Abstract
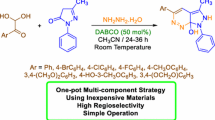
A study concerning the new substituted cinnoline synthesis is described. The use of a one-pot three-component method allows a simple regioselective and efficient synthesis of cinnoline derivatives via reaction of arylglyoxals with 1,3-cyclohexanedione and dimedone in the presence of hydrazine hydrate.






Similar content being viewed by others
References
G. Cignarella, D. Barlocco, G.A. Pinna, M.M. Curzu, Acta Chim. Slov. 41, 173 (1994)
G. Cignarella, D. Barlocco, G.A. Pinna, M. Loriga, M.M. Curzu, O. Tofanetti, M. Germini, P. Cazzulani, E. Cavaletti, J. Med. Chem. 32, 2277 (1989)
J.W. Lown, A.R. Morgan, S.F. Yen, Y.H. Wang, W.D. Wilson, Biochemistry 24, 4028 (1990)
K. Miyamoto, J. Matsumoto, S. Nakamura, Chem. Abstr. 113, 97619 (1990)
A.G. Siegfried, Fr. 1,393,596 (1965)
S.M. Yarnal, V.V. Badiger, J. Matsumoto, Chem. Abstr. 104, 224864 (1986)
P. Barraja, P. Diana, A. Lauria, A. Passannanti, A.N. Almerico, C. Minnei, S. Longu, D. Cengiu, C. Musiu, P.L. Colla, Bioorg. Med. Chem. 7, 1591 (1999)
G. Cirrincione, E. Almerico, P. Barraja, F. Mingoia, Farmaco 50, 849 (1995)
J. Savaranan, K.S. Manjunath, Indian J. Pharm. Sci. 60, 330 (1998)
M.S. Abbady, S.M. Radwan, E.A. Bakhite, Indian J. Chem. Sec B 32, 1281 (1993)
E. Gavini, C. Juliano, A. Mule, G. Pirisino, G. Murineddu, G.A. Pinna, Arch Pharm (Weinheim) 333, 341 (2000)
V. Saxena, S.K. Maiti, N. Kumar, A.K. Sharma, Indian J. Anim. Sci. 78, 1250 (2008)
S. Vikas, S. Darbhamulla, Afr. Health Sci. 9, 275 (2009)
F. Vargas, T. Zoltan, C. Rivas, A. Ramirez, T. Cordero, Y. Díaz, C. Izzo, Y.M. Cárdenas, V. López, L. Gómez, J. Ortega, A. Fuentes, J. Photochem. Photobiol. B: Biol. 92, 83 (2008)
C. Lunniss, C. Eldred, N. Aston, A. Craven, K. Gohil, B. Judkins, S. Keeling, L. Ranshaw, E. Robinson, T. Shipley, N. Trivedi, Bioorg. Med. Chem. Lett. 20, 137 (2010)
R.A. Conrad, W.A. White, US 4,379,929 (1983)
Y. Yu, S.K. Singh, A. Liu, T.-K. Li, L.F. Liu, E. LaVoie, J. Bioorg. Med. Chem. 11, 1475 (2003)
T. Nakao, M. Takai, H. Fukamachi, H. Ohashi, Jpn. Kokai Tokkyo Koho JP 11 189,586 [99189,586]
E. Lunt, K. Washbourn, W.R. Wragg, J. Chem. Soc. C. 1152 (1968)
Hennequin, Laurent Francois Andre. PCT Int. Appl. WO 02 12,228
N. Garcia-Dominguez, E. Ravina, L. Santana, C. Teran, G. Garcia-Mera, F. Orallo, M. Crespo, J.A. Fontenla, Arch. Pharma (Weinheim) 321, 735 (1988)
J. Geraint Ger. Offen. 2,438,413 (1975)
D. Holland, G. Jones, P.W. Marshall, G.D. Tringham, J. Med. Chem. 19, 1225 (1976)
J.C.E. Simpson, in Condensed Pyridazine and Pyrazine Rings. The Chemistry of Heterocyclic Compounds, ed. by A. Weisberg, (Interscience publishers, New York-London 1953), p. 347
G.M. Singerman, in The Chemistry of Heterocyclic Compounds, ed. by R. N. Castle, (Interscience publishers, New York. 1973), p. 1
N.J. Leonard, Chem. Rev. 37, 269 (1945)
T.L. Jacobs, in Heterocyclic Compounds, ed. by R. C. Elderfield, (Wiley, New York. 1957) p. 136
N. Haider, W. Holzer, “Product Class 9: Cinnolines,” in Science of Synthesis, 2004, pp. 251–313
D.J. Brown, Cinnolines and Phthalazines, Suppl. II, The Chemistry of Heterocyclic Compounds (Wiley, New York, 2005)
F.M. Abdelrazek, P. Metz, N.H. Metwally, S.F. El-Mahrouky, Arch. Pharm. Chem. Life Sci. (Weinheim) 339, 456 (2006)
B. Wünsch, S. Nerdinger, G. Höfner, Liebigs Ann. 7, 1303 (1995)
P.W. Neber, G. Knoller, K. Herrst, A. Trissler, Justus Liebigs Ann. Chem. 471, 113 (1929)
E.J. Alford, K. Schofield, J. Chem. Soc. 2102 (1952)
K. Pfannstiel, J. Janecke, Ber. Dtsch. Chem. Ges. 75, 1096 (1942)
H.E. Baumgarten, C.H. Anderson, J. Am. Chem. Soc. 80, 1981 (1958)
M.A.M. Gomaa, Tetrahedron Lett. 44, 3493 (2003)
M.S. Shvartsberg, I.D. Ivanchikova, Tetrahedron Lett. 41, 771 (2000)
H.J. Barber, E. Lunt, J. Chem. Soc. C. 1156 (1968)
V. Richter, Ber. Dtsch. Chem. Ges. 16, 677 (1883)
I.D. Jurberg, F. Gagosz. J. Organomet. Chem. 696, 37 (2011)
G. Halbritter, F. Knoch, H. Kisch, J. Organomet. Chem. 492, 87 (1995)
U. Dürr, F.W. Heinemann, H. Kisch, J. Organomet. Chem. 558, 91 (1998)
R.L. Zey, J. Heterocycl. Chem. 25, 847 (1988)
C. Zhu, M. Yamane, Tetrahedron 67, 4933 (2011)
O. Diels, K. Alder, Justus Liebigs Ann. Chem. 450, 237 (1926)
J. Khalafy, M. Rimaz, M. Ezzati, R.H. Prager, Bull. Korean Chem. Soc. 33, 2890 (2012)
Acknowledgments
The authors are grateful to Urmia University for financial support and to Dr. A. Choudhury (Missouri University of Science and Technology, USA) for his assistance with X-ray crystallography.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khalafy, J., Ezzati, M., Rimaz, M. et al. An efficient and facile regioselective synthesis of new substituted (E)-1-(3-aryl-7,8-dihydrocinnoline-5(6H)-ylidene)hydrazines and (1E,2E)-1,2-bis(3-aryl-7,8-dihydrocinnoline-5(6H)-ylidene)hydrazines. J IRAN CHEM SOC 11, 1067–1074 (2014). https://doi.org/10.1007/s13738-013-0378-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-013-0378-2