Abstract
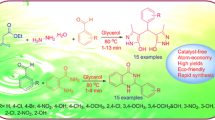
In this study, we have described a clean and facile condensation reaction of various aromatic aldehydes or cyclic ketones with 2-aminobenzamide under mild conditions in the presence of C–SO3H as an efficient catalyst derived from glycerol. This protocol furnishes an imperative avenue for the access of 2,3-dihydroquinazolin-4(1H)-ones in excellent yields with a range of structural diversity. The recovered catalyst has been engineered for subsequent runs with substantial activity.
Graphical Abstract











Similar content being viewed by others
References
D. Uraguchi, K. Sorimachi, M. Terada, J. Am. Chem. Soc. 126, 11804 (2004)
A. Domling, Chem. Rev. 106, 17 (2006)
D.J. Raman, M. Yus, Angew. Chem. Int. Ed. 44, 1602 (2005)
A. Corma, Chem. Rev. 95, 559 (1995)
T. Okuhara, Chem. Rev. 102, 3641 (2002)
B. Maheshwar Rao, G. Niranjan Reddy, T. Vijaikumar Reddy, B.L.A. Prabhavathi Devi, R.B.N. Prasad, J.S. Yadav, B.V. Subba Reddy, Tetrahedron Lett. 54, 2466 (2013)
V. Manneganti, R.B.N. Prasad, B.L.A. Prabhavathi Devi, Eur. J. Chem. 5, 167 (2014)
B.L.A. Prabhavathi Devi, K.N. Gangadhar, P.S.S. Prasad, B. Jagannadh, R.B.N. Prasad, Chemsuschem 2, 617 (2009)
K.N. Gangadhar, M. Vijay, R.B.N. Prasad, B.L.A. Prabhavathi Devi, Green Sustain. Chem. 3, 122 (2013)
G. Niranjan Reddy, B. Maheshwar Rao, M. Vijay, B.L.A. Prabhavathi Devi, R.B.N. Prasad, B.V. Subba Reddy, Can. J. Chem. 93, 341 (2015)
M.G. Arania, M.S. Niasaria, S. Naseh, Ultrason. Sonochem. 39, 494 (2017)
M.G. Arani, M.M. Arani, M.S. Niasari, J. Mol. Liq. 216, 59 (2016)
H. Wang, S. Xue, J. Mater. Sci. Mater. Electron. 27, 1 (2016)
M.G. Arani, M.M. Arani, M.S. Niasari, J. Mol. Catal. A Chem. 425, 31 (2016)
L.N. Moghadam, M.S. Niasari, J. Mol. Struct. 1146, 629 (2017)
H. Haghjooa, F.S. Sangsefidia, S.A. Hashemizadehb, M.S. Niasari, J. Mol. Liq. 225, 290 (2017)
B.L.V. Prabhavathi Devi, T. Vijai Kumar Reddy, K. Vijaya Lakshmi, R.B.N. Prasad, Bioresour. Technol. 153, 370 (2014)
P. Sivaguru, K. Parameswaran, M. Kiruthika, P. Vadivel, A. Lalitha, J. Iran. Chem. Soc. 12, 95 (2015)
K. Revathy, A. Lalitha, J. Iran. Chem. Soc. 12, 2045 (2015)
V. Murugan, M. Kulkarni, R.M. Anand, E.P. Kumar, B. Suresh, V.M. Reddy, Asian J. Chem. 18, 900 (2006)
G. Bonola, P.D. Re, M.J. Magistretti, E. Massarani, I. Setnikar, J. Med. Chem. 11, 1136 (1968)
Rexall Drug Co., U.S. Patent 3257397 (1966)
K. Okumura, T. Oine, Y. Yamada, G. Hayashi, M. Nakama, J. Med. Chem. 11, 348 (1968)
M.J. Hour, L.J. Huang, S.C. Kuo, Y. Xia, K. Bastow, Y. Nakanishi, E. Hamel, K.H. Lee, J. Med. Chem. 43, 4479 (2000)
H.L. Birch, G.M. Buckley, N. Davies, H.J. Dyke, E.J. Frost, P.J. Gilbert, D.R. Hannah, A.F. Haughan, M.J. Madigan, T. Morgan, W.R. Pitt, A.J. Ratcliffe, N.C. Ray, M.D. Richard, A. Sharpe, A.J. Taylor, J.M. Whitworth, S.C. Williams, Bioorg. Med. Chem. Lett. 15, 5335 (2005)
E. Cohen, B. Klarberg, J.R. Vaughan, J. Am. Chem. Soc. 81, 5508 (1959)
V. Alagarsamy, V.R. Solomon, M. Murugan, Bioorg. Med. Chem. 15, 4009 (2007)
J.I. Levin, P.I. Chan, T. Bailey, A.S. Katocs, A.M. Venkatesan, Bioorg. Med. Chem. Lett. 4, 1141 (1994)
Instituto De Angeli S.P.A. French Patent M 1893 (1963)
Shulton Inc. U.S. Patent 3265697 (1966)
N. Hirose, S. Kuriyama, S. Sohda, K. Sakaguchi, H. Yamamoto, Chem. Pharm. Bull. 21, 1005 (1973)
S. Schramm, E. Schmitz, E. Grundemann, J. Prakt. Chem. 326, 279 (1984)
J.P. Michael, Nat. Prod. Rep. 25, 166 (2008)
V. Alagarsamy, V.R. Solomon, K. Dhanabal, Bioorg. Med. Chem. 15, 235 (2007)
J. Imagawa, K. Sakai, Eur. J. Pharmacol. 131, 257 (1986)
R.Q. Dempcy, E.B. Skibo, Biochemistry 30, 8480 (1991)
M.S. Malamas, J. Millen, J. Med. Chem. 34, 1492 (1991)
R. Ramesh, A. Lalitha, RSC Adv. 5, 51188 (2015)
R. Ramesh, A. Lalitha, Res. Chem. Intermed. 41, 8009 (2015)
R. Ramesh, S. Maheswari, S. Murugesan, R. Sandhiya, A. Lalitha, Res. Chem. Intermed. 41, 8233 (2015)
R. Ramesh, P. Vadivel, S. Maheswari, A. Lalitha, Res. Chem. Intermed. 42, 7625 (2016)
R. Ramesh, A. Lalitha, ChemistrySelect 1, 2085 (2016)
R. Ramesh, R. Madhesh, J.G. Malecki, A. Lalitha, ChemistrySelect 1, 5196 (2016)
R. Ramesh, N. Nagasundaram, D. Meignanasundar, A. Lalitha, Res. Chem. Intermed. 43, 1767 (2017)
CrysAlis RED, Oxford Diffraction Ltd. Version 1.171.37.35g
O.V. Dolomanov, L.J. Bourhis, R.J. Gildea, J.A.K. Howard, H. Puschmann, J. Appl. Cryst. 42, 339 (2009)
G.M. Sheldrick, Acta Cryst. A64, 112 (2008)
Acknowledgements
R. Ramesh gratefully thank DST-Inspire Fellowship, New Delhi, India (No: DST/INSPIRE Fellowship/2012/690) for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ramesh, R., Sankar, G., Malecki, J.G. et al. Carbon–SO3H derived from glycerol: a green recyclable catalyst for synthesis of 2,3-dihydroquinazolin-4(1H)-ones. J IRAN CHEM SOC 15, 1–9 (2018). https://doi.org/10.1007/s13738-017-1202-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-017-1202-1