Abstract
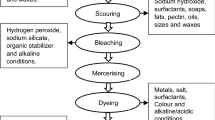
The textile industry is one of the major water consumers. Considering the large volume of water consumption, the textile industry is one of the producers of industrial wastewater. The dye removal from the wastewater generated by the textile industry is carried out in various ways, including physical, chemical, and biological methods or a combination thereof. Studies on wastewater treatment have devoted much attention to advanced oxidation processes. The solar photo-Fenton method has many applications for pollutant removal from aqueous solutions. The present study was conducted on the wastewater of Borujerd Textile Company (Borujerd city–Iran). The experiments were designed and performed using response surface methodology based on the Box–Behnken method. Solar lamp intensity (0–300 w/m2), process time (60–240 min), hydrogen peroxide concentration (100–600 mg/L), and iron ion concentration (10–60 mg/L) were selected as the operational variables. In addition, the removal rate of chemical oxygen demand was considered as the response of experimental design. Under optimal conditions (namely, \({I}_{SL}=150\frac{W}{{m}^{2}}\), \(t=157 {\text{min}}\), \({C}_{{H}_{2}{O}_{2}}=600\frac{mg}{L}\) and \({C}_{F{e}^{2+}}=60\frac{mg}{L}\)), the chemical oxygen demand removal rate reached the highest (i.e., 79.54%).






Similar content being viewed by others
Data availability
All data, models, or code generated or used during the study are available in a repository or online.
References
Aleksić M, Kušić H, Koprivanac N, Leszczynska D, Božić AL (2010) Heterogeneous Fenton type processes for the degradation of organic dye pollutant in water-the application of zeolite assisted AOPs. Desalination 257(1–3):22–29
Almeida LC, Garcia-Segura S, Arias C, Bocchi N, Brillas E (2012) Electrochemical mineralization of the azo dye Acid Red 29 (Chromotrope 2R) by photoelectron-Fenton process. Chemosphere 89(6):751–758
Antonin VS, Garcia-Segura S, Santos MC, Brillas E (2015) Degradation of Evans Blue diazo dye by electrochemical processes based on Fenton’s reaction chemistry. J Electroanal Chem 747:1–11
Baird RB, Eaton AD, Clesceri LS (2012) Standard methods for the examination of water and wastewater. In: Rice EW (ed) Washington. American public health association, DC
Bilińska L, Blus K, Gmurek M, Ledakowicz S (2019) Coupling of electrocoagulation and ozone treatment for textile wastewater reuse. Chem Eng J 358:992–1001
Bilińska L, Gmurek M, Ledakowicz S (2016) Comparison between industrial and simulated textile wastewater treatment by AOPs–biodegradability, toxicity and cost assessment. Chem Eng J 306:550–559
Bilińska L, Gmurek M, Ledakowicz S (2017) Textile wastewater treatment by AOPs for brine reuse. Process Saf Environ Prot 109:420–428
Blanco J, Torrades F, Morón M, Brouta-Agnésa M, García-Montaño J (2014) Photo-Fenton and sequencing batch reactor coupled to photo-Fenton processes for textile wastewater reclamation: feasibility of reuse in dyeing processes. Chem Eng J 240:469–475
Cabrera-Reina A, Miralles-Cuevas S, Rivas G, Pérez JS (2019) Comparison of different detoxification pilot plants for the treatment of industrial wastewater by solar photo-Fenton: are raceway pond reactors a feasible option? Sci Total Environ 648:601–608
Cardoso JC, Lucchiari N, Zanoni MVB (2015) Bubble annular photoeletrocatalytic reactor with TiO2 nanotubes arrays applied in the textile wastewater. J Environ Chem Eng 3(2):1177–1184
Dutta A, Das N, Sarkar D, Chakrabarti S (2019) Development and characterization of a continuous solar-collector-reactor for wastewater treatment by photo-Fenton process. Sol Energy 177:364–373
Ehzari H, Safari M, Samimi M, Shamsipur M, Gholivand MB (2022) A highly sensitive electrochemical biosensor for chlorpyrifos pesticide detection using the adsorbent nanomatrix contain the human serum albumin and the Pd: CdTe quantum dots. Microchem J 179:107424
El-sousy K, Hussen A, Hartani K, El–Aila H (2007) Elimination of organic pollutants using supported catalysts with hydrogen peroxide. Jordan J Chem 2(1):97–103
Florenza X, Solano AMS, Centellas F, Martínez-Huitle CA, Brillas E, Garcia-Segura S (2014) Degradation of the azo dye Acid Red 1 by anodic oxidation and indirect electrochemical processes based on Fenton’s reaction chemistry. Relationship between decolorization, mineralization and products. Electrochim Acta 142:276–288
Flox C, Ammar S, Arias C, Brillas E, Vargas-Zavala AV, Abdelhedi R (2006) Electro-Fenton and photoelectron-Fenton degradation of indigo carmine in acidic aqueous medium. Appl Catal B 67(1–2):93–104
Foteinis S, Monteagudo JM, Durán A, Chatzisymeon E (2018) Environmental sustainability of the solar photo-Fenton process for wastewater treatment and pharmaceuticals mineralization at semi-industrial scale. Sci Total Environ 612:605–612
Garcia-Segura S, Brillas E (2014) Advances in solar photoelectron-Fenton: decolorization and mineralization of the direct yellow 4 diazo dye using an autonomous solar pre-pilot plant. Electrochim Acta 140:384–395
Ghanbari F, Moradi M (2015) A comparative study of electrocoagulation, electrochemical Fenton, electro-Fenton and peroxi-coagulation for decolorization of real textile wastewater: electrical energy consumption and biodegradability improvement. J Environ Chem Eng 3(1):499–506
Iranifam M, Zarei M, Khataee AR (2011) Decolorization of CI basic yellow 28 solution using supported ZnO nanoparticles coupled with photoelectron-Fenton process. J Electroanal Chem 659(1):107–112
Jafari Mansoorian H, Bazrafshan E, Yari AR, Alizadeh M (2014) Removal of azo dyes from aqueous solution using Fenton and modified Fenton processes. Health Scope 3(2):e15507
Jorfi S, Pourfadakari S, Kakavandi B (2018) A new approach in sono-photocatalytic degradation of recalcitrant textile wastewater using MgO@Zeolite nanostructure under UVA irradiation. Chem Eng J 343:95–107
Kaur P, Kushwaha JP, Sangal VK (2017) Evaluation and disposability study of actual textile wastewater treatment by electro-oxidation method using Ti/RuO2 anode. Process Saf Environ Prot 111:13–22
Khataee AR, Zarei M, Moradkhannejhad L (2010) Application of response surface methodology for optimization of azo dye removal by oxalate catalyzed photoelectron-Fenton process using carbon nanotube-PTFE cathode. Desalination 258(1–3):112–119
Manjarrez Paba G, Baldiris Ávila R, Baena Baldiris D (2021) Application of environmental bacteria as potential methods of azo dye degradation systems. Global J Environ Sci Manage 7:131–154
Miklos DB, Remy C, Jekel M, Linden KG, Drewes JE, Hübner U (2018) Evaluation of advanced oxidation processes for water and wastewater treatment–A critical review. Water Res 139:118–131
Milidrag GP, Prica M, Kerkez D, Dalmacija B, Kulic A, Pilipovic DT, Tomin MB (2018) A comparative study of the decolorization capacity of the solar-assisted Fenton process using ferrioxalate and Al, Fe-bentonite catalysts in a parabolic trough reactor. J Taiwan Inst Chem Eng 93:436–449
Moghadam H, Samimi M (2022) Effect of condenser geometrical feature on evacuated tube collector basin solar still performance: productivity optimization using a Box-Behnken design model. Desalination 542:116092
Montgomery DC (2017) Design and analysis of experiments. Wiley, Hoboken
Moreira FC, Garcia-Segura S, Vilar VJ, Boaventura RA, Brillas E (2013) Decolorization and mineralization of Sunset Yellow FCF azo dye by anodic oxidation, electro-Fenton, UVA photoelectron-Fenton and solar photoelectron-Fenton processes. Appl Catal B 142:877–890
Nayna OK, Tareq SM (2019) Application of semiconductor nanoparticles for removal of organic pollutants or dyes from wastewater. Nanotechnology in water and wastewater treatment. Elsevier, Amsterdam, pp 267–290
Oturan MA, Aaron JJ (2014) Advanced oxidation processes in water/wastewater treatment: principles and applications. A review. Critic Rev Environ Sci Technol 44(23):2577–2641
Paździor K, Bilińska L, Ledakowicz S (2019) A review of the existing and emerging technologies in the combination of AOPs and biological processes in industrial textile wastewater treatment. Chem Eng J 376:120597
Peralta-Hernández JM, Meas-Vong Y, Rodríguez FJ, Chapman TW, Maldonado MI, Godínez LA (2008) Comparison of hydrogen peroxide-based processes for treating dye-containing wastewater: decolorization and destruction of Orange II azo dye in dilute solution. Dyes Pigm 76(3):656–662
Peralta-Hernández JM, Vijay S, Rodríguez-Narváez O, Pacheco-Álvarez MA (2018) Photo and solar Fenton processes for wastewater treatment. Electrochemical water and wastewater treatment. Butterworth-Heinemann, Oxford, pp 223–237
Ruiz EJ, Arias C, Brillas E, Hernández-Ramírez A, Peralta-Hernández JM (2011) Mineralization of acid yellow 36 azo dye by electro-Fenton and solar photoelectron-Fenton processes with a boron-doped diamond anode. Chemosphere 82(4):495–501
Samimi M, Safari M (2022) TMU-24 (Zn-based MOF) as an advance and recyclable adsorbent for the efficient removal of eosin B: characterization, equilibrium, and thermodynamic studies. Environ Prog Sustain Energy 41(5):e13859
Samimi M, Mohammadzadeh E, Mohammadzadeh A (2023) Rate enhancement of plant growth using Ormus solution: optimization of operating factors by response surface methodology. Int J Phytoremediation 25(12):1636–1642
Samimi M, Shahriari-Moghadam M (2023) The Lantana camara L. stem biomass as an inexpensive and efficient biosorbent for the adsorptive removal of malachite green from aquatic environments: kinetics, equilibrium and thermodynamic studies. Int J Phytoremed 25(10):1–9
Solano AMS, Garcia-Segura S, Martínez-Huitle CA, Brillas E (2015) Degradation of acidic aqueous solutions of the diazo dye Congo red by photo-assisted electrochemical processes based on Fenton’s reaction chemistry. Appl Catal B 168:559–571
Soltani RDC, Jorfi S, Safari M, Rajaei MS (2016) Enhanced sonocatalysis of textile wastewater using bentonite-supported ZnO nanoparticles: response surface methodological approach. J Environ Manage 179:47–57
Sulistyowati L, Nurhasanah; Riani, E., & Cordova, M. R. (2022) Heavy metals concentration in the sediment of the aquatic environment caused by the leachate discharge from a landfill. Global J Environ Sci Manage 9:323–336
Thiam A, Sirés I, Garrido JA, Rodríguez RM, Brillas E (2015) Decolorization and mineralization of Allura Red AC aqueous solutions by electrochemical advanced oxidation processes. J Hazard Mater 290:34–42
Torrades F, García-Montaño J (2014) Using central composite experimental design to optimize the degradation of real dye wastewater by Fenton and photo-Fenton reactions. Dyes Pigm 100:184–189
Yaseen DA, Scholz M (2019) Textile dye wastewater characteristics and constituents of synthetic effluents: a critical review. Int J Environ Sci Technol 16(2):1193–1226
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
MM performed supervision, writing—original draft preparation, writing—reviewing and editing, and investigation. NS provided data curation, methodology, software, and investigation
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Editorial responsibility: S. Mirkia.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharifi, N., Mohadesi, M. Textile wastewater treatment using solar photo-Fenton process. Int. J. Environ. Sci. Technol. 21, 2709–2722 (2024). https://doi.org/10.1007/s13762-023-05397-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-023-05397-4