Abstract
Purpose
We hypothesized that SARS-CoV-2 infection numbers reported by governmental institutions are underestimated due to high dark figures as only results from polymerase chain reaction (PCR) tests are incorporated in governmental statistics and testing capacities were further restricted as of July, 2022.
Methods
A point prevalence investigation was piloted by rapid antigen testing (RAT) among participants of the VACCELERATE volunteer registry. 2400 volunteers were contacted, of which 500 received a RAT including instructions for self-testing in the first week of July, 2022. Results were self-reported via e-mail.
Results
419 valid RAT results were collected until July 7th, 2022. Between July-1 and July-7, 2022, 7/419 (1.67%) tests were positive. Compared to reports of the German Federal Government, our results suggest a more than twofold higher prevalence. Three out of seven positive individuals did not have a PCR test and are therefore likely not to be displayed in governmental statistics.
Conclusion
Our findings imply that the actual prevalence of SARS-CoV-2 may be higher than detected by current surveillance systems, so that current pandemic surveillance and testing strategies may be adapted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With the global surge of Severe Acute Respiratory Syndrome-Coronavirus-2 (SARS-CoV-2) Omicron subvariants BA.4 and BA.5, its enhanced antibody escape, and reduced infection control and prevention measures in most countries, infections due to SARS-CoV-2 rose also among vaccinated and recovered individuals [1, 2]. At the same time, hospitalizations and death rates did not seem to rise significantly, meaning that the infections do not affect most people severely, but still influence people’s daily lives and cause massive work absences [3].
In Germany, the reported incidence of laboratory-based diagnosis, i.e., primarily via polymerase chain reaction (PCR) of SARS-CoV-2 infections increased rapidly since the beginning of June, 2022 [4]. Epidemiological predominance of the Omicron BA.5 subvariant was noted from the end of the same month, while the variant-of-concern (VOC) Omicron has been circulating with a share of 99% among the general population already for more than five months [5]. The numbers reported by the Federal Government are likely to differ substantially from the real incidence as most SARS-CoV-2-infected individuals are not tested via PCR anymore according to the national testing strategy by the Federal Ministry of Health (MOH) [6]. They were rather diagnosed by rapid antigen test (RAT), which may not be added to the national statistics.
We aimed to describe the point prevalence of SARS-CoV-2 infections among a representative random sample of volunteers in Germany, to elucidate a potential underreporting in notified numbers of the RKI or MOH, and to estimate the actual prevalence of SARS-CoV-2 infections.
Methods and results
The VACCELERATE Volunteer Registry is an online registry under the umbrella of the VACCELERATE consortium, where people interested in participating in SARS-CoV-2 clinical studies can sign up via an online survey (www.vaccelerate.eu/volunteer-registry) [7]. The VACCELERATE Volunteer Registry was approved by the Ethics Committee of the Medical Faculty of the University of Cologne (Cologne, Germany) (Study number 20–1536) and currently is active in 15 European countries.
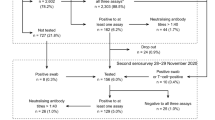
Among those registered in Germany, 32,962 volunteers ≥ 12 years were randomly selected with same odds for each registered participant to be shortlisted and selected for this evaluation (0.073%). Selected volunteers were invited to participate in this survey. A sample of 2400 registered individuals ≥ 12 years of age was contacted via email between June 23rd and 25th, 2022 (Fig. 1).
If a volunteer desired to participate, postal address was collected to allow sending of a RAT kit. Then, a SARS-CoV-2 rapid test (“NEWGENE COVID-19 Antigen Test Kit Schnelltest”, COVID-19-NG21, New Gene Bioengineering Co. Ldt., Hangzhou, People’s Republic of China; sensitivity of 100% with Ct ≤ 25, manufacturer specificity 99.2% [8]) was sent with instructions on how to self-test properly on the day of delivery. Five-hundred test-kits were sent on June 30th, 2022.
Results were then self-reported via email and collected until July 8th, 2022 and, therefore, resemble a point prevalence of July 1st–July 7th, 2022. Overall, 57% of participants were female, median age of participants was 44 years (Table 1). Up to this point, 419 participants had submitted a result, among those, seven participants tested positive for SARS-COV-2 via RAT (7/419, 1.67%). Six RAT test kits were damaged and, thus, unevaluable or were non-readable.
Among the SARS-CoV-2-positive individuals, information was collected on whether infection was already known and whether a PCR result had been performed to calculate any discrepancy between our survey and the governmental reports. All 7 RAT positive individuals had known about their infection before, 4/7 (57.1%) also had a registered positive PCR result, meaning that 42.8% of participants with a positive RAT were not represented in the federal statistics. Among RAT positive individuals, all 7 had received at least three doses of SARS-CoV-2 vaccine. None of them was hospitalized with COVID-19. Geographic distribution of participants was captured via postal code and showed an even distribution throughout Germany (Fig. 2). Evaluation of regional 7-day incidence compared to the weekly governmental report was not meaningful due to small sample number for each region [9].
Geographical distribution of test results in Germany, July 1st–4th, 2022. Explanation: the high number of participants including four individuals tested positive in the western part of Germany may explained by (1) a higher population density in the Rhein-Ruhr area and (2) by the site of the VACCELERATE Volunteer registry coordination office in Cologne, Germany and, therefore, a locally higher number of registered participants. The comparably high number of participants tested positive (four) in this area may be incidental and cannot be extrapolated from general incidence in Western Germany (state of North Rhine-Westphalia) at the time of this study
We report a point prevalence rate for SARS-CoV-2 infection of 1.67% (7/419, equals 1670 per 100,000 inhabitants) in our dataset. If calculated as a value for a representative random sample of individuals living in Germany, it would describe a 7-day-case number of 1,390,059 (calculated with the inhabitant number of 83,237,124 as of 31-Dec-2021 according to the Federal Statistical Office (Statistisches Bundesamt)). This means a 2.39 times higher number than the reports of the RKI dashboard (https://corona.rki.de/) by July 9th, 2022 (7-day incidence of 700.3 and 7-day case number 582,315).
Discussion and implications
With increasing SARS-CoV-2 infection numbers among immunized individuals due to the Omicron variant, the concurrent antibody escape of the BA.5 subvariant and without any contact precautions in place, SARS-CoV-2 infections spread even more rapidly than previously observed. This affects society with extremely high absolute numbers of simultaneously infected individuals. This can be observed as the current pandemic situation in Germany has affected the so-called critical infrastructure (healthcare, transport sector, industry, agriculture) which is burdened by massive personnel sick leave numbers. Recently, Germany faced noticeable delay of goods and service delivery. At the same time, numbers of hospital admissions and COVID-19 patients in need of intensive care were increasing as of July 8th, 2022 in Germany, aggravating the burden on the health care system [10]. Many infected individuals have been re-infected with the Omicron VOC despite previous SARS-CoV-2 vaccination [11]. Duration of antibody-mediated immunity seems to persist only shortly for SARS-CoV-2, while studies suggest that previous vaccination and infection—a so-called hybrid immunity—seems to protect best from re-infection [12, 13].
Facing this situation, political action regarding infection control prevention measures seems indispensable. With the third change to the National Testing Strategy of the German Federal Ministry of Health, capacities for SARS-CoV-2 testing were aimed to be utilized more precisely. Therefore, RAT will only be free-of-charge for defined risk groups from July 1st, 2022 in Germany [14]. This may further decrease the reliability of the reported SARS-CoV-2 incidence. Otherwise, the possibility to register self-tested RAT results in the governmental database would allow more precise reporting. Recent regional sewage water sampling of SARS-CoV-2 has suggested a twofold higher rate of SARS-CoV-2 infections than the reported incidence [15], which is in line with our finding of a more than twofold higher prevalence.
Policy makers may use pilot projects like ours to adjust strategies, as well as to guide the local or timely implementation of infection prevention measures. In an expansion of our project, larger and thus more representative groups may be tested regularly in the future in point prevalence studies, while at the same time systematic surveillance of sewage water may be helpful to complement real-time pandemic monitoring [16].
Our pilot investigation has some limitations. First, we assessed a point prevalence with a rather low participant number. Second, our investigation excluded children below 12 years of age, who may show different epidemiological patterns. Third, self-testing bears a potential for inferior test performance [17]. Fourth, the geographic distribution of reported test results varies substantially and regional epidemiological patterns regarding prevalence of SARS-CoV-2 infections are not displayed.
To conclude, determination of exact numbers of incidence and prevalence in a rapidly globally spreading disease is almost impossible [18]. Overall, we show that results of random point prevalence studies differ largely from reported governmental data. Such evaluations as well as wastewater analyses may help to determine more precisely a status of the pandemic, when mass testing is not feasible anymore due to capacities or the economic and logistical burden of such testing strategies, or when people infected with SARS-CoV-2 may refrain from confirmatory testing.
References
Tegally H, Moir M, Everatt J, et al. Emergence of SARS-CoV-2 Omicron lineages BA.4 and BA.5 in South Africa. Nat Med. 28(9):1785–1790. https://doi.org/10.1038/s41591-022-01911-2.
Tuekprakhon A, Nutalai R, Dijokaite-Guraliuc A, et al. Antibody escape of SARS-CoV-2 omicron BA.4 and BA.5 from vaccine and BA.1 serum. Cell. 2022;185:2422-2433.e13.
Callaway E. What Omicron’s BA.4 and BA.5 variants mean for the pandemic. Nature. 2022;606(7916):848–9.
Robert Koch-Institut. Wöchentlicher Lagebericht des RKI zur Coronavirus-Krankheit-2019 (COVID-19)-09.06.2022. Robert Koch-Institut: Berlin; 2022.
Robert Koch-Institut. Wöchentlicher Lagebericht des RKI zur Coronavirus-Krankheit-2019 (COVID-19)-23.06.2022. Robert Koch-Institut: Berlin; 2022.
Bundesministerium für Gesundheit, Die Nationale Teststrategie. 2022. https://doi.org/https://www.zusammengegencorona.de/testen/die-nationale-teststrategie/. Accessed 01 Jul 2022.
Salmanton-García J, Stewart FA, Heringer S, et al. VACCELERATE Volunteer Registry: a European study participant database to facilitate clinical trial enrolment. Vaccine. 2022;40:4090–7.
European Commission Directorate-General for Health and Food Safety Public Health, EU Common list of COVID-19 antigen tests 2022. https://doi.org/https://health.ec.europa.eu/system/files/2022-07/covid-19_eu-common-list-antigen-tests_en.pdf
Robert Koch-Institut. Wöchentlicher Lagebericht des RKI zur Coronavirus-Krankheit-2019 (COVID-19)-07.07.2022. Berlin; 2022.
Deutsche Interdisziplinäre Vereinigung für Intensiv- und Notfallmedizin, DIVI Intensivregister Tagesreport vom 08.07.2022; 2022.
Cohen JI, Burbelo PD. Reinfection with SARS-CoV-2: implications for vaccines. Clin Infect Dis. 2021;73:e4223–8.
Pilz S, Theiler-Schwetz V, Trummer C, et al. SARS-CoV-2 reinfections: overview of efficacy and duration of natural and hybrid immunity. Environ Res. 2022;209: 112911.
Feikin DR, Higdon MM, Abu-Raddad LJ, et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: results of a systematic review and meta-regression. Lancet. 2022;399:924–44.
Bundesministerium für Gesundheit. Dritte Verordnung zur Änderung der Coronavirus-Testverordnung. Berlin: Bundesministerium für Gesundheit; 2022.
Prado T, Fumian TM, Mannarino CF, et al. Wastewater-based epidemiology as a useful tool to track SARS-CoV-2 and support public health policies at municipal level in Brazil. Water Res. 2021;191: 116810.
Brumfield KD, Leddy M, Usmani M et al. Microbiome analysis for wastewater surveillance during COVID-19. mBio. 2022; e00591-22.
Lindner AK, Nikolai O, Kausch F et al. Head-to-head comparison of SARS-CoV-2 antigen-detecting rapid test with self-collected nasal swab versus professional-collected nasopharyngeal swab. Eur Respir J. 2021;57:e00591–22.
Estimating global, regional, and national daily and cumulative infections with SARS-CoV-2 through Nov 14, 2021: a statistical analysis. Lancet. 2022;399:2351–2380.
Acknowledgements
We thank Kerstin Albus, Eva Hagemeister, Sarah Heringer, Natalie Irchin, Janina Leckler, Fiona Stewart, and Jan Thielebeule for logistical and administrative support.
Funding
Open Access funding enabled and organized by Projekt DEAL. The German Volunteer Registry receives funding from the German Federal Ministry of Education and Research (Bundesministerium für Bildung und Forschung, Grant no. BMBF01KX2040).
Author information
Authors and Affiliations
Consortia
Contributions
JS: conceptualization, project administration, supervision, methodology, investigation, formal analysis, visualization, writing—original draft, writing—review and editing. JSG: methodology, resources, investigation, formal analysis, visualization, writing—review and editing. BW: resources, investigation, formal analysis, writing—review and editing. CT: investigation, formal analysis, writing—review and editing. CJ: investigation, formal analysis, writing—review and editing. JF: investigation, formal analysis, writing—review and editing. OAC: conceptualization, project administration, resources, supervision, methodology, investigation, formal analysis, writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
JS has received research grants by the Ministry of Education and Research (BMBF) and Basilea Pharmaceuticals Inc.; has received speaker honoraria by Pfizer Inc. and Gilead; has been a consultant to Gilead, Produkt&Markt GmbH, Alvea Vax. and Micron Research, and has received travel grants by German Society for Infectious Diseases (DGI e.V.) and Meta-Alexander Foundation. JSG reports speaker honoraria for Gilead and Pfizer. BW, CT, CJ, JF have nothing to disclose. OAC reports grants or contracts from Amplyx, Basilea, BMBF, Cidara, DZIF, EU-DG RTD (101037867), F2G, Gilead, Matinas, MedPace, MSD, Mundipharma, Octapharma, Pfizer, Scynexis; Consulting fees from Abbvie, Amplyx, Biocon, Biosys, Cidara, Da Volterra, Gilead, Matinas, MedPace, Menarini, Molecular Partners, MSG-ERC, Noxxon, Octapharma, Pardes, PSI, Scynexis, Seres; Honoraria for lectures from Abbott, Al-Jazeera Pharmaceuticals, Astellas, Grupo Biotoscana/United Medical/Knight, Hikma, MedScape, MedUpdate, Merck/MSD, Mylan, Pfizer; Payment for expert testimony from Cidara; Participation on a Data Safety Monitoring Board or Advisory Board from Actelion, Allecra, Cidara, Entasis, IQVIA, Janssen, MedPace, Paratek, PSI, Pulmocide, Shionogi; A patent at the German Patent and Trade Mark Office (DE 10 2021 113 007.7); Other interests from DGHO, DGI, ECMM, ISHAM, MSG-ERC, Wiley.
Ethical statement
The VACCELERATE Volunteer Registry was approved by the Ethics Committee of the Medical Faculty of the University of Cologne (Cologne, Germany) (Study number 20-1536).
Consent to participate
Informed consent to be contacted for this kind of study was obtained from all individual participants included in the study when they registered for the VACCELERATE volunteer registry. Specific Information includign a data protection statement on this specific study within VACCELERATE was provided when participants were contacted.
Consent to publish
All participants were informed about the intention to published anonymized results and consented via their participation.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stemler, J., Salmanton-García, J., Weise, B. et al. A pilot surveillance report of SARS-CoV-2 rapid antigen test results among volunteers in Germany, 1st week of July 2022. Infection 51, 465–469 (2023). https://doi.org/10.1007/s15010-022-01931-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-022-01931-7