Abstract
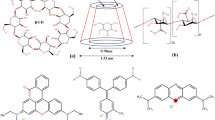
A novel type of polyurethane elastomer (PUE) containing nanocomposites of sodium alginate thiacalix[4]arene macrocycles including sodium alginate-thiacalix[4]arene and sodium alginate-tetra-sulfonated thaiacalix[4]arene (SA-TSTCA) was prepared. The key role of these nanocomposites merged into polyurethane elastomers is to function as chain extenders. To provide profound insight into the structural, thermal, mechanical, morphological and hydrophilic properties of the samples, several techniques were employed, namely Fourier transform infrared spectroscopy, thermal gravimetric analysis, scanning electron microscopy (SEM) and differential scanning calorimetry (DSC). Also the results were compared with the results of polyurethane elastomer based on glycerol (Gly) as chain extender. The results showed that the samples extended with nanocomposites as chain extender have better thermal and mechanical properties and between them the PUE containing SA-TSTCA showed higher thermal and mechanical properties. Furthermore, these elastomers have been used as adsorbent for removal of Malachite green (MG) for investigation of the effect of sulfonated groups introduced on the chain extender. The results revealed that SA-TSTCA-based polyurethane elastomer nanocomposite could well adsorb Malachite green from aqueous media by a batch adsorption method. The pseudo-first-order and pseudo-second-order kinetic models were employed to provide an in-depth study of the adsorption capacity of MG, suggesting that its adsorption onto PUE/SA-TSTCA elastomer was in accordance with pseudo-second-order kinetic process. A possible mechanism of adsorption was suggested where π–π stacking interactions, H-bonding interaction and electrostatic attraction controlled the MG adsorption.








Similar content being viewed by others
References
Narimani F, Lakouraj MM (2015) Swelling behavior and characterization of alcohol-specific superabsorbing gels based on acrylic acid and allyl tetrasodium thiacalix[4]arene tetrasulfonate. J Polym Res 22:28
Lakouraj MM, Norouzian RF, Balou S (2015) Preparation and cationic dye adsorption of novel Fe3O4 supermagnetic/thiacalix[4]arene tetrasulfonate self-doped/polyaniline nanocomposite: kinetics, isotherms, and thermodynamic study. J Chem Eng Data 60:2262–2272
Mohammadi A, Lakouraj MM, Barikani M (2014) Preparation and characterization of p-tert-butyl thiacalix[4]arene imbedded flexible polyurethane foam: An efficient novel cationic dye adsorbent. React Funct Polym 83:14–23
Ghaedi M, Ansari A, Habibi MH, Asghari AR (2014) Removal of malachite green from aqueous solution by zinc oxide nanoparticle loaded on activated carbon: kinetics and isotherm study. Eng Chem 20:17–28
Lakouraj MM, Mojerlou F, Zare EN (2014) Nanogel and superparamagnetic nanocomposite based on sodium alginate for sorption of heavy metal ions. Carbohydr Polym 106:34–41
Hosseini J, Zare EN, Ajloo D (2019) Experimental and theoretical calculation investigation on effective adsorption of lead(II) onto poly(aniline-co-pyrrole) nanospheres. J Mol Liq 296:111789
Zare EN, Lakouraj MM, Kasirian N (2018) Development of effective nano-biosorbent based on poly m-phenylenediamine grafted dextrin for removal of Pb(II) and methylene blue from water. Carbohydr Polym 201:539–548
Zare EN, Lakouraj MM (2014) Biodegradable polyaniline/dextrin conductive nanocomposites: synthesis, characterization, and study of antioxidant activity and sorption of heavy metal ions. Iran Polym J 23:257–266
Dong SH, Zheng B, Wang F, Huang F (2014) Supramolecular polymers constructed from macrocycle-based host–guest molecular recognition motifs. Account Chem Res 47:1982–1994
Kaya A, Onac C, Alpoguz HK, Yilmaz A, Ata N (2016) Removal of Cr(VI) through calixarene based polymer inclusion membrane from chrome plating bath water. Chem Eng J 283:141–149
Xiangdong ZH, Yuchen L, Chao ZH, Shicheng ZH, Jianmin CH (2014) Novel and high-performance magnetic carbon composite prepared from waste hydrochar for dye removal. ACS Sustain Chem Eng 2:969–977
Rudnicki P, HubickiZ Kołodynska D (2014) Evaluation of heavy metal ions removal from acidic waste water streams. J Chem Eng 252:362–373
Lofrano G, Carotenuto M, Libralato G, Domingos RF, Markus A (2016) Polymer functionalized nanocomposites for metals removal from water and wastewater: an overview. Water Res 92:22–37
Xiaoliang Q, Lei L, Liangliang SH, Zhipeng L, Tao Q, Yuna Q, Xuan W, Xiong W, Qianwen G, Jianliang SH (2019) Efficient decontamination of lead ions from wastewater by salecan polysaccharide-based hydrogels. ACS Sustain Chem Eng 7:11014–11023
Xiaoliang Q, Ruona L, Mengyu CH, Zhipeng L, Tao Q, Yuna Q, Shengzhe ZH, Minchao L, Qiankun Z, Jianliang SH (2019) Removal of copper ions from water using polysaccharide-constructed hydrogels. Carbohydr Polym 209:101–110
Xiaoliang Q, Zhipeng L, Liangliang SH, Tao Q, Yuna Q, Shengzhe ZH, Minchaol L, Qiankun Z, Jianliang SH (2019) Highly efficient dye decontamination via microbial salecan polysaccharide-based gels. Carbohydr Polym 219:1–11
Lakouraj MM, Hasanzadeh F, Zare EN (2014) Nanogel and super-paramagnetic nanocomposite of thiacalix[4]arene functionalized chitosan: synthesis, characterization and heavy metal sorption. Iran Polym J 23:933–945
Liua Q, Yang B, ZhangL Huanga R (2015) Adsorption of an anionic azo dye by cross-linked chitosan/bentonite composite. J Biol Macromol 72:1129–1135
Mohammadi A, Lakouraj MM, Barikani M (2015) Synthesis and investigation of properties of thiacalix[4]arene-based polyurethane elastomers. Polym Int 64:421–429
Daemi H, Barikani M, Barmar M (2013) Compatible compositions based on aqueous polyurethane dispersions and sodium alginate. Carbohydr Polym 92:490–496
Mojerlou F, Lakouraj MM, Barikani M, Mohammadi A (2019) Highly efficient polyurethane membrane based on nanocomposite of sulfonated thiacalix[4]arene-sodium alginate for desalination. Carbohydr Polym 205:353–361
Xiaoliang Q, Xianqin T, Ting S, Lei L, Qianwen G (2019) Honeycomb-like hydrogel adsorbents derived from salecan polysaccharide for wastewater treatment. Cellulose 26:8759–8773
Xiaoduo L, Jiefu T, Yuanyuan L, Ningfei S, Shu M, Yong X, Ziyu CH (2019) Enhanced dyes adsorption from wastewater via Fe3O4 nanoparticles functionalized activated carbon. J Hazard Mater 373:397–407
Mohammadi A, Barikani M, Barmar M (2013) Effect of surface modification of Fe3O4 nanoparticles on thermal and mechanical properties of magnetic polyurethane elastomer nanocomposites. J Mater Sci 48:7493–7502
Hua M, Zhang S, Pan B, Zhang W, Lv L, Zhang Q (2012) Heavy metal removal from water/wastewater by nanosized metal oxides: a review. J Hazard Mater 211–212:317–331
Yang D, Zou Y, Wang X, Yu S, Wen T, Wang X (2017) Rational design and synthesis of monodispersed hierarchical SiO2@layered double hydroxide nanocomposites for efficient removal of pollutants from aqueous solution. J Chem Eng 323:143–152
Pan B, Zhang W, Lv L, Zhang Q, Zhang S (2009) Development of polymeric and polymer-based hybrid adsorbents for pollutants removal from waters. J Chem Eng 151:19–29
Zheng K, Pan B, Zheng Q, Zhang W, Pan B, Han Y (2007) Enhanced adsorption of p-nitroaniline from water by a carboxylated polymeric adsorbent. Sep Purif Technol 57:250–256
Gong Q, Wu J, Gong X, Fan Y, Xia H (2013) Smart polyurethane foam with magnetic field controlled modulus and anisotropic compression property. RSC Adv 3:3241–3248
Benavides R, Oenning LW, Paula MMS, Silva LDA (2014) Properties of polystyrene/acrylic acid membranes after sulphonation reactions. J New Mater Electrochem Syst 17:049–132
Mohagheghian A, Vahidi-Kolur R, Pourmohseni M, Yang JK, Shirzad-Siboni M (2015) Application of scallop shell-Fe3O4nano-composite for the removal azodye from aqueous solutions. Water Air Soil Pollut 226:321
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mojerlou, F., Barikani, M., Lakouraj, M.M. et al. Efficiency of polyurethane elastomer containing sulfonated groups as chain extender in removing Malachite green. Iran Polym J 29, 1057–1069 (2020). https://doi.org/10.1007/s13726-020-00861-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13726-020-00861-1