Abstract
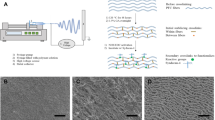
Expanded polytetrafluoroethylene (ePTFE) polymers do not support endothelialization because of nonconductive characteristics towards cellular attachment. Inner surface modification of the grafts can improve endothelialization and increase the long-term patency rate of the ePTFE vascular grafts. Here we reported a method of inner-surface modification of ePTFE vascular graft with extracellular matrix (ECM) and CD34 monoclonal antibodies (CD34 mAb) to stimulate the adhesion and proliferation of circulating endothelial progenitor cells on ePTFE graft to enhance graft endothelialization. The inner surface of ECM-coated ePTFE grafts were linked with CD34 mAb in the presence of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide/N-hydroxysuccinimide (EDC/NHS) solution and the physicochemical properties, surface morphology, biocompatibility, and hemocompatibility of the grafts were studied. The hydrophilicity of CD34 mAb-coated graft inner surface was significantly improved. Fourier transform infrared spectroscopy analysis confirmed ECM and CD34 mAb cross-linking in the ePTFE vascular grafts with our method. Scanning electron microscopy analysis showed protein layer covering uniformly on the inner surface of the modified grafts. The cell-counting kit-8 (CCK-8) assay confirmed that the modified graft has no obvious cytotoxicity. The modified graft showed a low hemolytic rate (0.9%) in the direct contact hemolysis test, suggesting the modification improved hemocompatibility of biopolymers. The modification also decreased adhesion of platelets, while significantly increased the adhesion of endothelial cells on the grafts. We conclude that our method enables ePTFE polymers modification with ECM and CD34 mAb, facilitates endothelialization, and inhibits platelet adhesion on the grafts, thus may increase the long-term patency rate of the prosthetic bypass grafts.







Similar content being viewed by others
References
Abbott WM, Callow A, Moore W, Rutherford R, Veith F, Weinberg S. Evaluation and performance standards for arterial prostheses. J Vasc Surg. 1993;17:746–56.
Albers M, Battistella VM, Romiti M, Rodrigues AA, Pereira CA. Meta-analysis of polytetrafluoroethylene bypass grafts to infrapopliteal arteries. J Vasc Surg. 2003;37:1263–9.
Ravi S, Chaikof EL. Biomaterials for vascular tissue engineering. Regen Med. 2010;5:107–28.
Faries PL, Logerfo FW, Arora S, Hook S, Pulling MC, Akbari CM, et al. A comparative study of alternative conduits for lower extremity revascularization: all-autogenous conduit versus prosthetic grafts. J Vasc Surg. 2000;32:1080–90.
Larsen CC, Kligman F, Tang C, Kottke-Marchant K, Marchant RE. A biomimetic peptide fluorosurfactant polymer for endothelialization of ePTFE with limited platelet adhesion. Biomaterials. 2007;28:3537–48.
Goh ET, Wong E, Farhatnia Y, Tan A, Seifalian AM. Accelerating in situ endothelialisation of cardiovascular bypass grafts. Int J Mol Sci. 2014;16:597–627.
Cines DB, Pollak ES, Buck CA, Loscalzo J, Zimmerman GA, McEver RP, et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood. 1998;91:3527–61.
Pearson JD. Endothelial cell function and thrombosis. Baillieres Best Pract Res Clin Haematol. 1999;12:329–41.
Avci-Adali M, Paul A, Ziemer G, Wendel HP. New strategies for in vivo tissue engineering by mimicry of homing factors for self-endothelialisation of blood contacting materials. Biomaterials. 2008;29:3936–45.
Rotmans JI, Heyligers JM, Verhagen HJ, Velema E, Nagtegaal MM, de Kleijn DP, et al. In vivo cell seeding with anti-CD34 antibodies successfully accelerates endothelialization but stimulates intimal hyperplasia in porcine arteriovenous expanded polytetrafluoroethylene grafts. Circulation. 2005;112:12–8.
Tseng DY, Edelman ER. Effects of amide and amine plasma-treated ePTFE vascular grafts on endothelial cell lining in an artificial circulatory system. J Biomed Mater Res. 1998;42:188–98.
Catto V, Farè S, Freddi G, Tanzi1 MC. Vascular tissue engineering: recent advances in small diameter blood vessel regeneration. ISRN Vascular Medicine 2014. doi:10.1155/2014/923030.
Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964–7.
Hristov M, Erl W, Weber PC. Endothelial progenitor cells: mobilization, differentiation, and homing. Arterioscler Thromb Vasc Biol. 2003;23:1185–9.
Melchiorri AJ, Hibino N, Yi T, Lee YU, Sugiura T, Tara S, et al. Contrasting biofunctionalization strategies for the enhanced endothelialization of biodegradable vascular grafts. Biomacromolecules. 2015;16:437–46.
Walachova K, Svorcik V, Bacakova L, Hnatowicz V. Colonization of ion-modified polyethylene with vascular smooth muscle cells in vitro. Biomaterials. 2002;23:2989–96.
Moby V, Boura C, Kerdjoudj H, Voegel JC, Marchal L, Dumas D, et al. Poly (styrenesulfonate)/poly(allylamine) multilayers: a route to favor endothelial cell growth on expanded poly(tetrafluoroethylene) vascular grafts. Biomacromolecules. 2007;8:2156–60.
Bu X, Yan Y, Zhang Z, Gu X, Wang M, Gong A, et al. Properties of extracellular matrix-like scaffolds for the growth and differentiation of endothelial progenitor cells. J Surg Res. 2010;164:50–7.
Danen EH, Sonnenberg A. Integrins in regulation of tissue development and function. J Pathol. 2003;201:632–41.
Williams SK, Kleinert LB, Patula-Steinbrenner V. Accelerated neovascularization and endothelialization of vascular grafts promoted by covalently bound laminin type 1. J Biomed Mater Res A. 2011;99:67–73.
Caiado F, Dias S. Endothelial progenitor cells and integrins: adhesive needs. Fibrogenesis Tissue Repair. 2012;5:4.
Vartanian KB, Kirkpatrick SJ, McCarty OJ, Vu TQ, Hanson SR, Hinds MT. Distinct extracellular matrix microenvironments of progenitor and carotid endothelial cells. J Biomed Mater Res A. 2009;91:528–39.
Motlagh D, Allen J, Hoshi R, Yang J, Lui K, Ameer G. Hemocompatibility evaluation of poly(diol citrate) in vitro for vascular tissue engineering. J Biomed Mater Res A. 2007;82:907–16.
Grunkemeier JM, Tsai WB, Horbett TA. Hemocompatibility of treated polystyrene substrates: contact activation, platelet adhesion, and procoagulant activity of adherent platelets. J Biomed Mater Res. 1998;41:657–70.
Raz O, Lev DL, Battler A, Lev EI. Pathways mediating the interaction between endothelial progenitor cells (EPCs) and platelets. PLoS ONE. 2014;9:e95156.
Mead LE, Prater D, Yoder MC, Ingram DA. Isolation and characterization of endothelial progenitor cells from human blood. Curr Protoc Stem Cell Biol 2008; Chapter 2: Unit 2C.1.
Chade AR, Zhu X, Lavi R, Krier JD, Pislaru S, Simari RD, et al. Endothelial progenitor cells restore renal function in chronic experimental renovascular disease. Circulation. 2009;119:547–57.
Mihaly J, Sterkel S, Ortner HM, Kocsis L, Hajba L, Furdyga E, et al. FTIR and FT-Raman spectroscopic study on polymer based high pressure digestion vessels. Croat Chem Acta. 2006;79:497–501.
de Mel A, Jell G, Stevens MM, Seifalian AM. Biofunctionalization of biomaterials for accelerated in situ endothelialization: a review. Biomacromolecules. 2008;9:2969–79.
Yin M, Yuan Y, Liu C, Wang J. Combinatorial coating of adhesive polypeptide and anti-CD34 antibody for improved endothelial cell adhesion and proliferation. J Mater Sci Mater Med. 2009;20:1513–23.
Ranjan AK, Kumar U, Hardikar AA, Poddar P, Nair PD, Hardikar AA. Human blood vessel-derived endothelial progenitors for endothelialization of small diameter vascular prosthesis. PLoS ONE. 2009;4:e7718.
Bacakova L, Filova E, Rypacek F, Svorcik V, Stary V. Cell adhesion on artificial materials for tissue engineering. Physiol Res. 2004;53:S35–45.
Petersen S, Strohbach A, Busch R, Felix SB, Schmitz KP, Sternberg K. Site-selective immobilization of anti-CD34 antibodies to poly(l-lactide) for endovascular implant surfaces. J Biomed Mater Res B Appl Biomater. 2014;102:345–55.
Chen JL, Li QL, Chen JY, Huang N. Adsoption CD34 antibody onto the col/hep coating film on titanium to improve cytocompatibility of titanium. Adv Mater Res. 2008;47:1411–4.
Lin Q, Ding X, Qiu F, Song X, Fu G, Ji J. In situ endothelialization of intravascular stents coated with an anti-CD34 antibody functionalized heparin-collagen multilayer. Biomaterials. 2010;31:4017–25.
Liu H, Li X, Niu X, Zhou G, Li P, Fan Y. Improved hemocompatibility and endothelialization of vascular grafts by covalent immobilization of sulfated silk fibroin on poly(lactic-co-glycolic acid) scaffolds. Biomacromolecules. 2011;12:2914–24.
Vashist SK, Dixit CK, MacCraith BD, O’Kennedy R. Effect of antibody immobilization strategies on the analytical performance of a surface plasmon resonance-based immunoassay. Analyst. 2011;136:4431–6.
Sohn YS, Lee YK. Site-directed immobilization of antibody using EDC-NHS-activated protein A on a bimetallic-based surface plasmon resonance chip. J Biomed Opt. 2014;19:051209.
Njatawidjaja E, Kodama M, Matsuzaki K, Yasuda K, Matsuda T, Kogoma M. Hydrophilic modification of expanded polytetrafluoroethylene (ePTFE) by atmospheric pressure glow discharge (APG) treatment. Surf Coat Technol. 2006;201:699–706.
Chong DS, Turner LA, Gadegaard N, Seifalian AM, Dalby MJ, Hamilton G. Nanotopography and plasma treatment: redesigning the surface for vascular graft endothelialisation. Eur J Vasc Endovasc Surg. 2015;49:335–43.
Jin G, Yao Q, Zhang S, Zhang L. Surface modifying of microporous PTFE capillary for bilirubin removing from human plasma and its blood compatibility. Mater Sci Eng C Mater Biol Appl. 2008;28:1480–8.
Hoshi RA, Van Lith R, Jen MC, Allen JB, Lapidos KA, Ameer G. The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts. Biomaterials. 2013;34:30–41.
Chandy T, Das GS, Wilson RF, Rao GH. Use of plasma glow for surface-engineering biomolecules to enhance bloodcompatibility of Dacron and PTFE vascular prosthesis. Biomaterials. 2000;21:699–712.
Zhou F, Jia X, Yang Y, Yang Q, Gao C, Zhao Y, et al. Peptide-modified PELCL electrospun membranes for regulation of vascular endothelial cells. Mater Sci Eng C Mater Biol Appl. 2016;68:623–31.
Nichols MD, Choudhary R, Kodali S, Reichert WM. Coagulation-induced resistance to fluid flow in small-diameter vascular grafts and graft mimics measured by purging pressure. J Biomed Mater Res B Appl Biomater. 2013;101:1367–76.
Freedman JE. Molecular regulation of platelet-dependent thrombosis. Circulation. 2005;112:2725–34.
Lamichhane S, Anderson JA, Remund T, Sun H, Larson MK, Kelly P, et al. Responses of endothelial cells, smooth muscle cells, and platelets dependent on the surface topography of polytetrafluoroethylene. J Biomed Mater Res A. 2016;104:2291–304.
Yoder MC, Mead LE, Prater D, Krier TR, Mroueh KN, Li F, et al. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood. 2007;109:1801–9.
Stroncek JD, Grant BS, Brown MA, Povsic TJ, Truskey GA, Reichert WM. Comparison of endothelial cell phenotypic markers of late-outgrowth endothelial progenitor cells isolated from patients with coronary artery disease and healthy volunteers. Tissue Eng Part A. 2009;15:3473–86.
Yoon CH, Hur J, Park KW, Kim JH, Lee CS, Oh IY, et al. Synergistic neovascularization by mixed transplantation of early endothelial progenitor cells and late outgrowth endothelial cells: the role of angiogenic cytokines and matrix metalloproteinases. Circulation. 2005;112:1618–27.
Tanaka M, Mochizuki A, Ishii N, Motomura T, Hatakeyama T. Study of blood compatibility with poly(2-methoxyethyl acrylate). Relationship between water structure and platelet compatibility in poly(2-methoxyethylacrylate-co-2-hydroxyethylmethacrylate). Biomacromolecules. 2002;3:36–41.
Lee JH, Lee HB. Platelet adhesion onto wettability gradient surfaces in the absence and presence of plasma proteins. J Biomed Mater Res. 1998;41:304–11.
Wang YX, Robertson JL, Spillman WJ, Claus RO. Effects of the chemical structure and the surface properties of polymeric biomaterials on their biocompatibility. Pharm Res. 2004;21:1362–73.
Yoder MC. Is endothelium the origin of endothelial progenitor cells? Arterioscler Thromb Vasc Biol. 2010;30:1094–103.
Gong X, Li Y, Gao Q, Cheng B, Shen B, Yan Z, et al. Adhesion behavior of endothelial progenitor cells to endothelial cells in simple shear flow. Acta Mech Sin. 2011;27:1071–80.
Melchiorri AJ, Hibino N, Fisher JP. Strategies and techniques to enhance the in situ endothelialization of small-diameter biodegradable polymeric vascular grafts. Tissue Eng Part B Rev. 2013;19:292–307.
Avci-Adali M, Ziemer G, Wendel HP. Induction of EPC homing on biofunctionalized vascular grafts for rapid in vivo self-endothelialization—a review of current strategies. Biotechnol Adv. 2010;28:119–29.
Shimizu T, Ito S, Kikuchi Y, Misaka M, Hirayama T, Ishimaru S, et al. Arterial conduit shear stress following bypass grafting for intermediate coronary artery stenosis: a comparative study with saphenous vein grafts. Eur J Cardiothorac Surg. 2004;25:578–84.
Acknowledgements
The authors thank all of the study participants and research staff of the Guangzhou Vascular Disease Center. This work was supported by Science Foundation of Guangdong Province (2010B080701034).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Ethical Statement
This study was approved by the Clinical Research and Experimental Animal Ethics Committee of the First Affiliated Hospital of Sun Yat-Sen University (IRB no. 2015_07) and institutional review board of Guangdong Provincial Health Bureau and Sun Yat-sen University.
Additional information
Lei Chen and Haipeng He are co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure S1
The schematic diagram shows the four consecutive stages in the development of the coated ePTFE grafts (TIFF 1317 kb)
Rights and permissions
About this article
Cite this article
Chen, L., He, H., Wang, M. et al. Surface Coating of Polytetrafluoroethylene with Extracellular Matrix and Anti-CD34 Antibodies Facilitates Endothelialization and Inhibits Platelet Adhesion Under Sheer Stress. Tissue Eng Regen Med 14, 359–370 (2017). https://doi.org/10.1007/s13770-017-0044-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-017-0044-3