Abstract
Background:
Osteoporosis is a metabolic bone disease characterized by low bone density resulting in increased fracture susceptibility. This research was constructed to uncover the potential therapeutic application of osteoblasts transplantation, generated upon culturing male rat bone marrow-derived mesenchymal stem cells (BM-MSCs) in osteogenic medium (OM), OM containing gold (Au-NPs) or gold/hydroxyapatite (Au/HA-NPs) nanoparticles, in ovariectomized rats to counteract osteoporosis.
Methods:
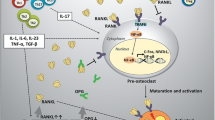
Forty rats were randomized into: (1) negative control, (2) osteoporotic rats, whereas groups (3), (4) and (5) constituted osteoporotic rats treated with osteoblasts yielded from culturing BM-MSCs in OM, OM plus Au-NPs or Au/HA-NPs, respectively. After 3 months, osterix (OSX), bone alkaline phosphatase (BALP), sclerostin (SOST) and bone sialoprotein (BSP) serum levels were assessed. In addition, gene expression levels of cathepsin K, receptor activator of nuclear factor-κb ligand (RANKL), osteoprotegerin (OPG) and RANKL/OPG ratio were evaluated using real-time PCR. Moreover, histological investigation of femur bone tissues in different groups was performed. The homing of implanted osteoblasts to the osteoporotic femur bone of rats was documented by Sex determining region Y gene detection in bone tissue.
Results:
Our results indicated that osteoblasts infusion significantly blunted serum BALP, BSP and SOST levels, while significantly elevated OSX level. Also, they brought about significant down-regulation in gene expression levels of cathepsin K, RANKL and RANKL/OPG ratio versus untreated osteoporotic rats. Additionally, osteoblasts nidation could restore bone histoarchitecture.
Conclusion:
These findings offer scientific evidence that transplanting osteoblasts in osteoporotic rats regains the homeostasis of the bone remodeling cycle, thus providing a promising treatment strategy for primary osteoporosis.



Similar content being viewed by others
References
Compston JE, McClung MR, Leslie WD. Osteoporosis. Lancet. 2019;393:364–76.
Akkawi I, Zmerly H. Osteoporosis: current Concepts. Joints. 2018;6:122–7.
El-Tawab SS, Sabs EK, Elweshahi HM, Ashry MH. Knowledge of osteoporosis among women in Alexandria (Egypt): a community based survey. Egypt Rheumatol. 2015;38:225–31.
Sobeih HS, Abd Elwahed AT. Knowledge and perception of women at risk for osteoporosis: educational intervention. Egypt Nurs J. 2018;15:169–77.
Agrawal VK, Gupta DK. Recent update on osteoporosis. Int J Med Sci Public Health. 2013;2:164–8.
Hossien YE, Tork HMM, El-Sabeely AA. Osteoporosis knowledge among female adolescents in Egypt. Am J NursSci. 2014;3:13–7.
Sözen T, Özışık L, Başaran NÇ. An overview and management of osteoporosis. Eur J Rheumatol. 2017;4:46–56.
Lin X, Xiong D, Peng YQ, Sheng ZF, Wu XY, Wu XP, et al. Epidemiology and management of osteoporosis in the people’s republic of china: current perspectives. Clin Interv Aging. 2015;10:1017–33.
Pino AM, Rosen CJ, Rodríguez JP. In osteoporosis, differentiation of mesenchymal stem cells (MSCs) improves bone marrow adipogenesis. Biol Res. 2012;45:279–87.
Ji MX, Yu Q. Primary osteoporosis in postmenopausal women. Chronic Dis Transl Med. 2015;1:9–13.
Feng X, McDonald JM. Disorders of bone remodeling. Annu Rev Pathol. 2011;6:121–45.
Luhmann T, Germershaus O, Groll J, Meinel L. Bone targeting for the treatment of osteoporosis. J Control Release. 2012;161:198–213.
Kular J, Tickner J, Chim SM, Xu J. An overview of the regulation of bone remodeling at the cellular level. Clin Biochem. 2012;45:863–73.
Perez JR, Kouroupis D, Li DJ, Best TM, Kaplan L, Correa D. Tissue engineering and cell-based therapies for fractures and bone defects. Front Bioeng Biotechnol. 2018;6:105.
Yao W, Lane NE. Target delivery of mesenchymal stem cells to bone. Bone. 2015;70:62–5.
Mahmoud NS, Ahmed HH, Mohamed MR, Amr KS, Aglan HA, Ali MAM, et al. Role of nanoparticles in osteogenic differentiation of bone marrow mesenchymal stem cells. Cytotechnology. 2020;72:1–22.
Yi C, Liu D, Fong CC, Zhang J, Yang M. Gold nanoparticles promote osteogenic differentiation of mesenchymal stem cells through p38 MAPK pathway. ACS Nano. 2010;4:6439–48.
Power RA, Iwaniec UT, Magee KA, Mitova-Caneva NG, Wronski TJ. Basic fibroblast growth factor has rapid bone anabolic effects in ovariectomized rats. Osteoporos Int. 2004;15:716–23.
Ahmed HH, Mahdy EE, Shousha WG, Rashed LA, Abdo SM. Potential role of bone marrow derived mesenchymal stem cells with or without injectable calcium phosphate composite in management of osteoporosis in rat model. Int J Pharm Pharm Sci. 2013;5:494–504.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C(T)) Method. Methods. 2001;25:402–8.
Banchroft JD, Stevens A, Turner DR. Theory and practice of histological techniques. 4th ed. New York: Churchil Livingstone; 1996.
Seibel MJ. Clinical application of biochemical markers of bone turnover. Arq Bras Endocrinol Metabol. 2006;50:603–20.
Sophocleous A, Idris AI. Rodent models of osteoporosis. Bonekey Rep. 2014;3:614.
Wang Z, Goh J, Das De S, Ge Z, Ouyang H, Chong JS, et al. Efficacy of bone marrow-derived stem cells in strengthening osteoporotic bone in a rabbit model. Tissue Eng. 2006;12:1753–61.
Zhou W, Liu Y, Shen J, Yu B, Bai J, Lin J, et al. Melatonin increases bone mass around the prostheses of ovx rats by ameliorating mitochondrial oxidative stress via the SIRT3/SOD2 signaling pathway. Oxid Med Cell Longev. 2019;2019:4019619.
Abuohashish HM, Ahmed MM, Al-Rejaie SS, Eltahir KE. The antidepressant bupropion exerts alleviating properties in an ovariectomized osteoporotic rat model. Acta Pharmacol Sin. 2015;36:209–20.
Kim BJ, Bae SJ, Lee SY, Lee YS, Baek JE, Park SY, et al. TNF-α mediates the stimulation of sclerostin expression in an estrogen-deficient condition. Biochem Biophys Res Commun. 2012;424:170–5.
Shaarawy M, Hasan M. Serum bone sialoprotein: a marker of bone resorption in postmenopausal osteoporosis. Scand J Clin Lab Invest. 2001;61:513–22.
Li CW, Liang B, Shi XL, Wang H. OPG/RANKL mRNA dynamic expression in the bone tissue of ovariectomized rats with osteoporosis. Genet Mol Res. 2015;14:9215–24.
Govindarajan P, Böcker W, El Khassawna T, Kampschulte M, Schlewitz G, Huerter B, et al. Bone matrix, cellularity, and structural changes in a rat model with high-turnover osteoporosis induced by combined ovariectomy and a multiple-deficient diet. Am J Pathol. 2014;184:765–77.
Pfeilschifter J, Köditz R, Pfohl M, Schatz H. Changes in proinflammatory cytokine activity after menopause. Endocr Rev. 2002;23:90–119.
Lu X, Beck GR Jr, Gilbert LC, Camalier CE, Bateman NW, Hood BL, et al. Identification of the homeobox protein Prx1 (MHox, Prrx-1) as a regulator of osterix expression and mediator of tumor necrosis factor alpha action in osteoblast differentiation. J Bone Miner Res. 2011;26:209–19.
Hamdy NA. Targeting the RANK/RANKL/OPG signaling pathway: a novel approach in the management of osteoporosis. Curr Opin Investig Drugs. 2007;8:299–303.
Costa AG, Cusano NE, Silva BC, Cremers S, Bilezikian JP. Cathepsin K: its skeletal actions and role as a therapeutic target in osteoporosis. Nat Rev Rheumatol. 2011;7:447–56.
Armstrong VJ, Muzylak M, Sunters A, Zaman G, Saxon LK, Price JS, et al. Wnt/beta-catenin signaling is a component of osteoblastic bone cell early responses to load-bearing and requires estrogen receptor alpha. J Biol Chem. 2007;282:20715–27.
Delhon I, Gutzwiller S, Morvan F, Rangwala S, Wyder L, Evans G, et al. Absence of estrogen receptor-related-α increases osteoblastic differentiation and cancellous bone mineral density. Endocrinology. 2009;150:4463–72.
Katsimbri P. The biology of normal bone remodeling. Eur J Cancer Care (Engl). 2017;26:e12740.
Song G, Habibovic P, Bao C, Hua J, van Blitterswijk CA, Yuan H, et al. The homing of bone marrow MSCs to non-osseous sites for ectopic bone formation induced by osteoinductive calcium phosphate. Biomaterials. 2013;34:2167–76.
Okabe YT, Kondo T, Mishima K, Hayase Y, Kato K, Mizuno M, et al. Biodistribution of locally or systemically transplanted osteoblast-like cells. Bone Joint Res. 2014;3:76–81.
Liu C, Zhang H, Tang X, Feng R, Yao G, Chen W, et al. Mesenchymal stem cells promote the osteogenesis in collagen-induced arthritic mice through the inhibition of TNF-α. Stem Cells Int. 2018;2018:4069032.
Leong WF, Zhou T, Lim GL, Li B. Protein palmitoylation regulates osteoblast differentiation through bmp-induced osterix expression. PLoS One. 2009;4:e4135.
Rahman MS, Akhtar N, Jamil HM, Banik RS, Asaduzzaman SM. TGF-β/BMP signaling and other molecular events: regulation of osteoblastogenesis and bone formation. Bone Res. 2015;3:15005.
Lam J, Takeshita S, Barker JE, Kanagawa O, Ross FP, Teitelbaum SL. TNF-alpha induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J Clin Invest. 2000;106:1481–8.
Zhao B, Grimes SN, Li S, Hu X, Ivashkiv LB. TNF-induced osteoclastogenesis and inflammatory bone resorption are inhibited by transcription factor RBP-J. J Exp Med. 2012;209:319–34.
Kobayashi Y, Maeda K, Takahashi N. Roles of Wnt signaling in bone formation and resorption. Jpn Dent Sci Rev. 2008;44:76–82.
Pramusita A, Mastutik G, Putra ST. Role of melatonin in down-regulation of receptor activator of nuclear factor kappa-B ligand: osteoprotegerin ratio in rat—bone-marrow mesenchymal stem cells. JKIMSU. 2018;7:12–21.
Davidge ST, Zhang Y, Stewart KG. A comparison of ovariectomy models for estrogen studies. Am J Physiol Regul Integr Comp Physiol. 2001;280:R904–7.
Sadat-Ali M, Al-Turki HA, Acharya S, Al-Dakheel DA. Bone marrow-derived osteoblasts in the management of ovariectomy induced osteoporosis in rats. J Stem Cells Regen Med. 2018;14:63–8.
Acknowledgements
The authors gratefully appreciate the financial support of the National Research Centre, Egypt (Thesis fund no. 71511). Also, the authors express sincere appreciation to Prof. Adel Bakeer Kholousy, Professor of Pathology, Faculty of Veterinary Medicine, Cairo University for his kind participation in histological examination of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no financial conflicts of interest.
Ethical statement
This study was conducted according to the ethical considerations for the experimental animals and approved by the Ethical Committee for Medical Research of the National Research Centre, Giza, Egypt, which follows the recommendations of the National Institutes of Health Guide for Care and Use of Laboratory Animals (Approval No.15106).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahmoud, N.S., Mohamed, M.R., Ali, M.A.M. et al. Osteoblast-Based Therapy—A New Approach for Bone Repair in Osteoporosis: Pre-Clinical Setting. Tissue Eng Regen Med 17, 363–373 (2020). https://doi.org/10.1007/s13770-020-00249-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-020-00249-5