Abstract
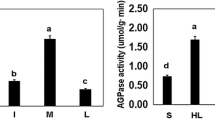
This research was conducted to evaluate the impact of different irrigation on starch biosynthesis pathway with emphasis on cotton yield. Experiments were conducted in split-plot factorial design. Significant differences were observed in irrigation levels. Starch and alpha-amylase activity increased for seeds rainfed source under 100%field-capacity irrigation. Protein increased in seed sources that were grown in 33 and 66% water-need irrigated 100%field-capacity in double water stress exposure. Although rainfed seeds had an early harvest, highest total yield was observed in seed sources that were grown in exposure to 33%field-capacity which irrigated 100%field-capacity. To identify genes involved in starch biosynthesis, cotton alpha-amylase and ADP-glucose pyrophosphorylase were studied in silico. The alpha-amylase protein encoded 409 amino acids with a molecular weight of 46,790 Dalton. Alpha-amylase and ADP-glucose-pyrophosphorylase proteins belonged to the glycosyl hydrolase and transferase family, respectively. Phylogenetic analysis revealed that cotton sequences were separated along with co-family plants in the Malvaceae family. Biochemical responses observed in reduced glucose and protein and to a lesser extent starch as the level of water stress increased. Seeds exposed to double water stress had less starch than triple stress exposure. The sowing of seeds in water shortage continually resulted in low yields. Seeds obtained from 66%field-capacity irrigation had less starch, stomata density and more protein. Higher levels of protein and glucose were present in seeds exposed to double stress as compared to triple stress. In areas with limited water resources, it is recommended that cotton seeds be propagated under irrigation under 66%field-capacity, which have adequate carbohydrate resources and produce better yields.











Similar content being viewed by others
References
Abdel Gadir A, Dougherty M (2012) Effect of different deficit-irrigation capabilities on cotton yield in the Tennessee Valley. Irrigat Drainage Sys Eng 01:2
Adam IK, Bello BA, Adam AA, Imam AA (2018) Analysis of phylogenetic sequence relationships between plant alpha amylases. Bayero J Pure Appl Sci (BAJOPAS) 11(2):81–93
Adda A, Regagba Z, Latigui A, Merah O (2014) Effect of salt stress on α-amylase activity, sugars mobilization and osmotic potential of Phaseolus vulgaris L. seeds var. ‘Cocorose’ and ‘Djadida’ during germination. J Biol Sci 14(5):370–375
Asoodeh A, Lagzian M (2012) Purification and characterization of a new glucoamylopullulanase from thermotolerant alkaliphilic Bacillus subtilis DR8806 of a hot mineral spring. J Process Biochem 47:806–815
Ballicora MA, Iglesias AA, Preiss J (2004) ADP-glucose pyrophosphorylase: a regulatory enzyme for plant starch synthesis. Photosynth Res 79:1–24
Biazus JPM, Souza RRD, Márquez JE, Franco TT, Santana JCC, Tambourgi EB (2009) Production and characterization of amylases from Zea mays malt. Braz Arch Biol Technol 52:991–1000
Blum A, Ebercon A (1981) Cell membrane stability as a measure of drought and heat tolerance in wheat 1. Crop Sci 21(1):43–47
Burhan A (2008) Highly thermostable, thermophilic, alkaline, SDS and chelator resistant amylase from a thermophilic Bacillus sp. isolate A3–15. Biores Technol 99:3071–3076
Bussis D, Kauder F, Heineke D (1998) Acclimation of potato plants to polyethylene glycol-induced water deficit. I. Photosynthesis and metabolism. J Exp Bot 49(13):49–60
Cochard H, Coll L, Le Roux X, Ame´glio T (2002) Unraveling the effects of plant hydraulics on stomatal closure during water stress in walnut. Plant Physiol 128:282–290
Dağdelen N, Başal H, Yılmaz E, Gürbüz T, Akçay S (2009) Different drip irrigation regimes affect cotton yield, water use efficiency and fiber quality in western Turkey. Agric Water Manag 96(1):111–120
Declerck N, Joyet P, Gaillardin C, Masson JM, Huber R, Wiegand G (2002) Engineering the thermo stability of Bacillus licheniformis α-amylase. Biol Bratisl 57:203–211
Elliott J, Deryng D, Müller C, Frieler K, Konzmann M, Gerten D, Glotter M, Flörke M, Wada Y, Best N, Eisner S, Fekete BM, Folberth C, Foster I, Gosling SN, Haddeland I, Khabarov N, Ludwig F, Masaki Y, Olin S, Rosenzweig C, Ruane AC, Satoh Y, Schmid E, Stacke T, Tang Q, Wisser D (2014) Constraints and potentials of future irrigation water availability on agricultural production under climate change. Proc Natl Acad Sci USA 111(9):3239–3244
Faghani E, Kolahi M, Sohrabi B, Goldson-Barnaby A (2019) Anatomic features and antioxidant activity of cotton seed (Gossypium hirsutum L.) genotypes under different irrigation regimes. J Plant Growth Regul 38:883–896
Faghani E, M Razzaghi, B Sohrabi, M Kolahi, A Chekani (2018) Investigation of physiological characteristics and vigor of cotton seeds under water stress and evaluation of their tolerance in Golestan province. Final report of complete project, cotton research institute. (code: 3-07-075195105)
Gasteiger E, Gattiker A, Hoogland C, Ivanyi I, Appel RD (2003) ExPASy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 31(13):3784–3788
Ghorbani M, Eskandari-Damaneh H, Cotton M, Choochani O, Borgi M (2021) Harnessing indigenous knowledge for climate change-resilient water management–lessons from an ethnographic case study in Iran. Clim Dev 1:1–4
Goldberg T, Hecht M, Hamp T, Karl T, Yachdav G (2014) LocTree3 prediction of localization. Nucleic Acids Res 42:350–355
Gossett DR, Millhollon EP, Lucas MC (1994) Antioxidant response to NaCl stress in salt-tolerant and salt-sensitive cultivars of cotton. Crop Sci 34(3):706–714
Hendriks JHM, Kolbe A, Gibon Y, Stitt M, Geigenberger P (2003) ADP-glucose pyrophosphorylase is activated by posttranslational redox-modification in response to light and to sugars in leaves of Arabidopsis and other plant species. Plant Physiol 133:838–849
Horváthová V, Janecek S, Sturdík E (2001) Amylolytic enzymes: molecular aspects of their properties. Gen Physiol Biophys 20:7–32
Isah T (2019) Stress and defense responses in plant secondary metabolites production. Biol Res 52(1):39–39
Janecek S, Svensson B, MacGregor EA (2014) α-Amylase: an enzyme specificity found in various families of glycoside hydrolases. Cell Mol Life Sci 71:1149–1170
Jin X, Ballicora MA, Preiss J, Geiger JH (2005) Crystal structure of potato tuber ADP-glucose pyrophosphorylase. Eur Mol Biol Organ (EMBO) 24:694–704
Jobling S (2004) Improving starch for food and industrial applications. Curr Opin Plant Biol 7:210–218
Jones R, Varner J (1967) The bioassay of gibberellins. Planta 72:155–161
Kanber R, Koç DL, Tekin S, Kapur B (2011) Effects of deficit irrigation on the yield and yield components of drip irrigated cotton in a mediterranean environment. Agric Water Manag 98:597–605
Kaneko M, Itoh H, Ueguchi-Tanaka M, Ashikari M, Matsuoka M (2002) The alpha-amylase induction in endosperm during rice seed germination is caused by gibberellin synthesized in epithelium. Plant Physiol 128:1264–1270
Kang Y, Wang R, Wan S, Hu W, Jiang S, Liu S (2012) Effects of different water levels on cotton growth and water use through drip irrigation in an arid region with saline ground water of Northwest China. Agric Water Manag 109:117–126
Kashif Rafiq Z, Farhan A, Farooq Sh, Muhammad Y, Tariq Sh, Durri Sh, Waseem H, Zahoor A, Chao Q, Yanli L, Amjad I, Wei W (2016) Response and tolerance mechanism of cotton Gossypium hirsutum L. to elevated temperature stress. Front Plant Sci 7:937
Kavakli IH, Kato C, Choi SB, Kim KH, Salamone PR, Ito H, Okita TW (2002) Generation, characterization, and heterologous expression of wild-type and up-regulated forms of Arabidopsis thaliana leaf ADP-glucose pyrophosphorylase. Planta J 215:430–439
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJ (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10(6):845–855
Khatibi S, Arjjumend H (2019) Water crisis in making in Iran. Grassroots J Nat Resour 2(3):45–54
Kramer PJ (1983) Problems in water relations of plants and cells. Int Rev Cytol 85:253–286
Linebarger CRL, Boehlein SK, Sewell AK, Shaw J, Hannah LC (2005) Heat stability of maize endosperm ADP-glucose pyrophosphorylase is enhanced by insertion of a cysteine in the N terminus of the small subunit. Plant Physiol 139:1625–1634
Liu Z, Li D, Gong P, Wu K (2004) Life table studies of the cottonbollworm, Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae), on different host plants. Environ Entomol 33(6):1570–1576
Liu L, Xia W, Li H, Zeng H, Wei B, Han S, Yin C (2018) Salinity inhibits rice seed germination by reducing α-amylase activity via decreased bioactive gibberellin content. Front Plant Sci 9:275
Lo SN, Garceau JJ (1975) A spectrophotometric method for quantitative analysis of sugar mixtures containing known sugars. Can J Chem Eng 53(5):582–587
Masuko T, Minami A, Iwasaki N, Majima T, Nishimura S-I, Lee YC (2005) Carbohydrate analysis by a phenol–sulfuric acid method in microplate format. Anal Biochem 339(1):69–72
Mieog JC, Janecek S, Ral JP (2017) New insight in cereal starch degradation: identification and structural characterization of four α-amylases in bread wheat. Amylase 1:35–49
Mollania N, Khajeh K, Hosseinkhani S, Dabirmanesh B (2010) Purification and characterization of a thermo stablephytate resistant α-amylase from Geobacillus sp. LH8. Int J Biol Macromol 46:27–36
Mugford ST, Fernandez O, Brinton J, Flis A, Krohn N, Encke B, Feil R, Sulpice R, Lunn JE, Stitt M, Smith AM (2014) Regulatory properties of ADP glucose pyrophosphorylase are required for adjustment of leaf starch synthesis in different photoperiods. Plant Physiol 166(4):1733–1747
Nielsen J, Borchert T (2000) Protein engineering of bacterial α-amylases. Biochem Biophys Acta 1543:253–274
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Biol 49(1):249–279
Noman A, Bashir R, Aqeel M, Anwar S, Iftikhar W, Zainab M, Zafar S, Baloch S, Islam W, Adnan M, Moral M (2016) Success of transgenic cotton (Gossypium hirsutum L.): fiction or reality? Cogent Food Agric 2:1207844
Onder D, Akiscan Y, Onder S, Mert M (2009) Effect of different irrigation water level on cotton yield and yield components. Afr J Biotech 8:8
Papastylianou P, Argyrokastritis I (2014) Effect of limited drip irrigation regime on yield, yield components, and fiber quality of cotton under Mediterranean conditions. Agric Water Manag 142:127–134
Peng J, Liu J, Zhang L, Luo J, Dong H, Ma Y, Zhao X, Chen B, Sui N, Zhou Z, Meng Y (2016) Effects of soil salinity on sucrose metabolism in cotton leaves. PLoS ONE 11:e0156241
Pilon C (2015) Physiological responses of cotton genotypes to water-deficit stress during reproductive development. Theses and Dissertations Retrieved from https://scholarworks.uark.edu/etd/1268
Regina A, Kosar-Hashemi B, Ling S, Li Z, Rahman S, Morell M (2010) Control of starch branching in barley defined through differential RNAi suppression of starch branching enzyme IIa and IIb. J Exp Bot 61:1469–1482
Rosa M, Prado C, Podazza G, Interdonato R, González JA, Hilal M, Prado FE (2009) Soluble sugars–metabolism, sensing and abiotic stress: a complex network in the life of plants. Plant Signal Behav 4(5):388–393
Saatsaz M (2020) A historical investigation on water resources management in Iran. Environ Dev Sustain 22(3):1749–1785
Salamone PR, Kavakli IH, Slattery CJ, Okita TW (2001) Directed molecular evolution of ADP-glucose pyrophosphorylase. PNAS 99:1070–1075
Sayyad-Amin P, Borzouei A, Jahansooz MR, Ajili F (2016) Changes in photosynthetic pigments and chlorophyll-a fluorescence attributes of sweet-forage and grain sorghum cultivars under salt stress. J Biol Phys 42:601–620
Shareef M, Fanjiang Z, Dongwei G, Bo Z, Muhammad F (2018) Drought induced interactive changes in physiological and biochemical attributes of cotton (Gossypium hirsutum). Int J Agric Biol 20:539–546
Singh Y, Rao SS, Regar P (2010) Deficit irrigation and nitrogen effects on seed cotton yield, water productivity and yield response factor in shallow soils of semi-arid environment. Agric Water Manag 97:965–970
Snider JL, Collins GD, Whitaker J, Chapman KD, Horn P (2016) The impact of seed size and chemical composition on seedling vigor, yield, and fiber quality of cotton in five production environments. Field Crop Res 193:186–195
Souza P, Oliveira E, Magalhães P (2010) Application of microbial α-amylase in industry-a review. Braz J Microbiol 41:850–861
Stanley D, Fitzgerald AM, Farnden KJF, Macrae EA (2002) Characterisation of putative α-amylases from apple (Malus domestica) and Arabidopsis thaliana. Biol Bratisl 57(11):137–148
Statistical Analysis System (SAS) (2001) User’s Guide: Statistics, Version 8.2. SAS Institute, NC, USA
Taliercio E (2008) Isolation and characterization of an ADP-glucose pyrophosphorylase gene from Gossypium hirsutum L. J Cotton Sci 12:273–279
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Todka D, Matsushima S, Morohashi Y (2000) Water stress enhances β-amylase activity in cucumber cotyledons. J Exp Bot 51(345):739–745
Unknown (2020) World Water Development Report–Water and Climate Change (UNESCO WWAP). https://en.unesco.org/
Waqas MA, Kaya C, Riaz A, Farooq M, Nawaz I, Wilkes A, Li Y (2019) Potential mechanisms of abiotic stress tolerance in crop plants induced by Thiourea. Front Plant Sci 10:1336
Yang J, Yan R, Roy A, Xu D, Poisson J (2015) The I-TASSER suite: protein structure and function prediction. Nat Methods 12(1):7–15
Yildirim E, Turan M, Guvenc I (2008) Effect of foliar salicylic acid applications on growth, chlorophyll and mineral content of cucumber (Cucumis sativus L.) grown under salt stress. J Plant Nutr 31:593–612
Zhang L, Ma H, Chen T, Pen J, Yu S, Zhao X (2014) Morphological and physiological responses of cotton (Gossypium hirsutum L.) plants to salinity. PLoS ONE 9(11):e112807
Zhu X, Sun L, Kuppu S, Hu R, Mishra N, Smith J, Esmaeili N, Herath M, Gore MA, Payton P, Shen G, Zhang H (2018) The yield difference between wild-type cotton and transgenic cotton that expresses IPT depends on when water-deficit stress is applied. Sci Rep 8(1):2538–2538
Zonta JH, Bezerra JRC, Sofiatti V, Brandão ZN (2015) Yield of cotton cultivars under different irrigation depths in the Brazilian semi-arid region. Rev Bras Eng Agríc Ambient 19:748–754
Acknowledgements
This work was funded by a grant from the Cotton Research Institute, Gorgan (Project code 3-07-075195105). The authors declare that there is no conflict of interest.
Funding
This research was supported by cotton research institute of Iran from Elham Faghani project (code: 3-07-075195105).
Author information
Authors and Affiliations
Contributions
MK contributed to bioinformatics analysis and write bioinformatics data. EF contributed to project administration, funder acquisition; physiology methodology, writing original draft. MK contributed to bioinformatics analysis. AG-B contributed to editing English paper. MHR contributed to doing experiments in farm and laboratory.
Corresponding author
Ethics declarations
Conflict of interest
All co-authors have seen and agree with the contents of manuscript, and there is no financial interest to report. So, we know of no conflicts of interest associated with this publication.
Ethical approval and consent to participate
Not applicable.
Additional information
Editorial responsibility: S. Mirkia.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Faghani, E., Kolahi, M., Kazemian, M. et al. Effect of irrigation regimes on starch biosynthesis pathway, cotton (Gossypium hirsutum) yield and in silico analysis of ADP-glucose-pyrophosphorylase. Int. J. Environ. Sci. Technol. 19, 10809–10830 (2022). https://doi.org/10.1007/s13762-022-04281-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04281-x