissions after adolescent suicide attempts during the pandemic. Pediatrics. 2022. https://doi.org/10.1542/peds.2021-055973.
Meherali S, Punjani N, Louie-Poon S, Abdul Rahim K, Das JK, Salam RA, et al. Mental health of children and adolescents amidst COVID-19 and past pandemics: a rapid systematic review. Int J Environ Res Public Health. 2021. https://doi.org/10.3390/ijerph18073432.
Acknowledgements
We thank Christina Pentek for her support.
Funding
Open Access funding enabled and organized by Projekt DEAL. This research received no external funding. The surveillance of invasive streptococcal infections in Germany at the German National Reference Laboratory for Streptococci was funded by the Robert Koch Institute, Berlin, Germany (funding number: 1369–235). The funder had no role in the design and conduct of the study.
Author information
Authors and Affiliations
Contributions
SCG and NB conceptualized and designed the study, carried out analyses, drafted the initial manuscript, and critically reviewed and revised the manuscript. TH, ROA, FAE, MA, J-CB, MAB, TB, MB, MB, FB, RC, CD, FD, JD, JD, FE, NE, ME, HF, MG, BG, CG, EH, YH, KH, H-GH, MH, GH, HK, CK-R, Pascal Lenz, Klaus Lohmeier, Andreas Müller, Frank Niemann, Michael Paulussen, FP, RP, MP, PR, TR, JR, HS, RS, PS, JNS, TS, PS, GS, US, CT, AT, BU, AvdH, TvH, VG, SW, TW, DW and GE, KOH, AL, collected data and critically reviewed and revised the manuscript. MvdL, AI, HC and T-TL collected data, carried analyses, and critically reviewed and revised the manuscript. UF-M and CD-S conceptualized and designed the study, coordinated and supervised data collection, and critically reviewed and revised the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The other authors have no conflicts of interest to disclose.
Supplementary Information
Below is the link to the electronic supplementary material.
15010_2023_2165_MOESM1_ESM.png
Figure S1. Monthly incidence rate ratios of invasive bacterial infections in children in North Rhine-Westphalia January 2020–March 2023 (reference period 2016–2019). a) S. pyogenes. b) S. pneumoniae. c) N. meningitidis. d) H. influenzae
15010_2023_2165_MOESM2_ESM.xlsx
Supplementary table 3 (XLSX 23 KB). Excel file containing monthly NRW case numbers, incidence rates, and incidence rate ratios with 95 % confidence intervals from the national reference laboratories
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Goretzki, S.C., van der Linden, M., Itzek, A. et al. Outbreak of severe community-acquired bacterial infections among children in North Rhine-Westphalia (Germany), October to December 2022. Infection 52, 1099–1111 (2024). https://doi.org/10.1007/s15010-023-02165-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-023-02165-x