Abstract
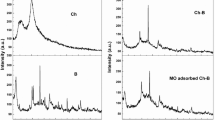
The public is still concerned about the effectiveness and efficiency of wastewater treatment. Investigating a better material to contribute to water purification is the objective of this research. Absorbent cotton-templated manganese oxide nanocomposites (AC/MnO2) were facilely synthesized by immersing adsorbent cotton into a potassium permanganate (KMnO4) solution through an in situ reduction with a biological template strategy. AC/MnO2 is attracted to hydration hydroxyl in aqueous solutions and has an excellent adsorption capacity for dye wastewater degradation in aqueous solutions. The morphology, structure, composition, and thermostability of AC/MnO2 were characterized by FE-SEM, XPS, XRD, FTIR, and TGA. This as-prepared nanomaterial was applied to degrade methylene blue. The factors that affected the degradation performance, including soaking time, initial methylene blue concentration, the amount of AC/MnO2, pH, and temperature, were investigated. The experiments indicated that the optimal conditions for adsorption were achieved at pH 5 using three wads of AC/MnO2 in 15 min, which were synthesized by soaking for 60 h. The isothermal adsorption behavior and kinetics were studied by measuring the absorbance intensities in reaction time to fit Langmuir and Freundlich's adsorption isotherms, pseudo-first-order kinetic models, and pseudo-second-order kinetic models. The correlation coefficient comparisons demonstrated that methylene blue degradation conformed to the Langmuir isotherm, indicating that the superficial behavior was like the monolayer and pseudo-second-order models, elucidating that this process contained both physical diffusion and chemical adsorption. The regeneration capability was assessed to demonstrate that AC/MnO2 can be recycled. The degradation in efficiency persisted at about 40% after five cycles.










Similar content being viewed by others
References
Asfaram A, Ghaedi M, Hajati S, Goudarzi A (2015) Ternary dye adsorption onto MnO2 nanoparticle-loaded activated carbon: Derivative spectrophotometry and modeling. RSC Adv 5(88):72300–72320. https://doi.org/10.1039/c5ra10815b
Ayub A, Raza ZA, Majeed MI, Tariq MR, Irfan A (2020) Development of sustainable magnetic chitosan biosorbent beads for kinetic remediation of arsenic contaminated water. Int J Biol Macromol 163:603–617. https://doi.org/10.1016/j.ijbiomac.2020.06.287
Babar M, Munir HMS, Nawaz A, Ramzan N, Azhar U, Sagir M, Tahir MS, Ikhlaq A, Mohamad Azmin NH, Mubashir M, Khoo KS, Chew KW (2022) Comparative study of ozonation and ozonation catalyzed by Fe-loaded biochar as catalyst to remove methylene blue from aqueous solution. Chemosphere 307:135738. https://doi.org/10.1016/j.chemosphere.2022.135738
Basfer NM, Mansour SF, Ahmed MK (2021) Physicochemical properties of hydroxyapatite modified with vanadium ions for degradation of methylene blue. J Mol Struct 1240:130562. https://doi.org/10.1016/j.molstruc.2021.130562
Bizani E, Fytianos K, Poulios I, Tsiridis V (2006) Photocatalytic decolorization and degradation of dye solutions and wastewaters in the presence of titanium dioxide. J Hazard Mater 136(1):85–94. https://doi.org/10.1016/j.jhazmat.2005.11.017
Darr JA, Zhang J, Makwana NM, Weng X (2017) Continuous hydrothermal synthesis of inorganic nanoparticles: applications and future directions. Chem Rev 117(17):11125–11238. https://doi.org/10.1021/acs.chemrev.6b00417
Devaraj N, Munichandraiah S (2008) Effect of crystallographic structure of MnO2 on its electrochemical capacitance properties. J Phys Chem C 112:4406–4417. https://doi.org/10.1021/jp7108785
DeVoy JE, Congiusta E, Lundberg DJ, Findeisen S, Bhattacharya S (2021) Post-consumer textile waste and disposal: Differences by socioeconomic, demographic, and retail factors. Waste Manag 136:303–309. https://doi.org/10.1016/j.wasman.2021.10.009
Ding B, Zheng P, Ma P, Lin J (2020) Manganese oxide nanomaterials: Synthesis, properties, and theranostic applications. Adv Mater 32(10):e1905823. https://doi.org/10.1002/adma.201905823
dos Santos AB, Cervantes FJ, van Lier JB (2007) Review paper on current technologies for decolourisation of textile wastewaters: Perspectives for anaerobic biotechnology. Bioresour Technol 98(12):2369–2385. https://doi.org/10.1016/j.biortech.2006.11.013
Ejikeme EM, Igbokwe PK (2016) Kinetics, isotherm and thermodynamics studies of swiss blue dye desorption from spent microwave prepared ‘hamburgar’ seed shell activated carbon. The International Journal of Engineering and Science (IJES) 5(7):47–60
El-Ghobashy MA, Salem IA, El-Dahrawy WM, Salem MA (2023) Fabrication of α-MnO2/Fe-Mn binary oxide nanocomposite as an efficient adsorbent for the removal of methylene blue from wastewater. J Mol Struct 1272:134118. https://doi.org/10.1016/j.molstruc.2022.134118
Han L, Liu P, Zhang H, Li F, Liu A (2017) Phage capsid protein-directed MnO2 nanosheets with peroxidase-like activity for spectrometric biosensing and evaluation of antioxidant behaviour. Chem Commun (camb) 53(37):5216–5219. https://doi.org/10.1039/c7cc02049j
Hashem AM, Abuzeid H, Kaus M, Indris S, Ehrenberg H, Mauger A, Julien CM (2018) Green synthesis of nanosized manganese dioxide as positive electrode for lithium-ion batteries using lemon juice and citrus peel. Electrochim Acta 262:74–81. https://doi.org/10.1016/j.electacta.2018.01.024
He Y, Jiang B, Chen J, Jiang Y, Zhang YX (2018) Synthesis of MnO2 nanosheets on montmorillonite for oxidative degradation and adsorption of methylene blue. J Colloid Interface Sci 510:207–220. https://doi.org/10.1016/j.jcis.2017.09.066
Ho YS (2006) Review of second-order models for adsorption systems. J Hazard Mater 136(3):681–689. https://doi.org/10.1016/j.jhazmat.2005.12.043
Hong X, Zhang G, Zhu Y, Yang H (2003) Sol–gel synthesis and characterization of mesoporous manganese oxide. Mater Res Bull 38(13):1695–1703. https://doi.org/10.1016/j.materresbull.2003.07.005
Ikhlaq A, Munir HMS, Khan A, Javed F, Joya KS (2018) Comparative study of catalytic ozonation and Fenton-like processes using iron-loaded rice husk ash as catalyst for the removal of methylene blue in wastewater. Ozone Sci Eng 41(3):250–260. https://doi.org/10.1080/01919512.2018.1525276
Iwuozor KO, Ighalo JO, Ogunfowora LA, Adeniyi AG, Igwegbe CA (2021) An empirical literature analysis of adsorbent performance for methylene blue uptake from aqueous media. J Environ Chem Eng 9(4):105658. https://doi.org/10.1016/j.jece.2021.105658
Jiang S, Yu T, Xia R, Wang X, Gao M (2019) Realization of super high adsorption capability of 2D δ-MnO2/GO through intra-particle diffusion. Mater Chem Phys 232:374–381. https://doi.org/10.1016/j.matchemphys.2019.05.004
Jiao C, Wei N, Liu D, Wang J, Liu S, Fu F, Liu T, Li T (2021) Sustainable Fenton-like degradation of methylene blue over MnO2-loaded poly(amidoxime-hydroxamic acid) cellulose microrods. Int J Biol Macromol 193(Pt B):1952–1961. https://doi.org/10.1016/j.ijbiomac.2021.11.026
Jittiarporn P, Badilescu S, Al Sawafta MN, Sikong L, Truong V-V (2017) Electrochromic properties of sol–gel prepared hybrid transition metal oxides – a short review. J Sci Adv Mater Devices 2(3):286–300. https://doi.org/10.1016/j.jsamd.2017.08.005
Kerli S, Kavgacı M, Soğuksu AK, Avar B (2022) Photocatalytic degradation of methylene blue, rhodamine-b, and malachite green by Ag @ ZnO/TiO2. Braz J Phys 52(1):1–11. https://doi.org/10.1007/s13538-021-01007-1
Krishna Moorthy A, Govindarajan Rathi B, Shukla SP, Kumar K, Shree Bharti V (2021) Acute toxicity of textile dye methylene blue on growth and metabolism of selected freshwater microalgae. Environ Toxicol Pharmacol 82:103552. https://doi.org/10.1016/j.etap.2020.103552
Kuan WH, Chen CY, Hu CY (2011) Removal of methylene blue from water by γ-MnO2. Water Sci Technol 64(4):899–903. https://doi.org/10.2166/wst.2011.262
Kubo M, Moriyama R, Shimada M (2019) Facile fabrication of HKUST-1 nanocomposites incorporating Fe3O4 and TiO2 nanoparticles by a spray-assisted synthetic process and their dye adsorption performances. Microporous Mesoporous Mater 280:227–235. https://doi.org/10.1016/j.micromeso.2019.02.010
Kumar KV, Gadipelli S, Wood B, Ramisetty KA, Stewart AA, Howard CA, Brett DJ, Rodriguez-Reinoso F (2019) Characterization of the adsorption site energies and heterogeneous surfaces of porous materials. J Mater Chem A 7(17):10104–10137
Lawrie K, Mills A, Hazafy D (2013) Simple inkjet-printed, UV-activated oxygen indicator. Sens Actuators B Chem 176:1154–1159. https://doi.org/10.1016/j.snb.2012.10.125
Li Z, Tang X, Liu K, Huang J, Xu Y, Peng Q, Ao M (2018) Synthesis of a MnO2/Fe3O4/diatomite nanocomposite as an efficient heterogeneous Fenton-like catalyst for methylene blue degradation. Beilstein J Nanotechnol 9:1940–1950. https://doi.org/10.3762/bjnano.9.185
Liu L, Liu Y, Wang X, Hu N, Li Y, Li C, Meng Y, An Y (2021) Synergistic effect of B-TiO2 and MIL-100(Fe) for high-efficiency photocatalysis in methylene blue degradation. Appl Surf Sci 561:149969. https://doi.org/10.1016/j.apsusc.2021.149969
Luo X, Liang H, Qu F, Ding A, Cheng X, Tang CY, Li G (2018) Free-standing hierarchical α-MnO2@CuO membrane for catalytic filtration degradation of organic pollutants. Chemosphere 200:237–247. https://doi.org/10.1016/j.chemosphere.2018.02.113
Mark Wainwright LA (2005) Review: The phenothiazinium chromophore and the evolution of antimalarial drugs. Tropical Med Int Health 10(6):501–511. https://doi.org/10.1111/j.1365-3156.2005.01417.x
Mary Ealias A, Saravanakumar MP (2019) A critical review on ultrasonic-assisted dye adsorption: Mass transfer, half-life and half-capacity concentration approach with future industrial perspectives. Crit Rev Environ Sci Technol 49(21):1959–2015. https://doi.org/10.1080/10643389.2019.1601488
Mondal K, Lalvani SB (2000) Modeling of mass transfer controlled adsorption rate based on the langmuir adsorption isotherm. Sep Sci Technol 35(16):2583–2599. https://doi.org/10.1081/ss-100102357
Nandi B, Goswami A, Purkait M (2009) Removal of cationic dyes from aqueous solutions by kaolin: Kinetic and equilibrium studies. Appl Clay Sci 42(3–4):583–590. https://doi.org/10.1016/j.clay.2008.03.015
Olusegun SJ, Freitas ETF, Lara LRS, Mohallem NDS (2021) Synergistic effect of a spinel ferrite on the adsorption capacity of nano bio-silica for the removal of methylene blue. Environ Technol 42(14):2163–2176. https://doi.org/10.1080/09593330.2019.1694083
Park J-H, Wang JJ, Seo D-C (2021) Comparison of catalytic activity for treating recalcitrant organic pollutant in heterogeneous Fenton oxidation with iron-impregnated biochar and activated carbon. J Water Process Eng 42:102141. https://doi.org/10.1016/j.jwpe.2021.102141
Pavan FA, Mazzocato AC, Gushikem Y (2008) Removal of methylene blue dye from aqueous solutions by adsorption using yellow passion fruit peel as adsorbent. Bioresour Technol 99(8):3162–3165. https://doi.org/10.1016/j.biortech.2007.05.067
Rafatullah M, Sulaiman O, Hashim R, Ahmad A (2010) Adsorption of methylene blue on low-cost adsorbents: A review. J Hazard Mater 177(1–3):70–80. https://doi.org/10.1016/j.jhazmat.2009.12.047
Ren L, Tang Z, Du J, Chen L, Qiang T (2021) Recyclable polyurethane foam loaded with carboxymethyl chitosan for adsorption of methylene blue. J Hazard Mater 417:126130. https://doi.org/10.1016/j.jhazmat.2021.126130
Salleh MAM, Mahmoud DK, Karim WAWA, Idris A (2011) Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280(1–3):1–13. https://doi.org/10.1016/j.desal.2011.07.019
Sharma P, Laddha H, Agarwal M, Gupta R (2022) Selective and effective adsorption of malachite green and methylene blue on a non-toxic, biodegradable, and reusable fenugreek galactomannan gum coupled MnO2 mesoporous hydrogel. Microporous Mesoporous Mater 338:111982. https://doi.org/10.1016/j.micromeso.2022.111982
Shayesteh H, Ashrafi A, Rahbar-Kelishami A (2017) Evaluation of Fe3O4@MnO2 core-shell magnetic nanoparticles as an adsorbent for decolorization of methylene blue dye in contaminated water: Synthesis and characterization, kinetic, equilibrium, and thermodynamic studies. J Mol Struct 1149:199–205. https://doi.org/10.1016/j.molstruc.2017.07.100
Shokoohi R, Nematollahi D, Samarghandi MR, Azarian G, Latifi Z (2020) Optimization of three-dimensional electrochemical process for degradation of methylene blue from aqueous environments using central composite design. Environ Technol Innov 18:100711. https://doi.org/10.1016/j.eti.2020.100711
Siddiqui SI, Manzoor O, Mohsin M, Chaudhry SA (2019) Nigella sativa seed based nanocomposite-MnO2/BC: An antibacterial material for photocatalytic degradation, and adsorptive removal of methylene blue from water. Environ Res 171:328–340. https://doi.org/10.1016/j.envres.2018.11.044
Simonin J-P (2016) On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chem Eng J 300:254–263. https://doi.org/10.1016/j.cej.2016.04.079
Su M, Li H, He X, Xu Z (2022) Significant enhancement of pesticide and organic dyes degradation by ion-exchange within a metal–organic framework. Polyhedron 215:115651. https://doi.org/10.1016/j.poly.2022.115651
Tan Y, Chen M, Hao Y (2012) High efficient removal of PB (II) by amino-functionalized Fe3O4 magnetic nano-particles. Chem Eng J 191:104–111. https://doi.org/10.1016/j.cej.2012.02.075
Theiss FL, Ayoko GA, Frost RL (2016) Synthesis of layered double hydroxides containing Mg2+, Zn2+, Ca2+ and Al3+ layer cations by co-precipitation methods—a review. Appl Surf Sci 383:200–213. https://doi.org/10.1016/j.apsusc.2016.04.150
Tong H, Yang C, Lv Y, Wang L, Chen K, Zhou X (2021) Fabrication of tubular porous titanium membrane electrode and application in electrochemical membrane reactor for treatment of wastewater. J Ind Eng Chem 96:269–276. https://doi.org/10.1016/j.jiec.2021.01.030
Wang H-Q, Zheng M-B, Chen J-H, Ji G-B, Cao J-M (2010) Synthesis of MnO2 microfiber with secondary nanostructure by cotton template. Journal of Nanotechnology 2010:1–5. https://doi.org/10.1155/2010/479172
Wang Q, Ma C, Tang J, Zhang C, Ma L (2018) Eggshell membrane-templated MnO2 nanoparticles: Facile synthesis and tetracycline hydrochloride decontamination. Nanoscale Res Lett 13(1):255. https://doi.org/10.1186/s11671-018-2679-y
Wang Y, Rao S, Mao P, Zhang F, Xiao P, Peng L, Zhu Q (2020) Controlled synthesis of Fe3O4@C@manganese oxides (MnO2, Mn3O4 and MnO) hierarchical hollow nanospheres and their superior lithium storage properties. Electrochim Acta 337:135739. https://doi.org/10.1016/j.electacta.2020.135739
Yan G, Wang P, Li Y, Qin Z, Lan S, Yan Y, Zhang Q, Chen X (2021) MnO2 adsorption ocidation mechanism. Adsorpt Sci Technol 2021:12. https://doi.org/10.1155/2021/3069392
Zhang L, Lian J, Wu L, Duan Z, Jiang J, Zhao L (2014) Synthesis of a thin-layer MnO2 nanosheet-coated Fe3O4 nanocomposite as a magnetically separable photocatalyst. Langmuir 30(23):7006–7013. https://doi.org/10.1021/la500726v
Zhang W, Zhang B, Jin H, Li P, Zhang Y, Ma S, Zhang J (2018) Waste eggshell as bio-template to synthesize high capacity δ-MnO2 nanoplatelets anode for lithium ion battery. Ceram Int 44(16):20441–20448. https://doi.org/10.1016/j.ceramint.2018.08.038
Zhang H, Xu F, Xue J, Chen S, Wang J, Yang Y (2020) Enhanced removal of heavy metal ions from aqueous solution using manganese dioxide-loaded biochar: Behavior and mechanism. Sci Rep 10(1):6067. https://doi.org/10.1038/s41598-020-63000-z
Zhao P, Yao M, Ren H, Wang N, Komarneni S (2019) Nanocomposites of hierarchical ultrathin MnO2 nanosheets/hollow carbon nanofibers for high-performance asymmetric supercapacitors. Appl Surf Sci 463:931–938. https://doi.org/10.1016/j.apsusc.2018.09.041
Zia J, Aazam ES, Riaz U (2020) Facile synthesis of MnO2 nanorods and ZnMn2O4 nanohexagons: A comparison of microwave-assisted catalytic activity against 4-nitrophenol degradation. J Market Res 9(5):9709–9719. https://doi.org/10.1016/j.jmrt.2020.06.048
Acknowledgements
This research was supported by the University of Malaya and the Taiyuan Institute of Technology. We are grateful to Qi Wang for the fruitful discussion.
Author information
Authors and Affiliations
Contributions
QW contributed to the study’s conception and design. JW, QW, and WW performed material preparation, data analysis, and analysis. SMK contributed to supervising JW and reviewing and editing the manuscript. JW wrote the first draft of the manuscript, and all authors commented on previous versions. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest in this work. They also declare that they do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Additional information
Editorial responsibility: Jing Chen.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, J., Wang, Q., Wu, W. et al. Absorbent cotton-templated manganese oxide nanocomposites for methylene blue promote isothermal adsorption. Int. J. Environ. Sci. Technol. 21, 417–430 (2024). https://doi.org/10.1007/s13762-023-04983-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-023-04983-w