Abstract
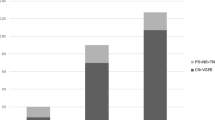
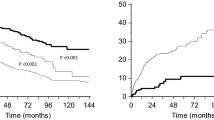
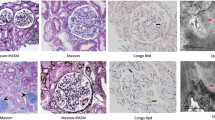
We report a 58-year-old Japanese woman who presented with nephrotic syndrome. Steroid therapy and cyclosporine A administration were initiated, but hematological remission and renal response were not achieved. Renal biopsy revealed amyloid deposits in the mesangial region and the small arteries. Proteomic analysis based on laser microdissection and mass spectrometry showed that the amyloid deposits were composed of the constant region of the lambda light chain. She received vincristine, adriamycin, and dexamethasone therapy followed by high-dose melphalan and autologous stem cell transplantation, resulting in hematological complete remission and renal response with negative urinary Bence-Jones protein and proteinuria. Renal biopsy was performed four times during follow-up, demonstrating that amyloid deposits decreased gradually, while glomeruli showing global sclerosis increased from 3 to 62%. This case suggests that glomerular amyloid deposits can be cleared via tissue remodeling, if stem cells producing amyloid precursors are completely replaced by unrelated cells after stem cell transplantation.



Similar content being viewed by others
Abbreviations
- AL:
-
Amyloid light chain
- VAD:
-
Vincristine, adriamycin, and dexamethasone
- HDM:
-
High-dose melphalan
- ASCT:
-
Autologous stem cell transplantation
- PSL:
-
Prednisolone
- CyA:
-
Cyclosporine A
- BJP:
-
Bence-Jones protein
- PAM:
-
Periodic acid methenamine silver
- PAS:
-
Periodic acid-Schiff
- PAM:
-
Periodic acid silver methenamine
- LMD/MS:
-
Laser microdissection and mass spectrometry
- RBx:
-
Renal biopsy
References
Milani P, et al. Light chain amyloidosis. Mediterr J Hematol Infect Dis. 2018;10(1):e2018022.
Dember LM. Amyloidosis-associated kidney disease. J Am Soc Nephrol. 2006;17(12):3458–71.
Said SM, Sethi S, Valeri AM, et al. Renal amyloidosis: origin and clinicopathologic correlations of 474 recent cases. Clin J Am Soc Nephrol. 2013;8(9):1515–23.
Kuroda T, Tanabe N, Kobayashi D, et al. Significant association between renal function and amyloid-positive area in renal biopsy specimens in AL amyloidosis. BMC Nephrol. 2012;13:118.
Wechalekar AD, Gillmore JD, Hawkins PN. Systemic amyloidosis. Lancet (London, England). 2016;387(10038):2641–54.
Nasr SH, Said SM, Valeri AM, et al. The diagnosis and characteristics of renal heavy-chain and heavy/light-chain amyloidosis and their comparison with renal light-chain amyloidosis. Kidney Int. 2013;83(3):463–70.
Gono T, Matsuda M, Dohi N, et al. Nephrotic syndrome due to primary AL amyloidosis, successfully treated with VAD and subsequent high-dose melphalan followed by autologous peripheral blood stem cell transplantation. Intern Med. 2003;42(1):72–7.
Perz JB, Schonland SO, Hundemer M, et al. High-dose melphalan with autologous stem cell transplantation after VAD induction chemotherapy for treatment of amyloid light chain amyloidosis: a single centre prospective phase II study. Br J Haematol. 2004;127(5):543–51.
Jaccard A, Moreau P, Leblond V, et al. High-dose melphalan versus melphalan plus dexamethasone for AL amyloidosis. NEJM. 2007;357(11):1083–93.
Sethi S, Theis JD, Leung N, et al. Mass spectrometry-based proteomic diagnosis of renal immunoglobulin heavy chain amyloidosis. Clin J Am Soc Nephrol. 2010;5(12):2180–7.
Cibeira MT, Sanchorawala V, Seldin DC, et al. Outcome of AL amyloidosis after high-dose melphalan and autologous stem cell transplantation: long-term results in a series of 421 patients. Blood. 2011;118(16):4346–52.
Milani P, Merlini G, Palladini G. Novel therapies in light chain amyloidosis. Kidney Int Rep. 2018;3(3):530–41.
Gillmore JD, Hawkins PN. Amyloidosis and the respiratory tract. Thorax. 1999;54(5):444–51.
Katoh N, Matsuda M, Tsuchiya-Suzuki A, et al. Regression of gastroduodenal amyloid deposition in systemic AL amyloidosis after intensive chemotherapies. Br J Haematol. 2011;153(4):535–8.
Couchman JR, Beavan LA, McCarthy KJ. Glomerular matrix: synthesis, turnover and role in mesangial expansion. Kidney Int. 1994;45(2):328–35.
Yamazaki O, Ubara Y, Suwabe T, et al. Successful treatment of primary AL amyloidosis by VAD therapy, high-dose melphalan, and autologous peripheral stem cell transplantation. J Clin Exp Nephrol. 2009;13(5):522–5.
Iwadate D, Hasegawa E, Hoshino J, et al. The long-term outcomes after VAD plus SCT therapy in a patient with AL amyloidosis and severe factor X deficiency. Intern Med. 2018;57(5):701–6.
Nakayama M, Kashiwagi M, Katafuchi R, et al. Resolution of primary amyloidosis by melphalan and prednisolone: a case report. Clin Nephrol. 2005;63(3):215–20.
Acknowledgements
We are grateful to Masanori Suzuki for performing electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors also declare that they have no conflicts of interest.
Ethical standards
The present study adhered to the Declaration of Helsinki and the patient gave written consent for the details of her case to be published.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Toriu, N., Sawa, N., Hiramatsu, R. et al. Regression of renal amyloid deposits by VAD therapy plus autologous stem cell transplantation in a patient with primary AL amyloidosis. CEN Case Rep 9, 6–10 (2020). https://doi.org/10.1007/s13730-019-00416-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-019-00416-1