Abstract
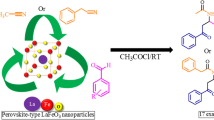
LiF nano cubic, with an average particle size of 21.6 nm, was synthesized sonochemically, using tetrabutylammonium fluoride as the fluoride source and characterized by SEM-EDX and XRD. This was applied, as a heterogeneous nano-catalyst, to synthesize some β-aryl-β-mercapto ketone derivatives via thia-Michael addition reactions with excellent yields at room temperature. In this catalytic system, the products of undesirable side reactions resulting from 1,2-addition, polymerization, bis-addition and oxidative coupling of thiols were not observed. The products were characterized by elemental analysis, IR, NMR and mass, and also single crystal X-ray determination for 3-(4-chlorophenylthio)-1,3-diphenylpropan-1-one, 1-phenyl-3-(phenylthio)-3-p-tolylpropan-1-one, 3-(p-tolylthio)-1-phenyl-3-p-tolylpropan-1-one and 3-(4-chlorophenylthio)-1-phenyl-3-p-tolylpropan-1-one. Following the recent search on the CSD, it is found that there is no report on the structure determination of [R1]C[H][CH2C(O)R2][S-Ar] β-aryl-β-mercapto ketones with the S-C6H5, S-C6H4-p-CH3 and S-C6H4-p-Cl segments (as S-Ar).







Similar content being viewed by others
References
Y. Hui, J. Jiang, W. Wang, W. Chen, Y. Cai, L. Lin, X. Liu, X. Feng, Angew. Chem. Int. Ed. 49, 4290 (2010)
Y. Qian, G.-Y. Ma, Y. Yang, K. Cheng, Q.-Z. Zheng, W.-J. Mao, L. Shi, J. Zhao, H.-L. Zhu, Bioorg. Med. Chem. 18, 4310 (2010)
C. González-Rodríguez, S.R. Parsons, A.L. Thompson, M.C. Willis, Chem. Eur. J. 16, 10950 (2010)
A. Kumar, Akanksha, Tetrahedron Lett. 48, 8730 (2007)
M.S. Abaee, S. Cheraghi, S. Navidipoor, M.M. Mojtahedi, S. Forghani, Tetrahedron Lett. 53, 4405 (2012)
M. Bandini, P.G. Cozzi, M. Giacomini, P. Melchiorre, S. Selva, A. Umani-Ronchi, J. Org. Chem. 67, 3700 (2002)
M.M. Alam, R. Varala, S.R. Adapa, Tetrahedron Lett. 44, 5115 (2003)
K. Suzuki, A. Ikegawa, T. Mukaiyama, Bull. Soc. Chem. Jpn. 55, 3277 (1982)
A. Sarkar, S.R. Roy, A.K. Chakraborti, Chem. Commun. 47, 4538 (2011)
S. Banerjee, J. Das, R.P. Alvarez, S. Santra, New J. Chem. 34, 302 (2010)
M. Verziu, M. Florea, S. Simon, V. Simon, P. Filip, V.I. Parvulescu, C. Hardacre, J. Catal. 263, 56 (2009)
M. Cavazza, F. Pietra, Tetrahedron Lett. 45, 3633 (2004)
S. Gao, C. Tseng, C.H. Tsai, C.-F. Yao, Tetrahedron 64, 1955 (2008)
A. Heydari, S. Khaksar, J. Akbari, M. Esfandyari, M. Pourayoubi, M. Tajbakhsh, Tetrahedron Lett. 48, 1135 (2007)
Z. Shobeiri, M. Pourayoubi, A. Heydari, T.M. Percino, M.A. Leyva Ramírez, C. R. Chim. 14, 597 (2011)
F.H. Allen, Acta Cryst. Sect. B 58, 380 (2002)
R. Sarraf-Mamoory, S. Nadery, N. Riahi-Noori, Chem. Eng. Comm. 194, 1022 (2007)
S.K. Garg, R. Kumar, A.K. Chakraborti, Synlett 1370 (2005)
S.K. Garg, R. Kumar, A.K. Chakraborti, Tetrahedron Lett. 46, 1721 (2005)
G.L. Khatik, G. Sharma, R. Kumar, A.K. Chakraborti, Tetrahedron 63, 1200 (2007)
Y. Abrouki, M. Zahouily, A. Rayadh, B. Bahlaouan, S. Sebti, Tetrahedron Lett. 43, 8951 (2002)
J. Skarżewski, M. Zielińska-Błajet, I. Turowska-Tyrk, Tetrahedron Asymmetry 12, 1923 (2001)
C.-M. Chu, S. Gao, M.N.V. Sastry, C.-F. Yao, Tetrahedron Lett. 46, 4971 (2005)
M. Zahouily, Y. Abrouki, A. Rayadh, S. Sebti, H. Dhimane, M. David, Tetrahedron Lett. 44, 2463 (2003)
J.S. Yadav, B.V.S. Reddy, G. Baishya, J. Org. Chem. 68, 7098 (2003)
K. Tokuno, M. Matsui, F. Miyoshi, Y. Asao, T. Ohashi, K. Kihara, Acta Cryst. Sect. C 42, 85 (1986)
M. Yaeghoobi, H. Khaledi, Z. Abdullah, N. Abd Rahman, Acta Cryst. Sect. E67, o1693 (2011)
G. Morgant, X. Labouze, B. Viossat, J.-C. Lancelot, N.H. Dung, Acta Cryst. Sect. C 52, 923 (1996)
S. Koda, T. Kimura, T. Kondo, H. Mitome, Ultrason. Sonochem. 10, 149 (2003)
Oxford Diffraction Ltd., Xcalibur CCD System, Crys Alis Software System, Version 1.171, (Oxford, England, 2006)
A. Altomare, G. Cascarano, C. Giacovazzo, A. Guagliardi, J. Appl. Cryst. 26, 343 (1993)
G.M. Sheldrick, Acta Cryst. Sect. A 64, 112 (2008)
L.J. Farrugia, J. Appl. Cryst. 32, 837 (1999)
Acknowledgments
The authors gratefully acknowledge the support of this work by the Ferdowsi University of Mashhad. We are grateful to CEITEC-Central European Institute of Technology for determining the structural analysis by single-crystal X-ray diffraction method (CZ.1.05/1.1.00/02.0068).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shobeiri, Z., Pourayoubi, M., Nečas, M. et al. Sonochemical synthesis of lithium fluoride nano cubic as an active and simple catalyst for thia-Michael addition process: synthesis and structural characterization of β-aryl-β-mercapto ketones. J IRAN CHEM SOC 12, 245–255 (2015). https://doi.org/10.1007/s13738-014-0479-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-014-0479-6