Abstract
Purpose of the Review
The use of bioelectrical impedance analysis (BIA) for monitoring body composition during the ketogenic diet has experienced a rapid surge. This scoping review aimed to assess the validity of procedures applying BIA in the ketogenic diet and to suggest best practices for optimizing its utilization.
Recent Findings
We conducted a systematic scoping review of peer-reviewed literature involving BIA for assessing body composition in individuals adhering to a ketogenic diet. Searches of international databases yielded 1609 unique records, 72 of which met the inclusion criteria and were reviewed. Thirty-five studies used foot-to-hand technology, 34 used standing position technology, while 3 did not declare the technology used. Raw bioelectrical parameters were reported in 21 studies. A total of 196 body mass components were estimated, but predictive equations were reported in only four cases.
Summary
Most research on BIA during ketogenic diets did not report the equations used for predicting body composition, making it impossible to assess the validity of BIA outputs. Furthermore, the exceedingly low percentage of studies reporting and analyzing raw data makes it challenging to replicate methodologies in future studies, highlighting that BIA is not being utilized to its full potential. There is a need for more precise technology and device characteristics descriptions, full report of raw bioelectrical data, and predictive equations utilized. Moreover, evaluating raw data through vectorial analysis is strongly recommended. Eventually, we suggest best practices to enhance BIA outcomes during ketogenic diets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Body Composition Basics
Body composition describes the components of body mass, which can be organized into five levels in an increasing order of complexity [1]. Starting from the fifth and last level, defined as the whole-body level, dimensional characteristics such as body mass, stature, breadths, lengths, and circumferences allow for a preliminary analysis of body composition. These parameters can be measured and evaluated with inexpensive and easy-to-use tools, such as scales, stadiometers, bone calipers, or measuring tapes. Notable, these dimensions result from the combination of different tissues, whose accurate investigation requires the use of more sophisticated and costly methods, such as imaging techniques [2]. Indeed, the tissue level includes adipose tissue, skeletal muscle mass (SMM), as well as bone tissue, along with other components specific to organs and systems. Each tissue is consisted of different cells immersed in the extracellular fluid, each characterized by a different amount of water inside. Indeed, the percentage of water content varies from around 10% in adipocytes to approximately 80% in myocytes, which is typical for these cell types [3]. Many of these cells engage in metabolic activity, contributing to whole body energy expenditure and oxygen consumption. Collectively, they constitute the body cell mass (BCM), which can be measured using neutron activation techniques [4]. Just as tissues are composed of cells, these cells derive from the combination of various molecules such as lipids, water, minerals, and proteins. At this level, parameters as fat mass (FM), fat-free mass (FFM) or lean body mass (LBM), total body water (TBW), and bone mineral content (BMC) are among the most well-known and monitored in research both for their relationships with health and due to the wide range of tools enabling their analysis [5, 6]. Smaller elements like atoms are part of the first level of body composition and represent the building blocks of any mass. Aside from the atomic level, whose components are challenging to quantify and thus receive limited consideration in research, the following four levels of body composition are illustrated in Fig. 1.
Main body mass components at molecular, cellular, tissue, and whole-body level of body composition. * = Formed by all cells involved in metabolic processes, including the protoplasm in fat cells; FFM= fat-free mass; LBM= lean body mass; LM= lean mass; ALM= Appendicular lean mass; ICW= Intracellular water; ECW= Extracellular water; NMA= nonmetabolically active; IAT= Internal adipose tissue formed by visceral and non-visceral adipose tissue; SAT= Subcutaneous adipose tissue
The Evolution of Bioelectrical Impedance Analysis
Among the most used tools in scientific research for monitoring body composition is bioelectrical impedance analysis (BIA) [5, 7]. This is due to its ease of use, portability, low cost, and the wide range of parameters it allows for evaluation. BIA is based on the conductive properties of the tissues, where fluids represent the main source of resistance (R) and cellular membranes and other tissue interfaces represent capacitive elements, identified in the reactance (Xc) [4]. The arctangent between R and Xc, calculable as (Xc/R)*(180/π), results in the phase angle (PhA), a biomarker positively associated with the intracellular-to-extracellular water (ICW/ECW) ratio [8, 9]. The history of BIA begins in 1950 when American cardiologist Hans Nyboer discovers the relationship between blood flow and bioelectrical parameters [10]. Subsequently, in 1962, French physician August Luis Thomasset developed the first device to measure bioimpedance in a supine position using a foot-to-hand technology [11]. A few years later, Hoffer et al. (1969) allowed the foot-to-hand technology to evolve into a tetrapolar method by using two pairs of electrodes instead of the two needles previously employed by Thomasset. The clear relationship between bioimpedance and body composition parameters [13] led to the development of the first equations to predict TBW and FFM [14,15,16]. At this point, FM began to be calculated as the difference between FFM and body mass, even after obtaining FFM under the assumption that it is approximately 73% composed of fluids [17]. These calculations and assumptions, not always valid in all contexts, led to defining BIA as a hydration-dependent method, especially for estimating FM [3, 18]. Subsequently, multicomponent models were used to develop prediction equations for FM, making BIA no longer entirely dependent on hydration status [19, 20]. In 1994, Piccoli et al. [21] suggested that for BIA to achieve high performance in quantifying body composition, a large number of predictive equations specific to age, gender, level of physical activity, and geographical characteristics would be necessary. Alongside this criticism, Piccoli et al. [21] proposed a remedy that involved the analysis of raw data, which he defined as bioelectrical impedance vector analysis (BIVA). In BIVA, R and Xc are standardized based on the subject's stature and considered on the x-axis and y-axis of a Cartesian plane, respectively. The resulting point/vector in representative of the fluid content and its distribution between intra and extracellular compartments [21]. Indeed, the vector length is inversely associated with TBW, while the vector direction graphically represent the PhA [21]. The history of BIA unfolds in the subsequent years, marked by the development of the first equations for predicting skeletal muscle mass (SMM) utilizing magnetic resonance as a reference method [22]. This represented a significant advantage for BIA since FFM broadly encompasses everything not attributable to body fat, thus including a wide variety of components, some of which are metabolically more active (e.g., muscle) than others (bone). In fact, it is known that dosing certain macronutrients would be more appropriate when conducted relative to kilograms of SMM rather than FFM, as in the case of proteins in diets aimed at muscle hypertrophy [23, 24]. Furthermore, there are studies in which SMM increased, even though FFM remained unchanged following specific interventions [25, 26]. This underscores how monitoring FFM alone can mask substantial changes, akin to the situation when observing overall body weight without dissecting it into its relevant components [25]. Advancements in BIA include the production of new device technologies enabling measurements in a standing position, or analyzers integrated into joysticks (hand-to-hand) and home scales (leg-to-leg) [27, 28]. Unfortunately, the use of different technologies results in different outputs, thus preventing the comparison of data obtained from different devices. [29•, 30].While more than 100 equations were available at the end of 2000s [31], BIVA innovation continues with new references for different populations [32••, 33, 34] and a new approach based on standardizing R and Xc on arm, waist, and calf circumferences called specific BIVA [35], capable of providing information about FM in addition to the ICW/ECW ratio [9, 36]. Figure 2 summarizes the key events in the history of BIA.
The Utility of BIA in Ketogenic Diet
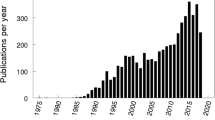
The use of BIA is beneficial in all cases where it is necessary to monitor changes in body composition, such as in the context of nutritional interventions. Among the different diets, the ketogenic diet is one of the most controversial that has shown, in the last years, a remarkable surge of scientific studies. Ketogenic diet is a low-carbohydrate, normal protein, high-fat dietary approach designed to induce a metabolic state called ketosis, where the body primarily utilizes fat for energy production [37]. The ketogenic diet (also defined as protein sparing modified fast) [38], was used for the first time as medical therapy by Dr. Wilder in 1921 as dietary approach to epilepsy in alternative to complete fasting [39]. The term “protein sparing modified fast” can be explained by the tentative to reach the metabolic state called ketosis (usually obtained during fasting) without an increased/excessive protein catabolism. In 1926 the first 4:1 ketogenic diet protocol was published [40]; the protocol involved1 g/protein/kg/day, 10 to 15 g/carbohydrates/day, and the remainder of the calories in fat (CHO + PRO: FAT grams, ratio 4:1). This proportion has been substantially maintained along the years [41,42,43,44] with some modifications but, in general, the scheme is based on less than 20 /30 g of carbohydrates or less than 5% of total daily energy intake from carbohydrate [45]. Recently, a clear classification of different types of ketogenic diets has been published [46]. In the recent year the interest about Ketogenic diet exceeded the limits of the original use in epilepsy and moved to include other clinical conditions such as inflammation [47, 48], metabolic syndrome, type 2 diabetes, polycystic ovary syndrome, cancer and others not communicable diseases [37]. The ketogenic diet is emerging as a cornerstone in public health strategies and interventions, given its potential to swiftly mitigate all modifiable risk factors, particularly addressing the complex issue of obesity and its associated metabolic sequelae [49••].
The suggested positive effects exerted by the ketogenic diet on health, relay on the “physiological ketosis” described by Hans Krebs in 1966 [50] who remarked the differences from the pathological ketosis (e.g., in decompensated diabetes [51]). Whilst the pathological ketosis is characterized by low systemic pH, no insulin, hyperglycaemia and very high level of ketone bodies (> 7/8 mmol/L), the physiological one has normal pH, low but within physiological ranges of both insulin and blood glucose. The levels of ketone bodies, produced from fats by the liver during carbohydrate restriction, varies typically between 0.3 up to 4 mmol/L [37, 44]. As said, ketone bodies are produced mainly in the liver (ketogenesis) from Acetyl-CoA. The condensation of two acetyl CoA leads to the production of an acetoactylCoa in a reaction catalyzed by the acetoacetyl-CoA thiolase, then the acetoacetyl Coa becomes 3HMGCoA through the addition of a third acetyl-CoA (catalyzed by the mitochondrial HMG-CoA synthetase). The HMG-CoA lyase catalyzes the passage from HMG-CoA to acetoacetate plus acetyl-CoA. Acetoacetate is the first ketone body which can be reduced by the NADH-dependent β-hydroxybutyrate dehydrogenase into the other ketone body: the β-Hydroxybutyrate and, by a spontaneous decarboxylation, to acetone, the third, volatile ketone. ketone bodies are not merely a byproduct of a CHO restricted condition but act as metabolic agents on different pathways [52]. One of the main fields of ketogenic diets’ use is weight control or weight loss and relatively changes of body composition variables. Considering fluids, the ketogenic diet typically leads to a significant reduction in carbohydrate intake, which in turn causes the body to release stored glycogen. Glycogen is stored with water, so as glycogen is depleted, the body also loses water weight. This initial loss of fluids is often noticeable, and monitoring fluid changes helps individuals understand short-term shifts in weight and hydration status. Moreover, during low carbohydrate availability periods, preserving skeletal muscle mass is a key concern for individuals. In fact, monitoring changes in SMM helps individuals ensure that their weight loss is predominantly from fat, rather than muscle. Given the primary body composition parameters of interest in ketogenic diet research, the utilization of BIA is progressively on the rise within this particular context [53].
Aim of the Study
Recently, it has been pointed out that BIA is not always applied with the correct procedures or fully exploited to its potential [28, 32••, 54]. This is due to the rapid evolution of BIA and the numerous innovations stemming from various studies that would need to be summarized. The list of procedures that delineate a proper BIA begins with an extensive description of device technology and the raw measured parameters. These procedures require the selection of specific predictive equations tailored to the subject under examination, and reporting them is crucial not only for ensuring the validity of the outputs but also for enabling future comparisons, given that each equation can yield different results [28, 31]. For these reasons, evaluating raw parameters through BIVA, in conjunction with body composition estimations, can facilitate a more comprehensive and qualitative analysis [32••, 55]. Therefore, the objective of this review was to systematically select all studies in which BIA has been used to analyze body composition in ketogenic diet interventions and consider whether the methods applied can be considered satisfactory. The hypothesis underlying this scoping review was that the BIA procedures might have shortcomings or that it had not always been utilized to its full capacity. Given the potential of BIA in evaluating parameters of particular interest to the ketogenic diet, the secondary goal was to define best practices to optimize its use, ensuring the validity of BIA-based assessments.
Methods
Search Strategy
The present study was carried out following the Preferred Reporting Items for Systematic reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guidelines [56]. The literature search was carried out by consulting the main biomedical databases such as Pubmed (Medline), and Scopus on Dec 31st, 2023, identifying potential eligible studies without any restriction related to year of publication. We composed different query strings depending on the variability of the databases functioning, using the following terms: ((ketogenic diet) OR (keto diet)) AND ((body composition) OR (fat mass) OR (body fluids) OR (total body water) OR (extracellular water) OR (intracellular water) OR (FM) OR (TBW) OR (ECW) OR (ICW) OR (bioelectrical impedance analysis) OR (bioimpedance) OR (BIA)). To ensure the inclusion of all potentially relevant sources, supplementary records were searched through cross-referencing.
Eligibility Criteria
Articles were included in the review if they met the following inclusion criteria: i) using BIA for assessing body composition in keto diet (< 50 g/d, or < 10% of daily energy intake) in humans; ii) accessible in English full text. Exclusion criteria were research protocols, thesis/dissertations, abstracts, letters to the editor, case reports, reviews, book chapters, guidelines, position papers, and unpublished works. Articles that did not meet the eligibility criteria were excluded via screening title, abstract, and full-text review.
Data Extraction
Based on the initial titles retrieved, duplicates were removed. After concluding the search, all records were compiled into the Endnote for Windows version X9, 2018 (Clarivate, Philadelphia, USA) software to delete all duplicates showing the same: a) title, authors and year of publication and b) title, authors, and journal title. The records remaining after the deletion of duplicates were exported to an Excel file for Windows version 16.75.2 (Microsoft, Washinton, EUA) organized based on essential information for screening, such as authors’ names, publication year, journal title, digital object identification (DOI), article title and abstract. Abstracts identified from the literature searches were screened for potential inclusion by two authors (F.C. and A.P.). The major characteristics extracted from the included studies were year of publication, study design and duration, sample size, BIA technology and device, bioelectrical measures and body composition estimations, and characteristics of the predictive equations used for estimating body composition.
Results
The seventy-two selected studies involved 3,825 participants (age ranging 2 – 78 y, body mass index ranging 15.3 – 50.9 kg/m2) of both genders. The procedure of the systematic search and the characteristics of the included studies are reported in Fig. 3 and Supplementary Table 1, respectively.
Technologies and Bioelectrical Raw Data
The layout of measurements for each of the four bioimpedance technologies is illustrated in Fig. 4, along with the corresponding number of studies that utilized them. Thirty-five studies employed a foot-to-hand technology [17, 57,58,59,60,61,62,63,63, 64•, 65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89], 34 utilized a standing position/segmental technology [90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124], and 3 did not report any information about the technology used [117, 125, 126]. No study employed the hand-to-hand or leg-to-leg technology. Five studies reported raw bioelectrical parameters of R, Xc, and PhA [64•, 73, 81, 82, 89], three reported only R and Xc [17, 65, 83], and 12 reported only PhA [61, 69, 71, 72, 80, 85, 86, 98, 99, 103, 105, 127].
Prediction of Body Mass Components
Figure 5 shows the predicted variables in the 72 included studies, indicating for each variable the number of studies in which it was considered and the percentage relative to the total predictions (N = 195).
Out of the 72 studies, 66 predicted FM [57,58,59,60,61, 63, 65,66,67,68,69, 71,72,73,74,75,76,77,78,79,80,81,82,83,84,85, 87, 88, 90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127], 41 predicted FFM [57,58,59, 63, 65,66,67, 69, 73,74,75,76,77,78, 80,81,82, 84, 85, 88, 90, 93, 98,99,100,101, 103, 105, 108,109,110, 114, 115, 117, 118, 120,121,122,123,124, 127], 24 predicted TBW [57, 58, 62, 69,70,71,72, 84, 89, 98,99,100,101, 103, 105, 108,109,110, 114,115,116, 118, 120, 121], 16 predicted ECW [58, 62, 69,70,71,72, 78, 89, 98, 99, 101, 103, 109, 118, 120, 122], 13 predicted ICW [58, 62, 69, 71, 78, 98, 99, 101, 103, 109, 118, 120, 121], 9 predicted SMM [58, 88, 96, 100, 107, 109, 110, 115, 121], 7 predicted visceral fat area [104, 107, 112, 115, 117, 121, 124], 6 predicted BCM [61, 67, 69, 85, 86, 89] and lean body mass [17, 79, 95, 106, 111, 126], 3 predicted muscle mass [115, 116, 122], 2 predicted lean mass [71, 94], and 1 predicted appendicular muscle mass [121],. Predictive equations have been reported in only three studies for the estimation of FM [76, 87], FFM [76], and LBM [17]. Ferraris et al. (2019) estimated FM using the following equation provided by Houtkooper et al. (1992): FM = body mass – FFM, where FFM = 0.58 * (R) + 0.24 * (weight) + 2.69. Pandurevic et al. (2023) estimated FM and FFM used the equation developed by Sun et al. (2003): FM = Weight – FFM, where FFM = − 10.68 + 0.65 × (stature2/R) + 0.26 × body mass + 0.02 × R. Vazquez and Janosky (1991) estimated LBM using eight different equations: 1) LBM = 0.6483 * (stature)2 + 0.1699* (body mass) + 5.091, by Segal et al. (1985); 2) LBM = 0.21411 * (stature) + 0.36273 * (stature2/R) + 0.1329 * (body mass) – 5.61911 * gender (where 1 is for female and 0 for male) – 8.98751, by Segal et al. (1985); 3) LBM = 0.821 * (stature2/R) + 4.917, by Lukaski et al. (1986); 4) LBM = 0.00085 * stature2—0.02375 * R + 0.3736 * (body mass)—0.1531 * (age) – 4.2921 * gender (where 1 is for female and 0 for male) + 17.7868, by Van Loan et al. (1987); 5) LBM = 0.00091186 * (stature)2—0.01466 * (R) + 0.29990 * (body mass)—0.07012 * (age) + 9.37938, by Segal et al. (1988); 6) LBM = 0.000985 * (stature)2—0.0387* (R) + 0.158 * (body mass) – 0.124 * (age) + 29.612, by Gray et al. (1989); 7) LBM = 0.698 * (stature2/R) + 3.5 * gender (where 0 is for female and 1 for male) + 9.4, by Deurenberg et al. (1989); 8) LBM = TBW/0.73, where TBW = 0.382 * (stature2/R) + 0.105 * (body mass) + 8.315, by Kushner et al. (1990).
Discussion
BIA enables the assessment of key body composition parameters relevant to the ketogenic diet [53, 136]. The main purpose of this scoping review was to select all studies that utilized BIA in ketogenic diet programs, assessing the validity of the procedures employed in analyzing body composition. A second aim was to suggest best practices for fully leveraging the potential of this technique. The results indicated that methods applied to BIA’s use are, generally, not well described, undermining the validity of the extracted data. This arises from the high number of studies that have not reported the prediction formulas used to estimate body composition, and the few compliant with this first point did not always used specific formulas suitable for the participants under examination. Moreover, few studies directly evaluated raw parameters, and none of them utilized classic or specific BIVA to monitor body fluids, their distribution, as well as FM.
Technologies and Bioelectrical Raw Data
BIA can currently be performed using four different types of devices, categorized into four distinct technologies. The studies included in this review have employed foot-to-hand and standing position, also known as segmental technology, while no study has utilized hand-to-hand and leg-to-leg technologies. However, three studies did not declare the technology used, nor the name of the device [117, 125, 126]. Reporting these types of information is relevant for comparison purposes, given the lack of agreement between the absolute data measured by different technologies [29•, 30]. Moreover, only 21 out of 72 studies reported raw data measured with BIA. Double-indirect methods for body composition analysis use raw measurements, such as R, Xc, and PhA, within regression models to estimate body mass components [3]. Hence, it is advisable to consistently include raw measurements, as they constitute the independent variables necessary for predictive models. Moreover, these raw data can be evaluated independently without necessarily being incorporated into predictive equations. For example, PhA has proven to be a reliable biomarker of fluid distribution between compartments, as well as being correlated with cellular integrity and density [64•]. By monitoring the phase angle, it's possible to qualitatively assess any compromise or improvement in cellular integrity, which plays a crucial role in determining muscle quality [64•]. Furthermore, through BIVA, it is possible to monitor PhA, representing the vector's distance from the x-axis, along with vector length which is a strong predictor of TBW [137] in classic BIVA [32••] or FM in specific BIVA [138]. This could be particularly useful in ketogenic diets to monitor glycogen depletion leading to intracellular water loss [49••]. Additionally, one of the goals during a ketogenic diet is to avoid excessive loss of SMM, a component primarily formed by fluids, especially intracellular ones [53]. In this regard, leftward vector displacements within the R-Xc graph identify increases in the ICW/ECW and then in SMM for both BIVA approaches [5]. Evaluating raw parameters without relying on predictive equations could help avoid dependence on estimation errors inherent in regression models. Such errors are even more pronounced when formulas unsuitable for the subject at hand are used, and unfortunately, there are still no formulas available for all populations to date [32••].
Prediction of Body Mass Components
A total of 196 estimates for various body mass components resulted from the 72 studies included in this review. However, only 4 studies reported the predictive formulas used, allowing for their discussion. Vasquez and and Janosky (1991) used eight different equations for estimating LBM. Considering that participants were adult females with obesity (Supp. Table 1) the use of the four formulas [130,131,132] appeared to be inappropriate since they were developed on normal weight females. One of the 8 formulas, instead, was developed on overweight subjects but mixed adults and elderly individuals [134]. It is worth noting that one equation estimated FFM from TBW, assuming that it can be composed of 73% fluids, a condition that is not always valid, especially in obese subjects who may exhibit overhydration [139]. In contrast, the other two formulas involved in the same study [17] may be valid since they were developed including obese subjects [15, 133]. Ferraris et al. [87] included participants aged 2 to 17 years and used the equation provided by Houtkooper et al. [128],which is developed on subjects similar age. Finally, Pandurevic et al. [76] considered adult female subjects and used the made by Sun et al. [129], which suffers from having been developed by mixing different populations (under 18, adults, and elderly people). In the remaining 68 studies, where the utilized predictive equations were not disclosed, evaluating the validity of the regression models concerning the characteristics of the involved participants becomes unfeasible. This is because the predictive equations, in addition to being specific to technologies, are also population-specific, and the use of different formulas can yield different results for the same parameter [17]. To date, a wide range of predictive equations is available in the literature, especially for the parameters that were most considered in the 72 studies, such as FM, FFM, fluids, SMM, BCM, and ALSM [140,141,142,143,144,145,146,147,148,149]. Conversely, to our knowledge, no equations are available for VFA and the algorithms used by some studies may be proprietary to the manufacturers. Moreover, the term muscle mass was incorrectly used interchangeably with FFM. Therefore, reporting the predictive equations used and adhering to correct terminology in accordance with the body composition basics (Fig. 1) not only allows future studies to replicate the procedures but also provides physicians and nutritionists with reference data in the clinical context.
Best Practices for Using BIA in Ketogenic Diet
In light of the above, the following best practices are discussed for improving the use of BIA in ketogenic diet studies:
-
Technology and device characteristics: these should always be reported because each device provides measurements that are not comparable with other technologies [29•].
-
Raw BIA data: reporting raw data allows for their use in new predictive equations or as reference data. Additionally, the assessment of PhA provides an indication of fluid distribution among compartments. Increases in PhA identify increases in the ICW/ECW ratio and vice versa [8, 64•].
-
Predictive equations: the equations should be reported among the methods and their selection should be justified. They are specific to technology and should be developed on subjects with similar characteristics to those being tested. Currently, there are various predictive equations for both the general population (under 18, adults, and elderly people) and athletes, developed for a wide range of body composition parameters and versus multicompartmental models using gold standard procedures [6, 150••].
-
Raw data analysis through classic or specific BIVA: when valid prediction equations are not available in the literature, the use of BIVA may avoid systematic estimation errors. Furthermore, reference tolerance ellipses to incorporate into R-Xc graphs are available for the general population and athletes [9, 32••].
The suggested best practices are schematized in Fig. 6.
Review Limitations and Future Perspectives
Some limitations about this review should be mentioned. Data on changes in body composition were not extracted. However, as hypothesized, the almost complete absence of information on the predictive equations used in the studies does not allow for certainty regarding the validity of the body composition outputs. Therefore, future systematic reviews will be necessary to analyze changes in body composition on a bioelectrical basis when enough studies have adopted best practices in its use during ketogenic diet. Another limitation is the failure to provide a list of predictive equations for each technology and each body composition variable. However, to date, such formulas are widespread in the literature, and no study has collected and cataloged them for different independent factors such as age, sex, health status, and level of physical activity. In this regard, a systematic review should be conducted to categorize all BIA-based prediction equations, facilitating their retrieval in the literature.
Conclusions
Until now, BIA has been employed in studies examining the efficacy of the ketogenic diet using methodologies with some limitations. The primary concern is the almost complete absence of descriptions (predictive equations) of the procedures used to estimate body composition. This lack of information compromises the validity of the obtained data and hinders future comparisons. Moreover, the majority of studies do not provide raw parameters, forfeiting the opportunity to analyze them qualitatively, thus avoiding potential intrinsic estimation errors in quantitative analysis of body mass components. We tried to suggest some best practices to enhance the use of BIA during ketogenic diet interventions, encompassing the description of BIA technologies and raw data, the selection of predictive equations, and the adoption of alternative analyses such as BIVA, which allows for qualitative monitoring of fluids and body fat.
Availability of Data and Materials
Not applicable.
Abbreviations
- BCM:
-
Body cell mass
- BMC:
-
Bone mineral content
- BIA:
-
Bioelectrical impedance analysis
- BIVA:
-
Bioelectrical impedance vector analysis
- CHO:
-
Carbohydrate
- ECW:
-
Extracellular water
- FM:
-
Fat mass
- FFM:
-
Fat-free mass
- ICE:
-
Intracellular water
- LBM:
-
Lean body mass
- LM:
-
Lean mass
- LSM:
-
Lean soft mass
- PhA:
-
Phase angle
- PRO:
-
Protein
- R:
-
Resistance
- SMM:
-
Skeletal muscle mass
- TBW:
-
Total body water
- VFA:
-
Visceral fat area
- Xc:
-
Reactance
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Wang ZM, Pierson RN, Heymsfield SB. The five-level model: A new approach to organizing body-composition research. Am J Clin Nutr. 1992;56(1):19–28.
Ellis KJ. Human body composition: in vivo methods. Physiol Rev United States. 2000;80:649–80.
Lohman TG, Milliken LA. ACSM’s Body composition assessment. Human Kinetics; 2020.
Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg P, Elia M, Gomez JM, et al. Bioelectrical impedance analysis–part I: review of principles and methods. Clin Nutr England. 2004;23:1226–43.
Campa F, Toselli S, Mazzilli M, Gobbo LA, Coratella G. Assessment of body composition in athletes: A narrative review of available methods with special reference to quantitative and qualitative bioimpedance analysis. Nutrients. 2021;13. Available from: https://www.mdpi.com/2072-6643/13/5/1620
Silva AM. Structural and functional body components in athletic health and performance phenotypes. Eur J Clin Nutr England. 2019;73:215–24.
Ramos IE, Coelho GM, Lanzillotti HS, Marini E, Koury JC. Fat-free mass using bioelectrical impedance analysis as an alternative to dual-energy x-ray absorptiometry in calculating energy availability in female adolescent athletes. Int J Sport Nutr Exerc Metab United States. 2022;32:350–8.
Campa F, Matias CN, Marini E, Heymsfield SB, Toselli S, Sardinha LB, et al. Identifying athlete body fluid changes during a competitive season with bioelectrical impedance vector analysis. Int J Sports Physiol Perform. 2020;15:361–7.
Marini E, Campa F, Buffa R, Stagi S, Matias CN, Toselli S, et al. Phase angle and bioelectrical impedance vector analysis in the evaluation of body composition in athletes. Clin Nutr England. 2020;39:447–54.
Nyboer J. Electrical impedance plethysmography; a physical and physiologic approach to peripheral vascular study. Circulation United States. 1950;2:811–21.
Thomasset MA. Bioelectric properties of tissue. Impedance measurement in clinical medicine. Significance of curves obtained. Lyon Med France. 1962;94:107–18.
Hoffer EC, Meador CK, Simpson DC. Correlation of whole-body impedance with total body water volume. J Appl Physiol United States. 1969;27:531–4.
Lukaski HC, Johnson PE, Bolonchuk WW, Lykken GI. Assessment of fat-free mass using bioelectrical impedance measurements of the human body. Am J Clin Nutr United States. 1985;41:810–7.
Kushner RF, Schoeller DA. Estimation of total body water by bioelectrical impedance analysis. Am J Clin Nutr United States. 1986;44:417–24.
Segal KR, Gutin B, Presta E, Wang J, Van Itallie TB. Estimation of human body composition by electrical impedance methods: a comparative study. J Appl Physiol United States. 1985;58:1565–71.
Lukaski HC, Bolonchuk WW. Estimation of body fluid volumes using tetrapolar bioelectrical impedance measurements. Aviat Space Environ Med United States. 1988;59:1163–9.
Vazquez JA, Janosky JE. Validity of bioelectrical-impedance analysis in measuring changes in lean body mass during weight reduction. Am J Clin Nutr United States. 1991;54:970–5.
Houtkooper LB, Lohman TG, Going SB, Howell WH. Why bioelectrical impedance analysis should be used for estimating adiposity. Am J Clin Nutr United States. 1996;64:436S-448S.
Heitmann BL. Evaluation of body fat estimated from body mass index, skinfolds and impedance. A comparative study. Eur J Clin Nutr England. 1990;44:831–7.
Svendsen OL, Haarbo J, Heitmann BL, Gotfredsen A, Christiansen C. Measurement of body fat in elderly subjects by dual-energy x-ray absorptiometry, bioelectrical impedance, and anthropometry. Am J Clin Nutr. 1991;53(5):1117–23.
Piccoli A, Rossi B, Pillon L, Bucciante G. A new method for monitoring body fluid variation by bioimpedance analysis: the RXc graph. Kidney Int United States. 1994;46:534–9.
Janssen I, Heymsfield SB, Baumgartner RN, Ross R. Estimation of skeletal muscle mass by bioelectrical impedance analysis. J Appl Physiol United States. 2000;89:465–71.
Murphy CH, Hector AJ, Phillips SM. Considerations for protein intake in managing weight loss in athletes. Eur J Sport Sci England. 2015;15:21–8.
Stokes T, Hector AJ, Morton RW, McGlory C, Phillips SM. Recent perspectives regarding the role of dietary protein for the promotion of muscle hypertrophy with resistance exercise training. Nutrients Switzerland. 2018;10(2):180.
Bartolomei S, Stout JR, Fukuda DH, Hoffman JR, Merni F. Block vs weekly undulating periodized resistance training programs in women. J strength Cond Res United States. 2015;29:2679–87.
Osco KM, Campa F, Coratella G, Correa BD, de Alencar Silva BS, Dos Santos VR, et al. Resistance but not elastic tubes training improves bioimpedance vector patterns and body composition in older women: A randomized trial. Exp Gerontol England. 2021;154:111526.
Oshima Y, Shiga T, Namba H, Kuno S. Estimation of whole-body skeletal muscle mass by bioelectrical impedance analysis in the standing position. Obes Res Clin Pract Netherlands. 2010;4:e1-82.
Campa F, Gobbo LA, Stagi S, Cyrino LT, Toselli S, Marini E, et al. Bioelectrical impedance analysis versus reference methods in the assessment of body composition in athletes. Eur J Appl Physiol. 2022. https://doi.org/10.1007/s00421-021-04879-y.
• Dellinger JR, Johnson BA, Benavides ML, Moore ML, Stratton MT, Harty PS, et al. Agreement of bioelectrical resistance, reactance, and phase angle values from supine and standing bioimpedance analyzers. Physiol Meas. 2021; Available from: http://iopscience.iop.org/article/10.1088/1361-6579/abe6fa. Highlights the lack of agreement among different BIA technologies, suggesting the need for specific reference values and predictive equations tailored to each bioelectrical device technology.
Stratton MT, Smith RW, Harty PS, Rodriguez C, Johnson BA, Dellinger JR, et al. Longitudinal agreement of four bioimpedance analyzers for detecting changes in raw bioimpedance during purposeful weight gain with resistance training. Eur J Clin Nutr England. 2021;75:1060–8.
Campa F, Matias CN, Moro T, Cerullo G, Casolo A, Teixeira FJ, et al. Methods over materials: the need for sport-specific equations to accurately predict fat mass using bioimpedance analysis or anthropometry. Nutrients. 2023;15. Available from: https://www.mdpi.com/2072-6643/15/2/278.
•• Campa F, Coratella G, Cerullo G, Stagi S, Paoli S, Marini S, et al. New bioelectrical impedance vector references and phase angle centile curves in 4,367 adults: The need for an urgent update after 30 years. Clin Nutr England. 2023;42:1749–58. Presents the latest update of the BIVA method with specific references for adults, including the new percentile curves for the phase angle.
Marini E, Sergi G, Succa V, Saragat B, Sarti S, Coin A, et al. ( Biva ) for assessing body composition in the elderly. J Nutr Health Aging. 2013;17:515–21.
Reljic D, Zarafat D, Jensen B, Herrmann HJ, Neurath MF, Konturek PC, et al. Phase angle and vector analysis from multifrequency segmental bioelectrical impedance analysis: new reference data for older adults. J Physiol Pharmacol an Off J Polish Physiol Soc. Poland; 2020;71.
Buffa R, Saragat B, Cabras S, Rinaldi AC, Marini E. Accuracy of specific BIVA for the assessment of body composition in the United States population. PLoS One United States. 2013;8:e58533.
Campa F, Matias CN, Nunes CL, Monteiro CP, Francisco R, Jesus F, et al. Specific bioelectrical impedance vector analysis identifies body fat reduction after a lifestyle intervention in former elite athletes. Biology Basel. 2021;10(6):524.
Paoli A, Rubini A, Volek JS, Grimaldi KA. Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur J Clin Nutr England. 2013;67:789–96.
Walters JK, Hoogwerf BJ, Reddy SS. The protein-sparing modified fast for obesity-related medical problems. Cleve Clin J Med United States. 1997;64:242–4.
Wilder RJ. The effects of ketonemia on the course of epilepsy. Mayo Clin Proc. 2:307–8. Available from: https://api.semanticscholar.org/CorpusID:76722699
Peterman MG. The ketogenic diet in the treatment of epilepsy: a preliminary report. J Nerv Ment Dis. 1926;64. Available from: https://journals.lww.com/jonmd/fulltext/1926/07000/the_ketogenic_diet_in_the_treatment_of_epilepsy__a.74.aspx
Caprio M, Infante M, Moriconi E, Armani A, Fabbri A, Mantovani G, et al. Very-low-calorie ketogenic diet (VLCKD) in the management of metabolic diseases: systematic review and consensus statement from the Italian Society of Endocrinology (SIE). J Endocrinol Invest Italy. 2019;42:1365–86.
Muscogiuri G, Barrea L, Laudisio D, Pugliese G, Salzano C, Savastano S, et al. The management of very low-calorie ketogenic diet in obesity outpatient clinic: a practical guide. J Transl Med. 2019;17:356. https://doi.org/10.1186/s12967-019-2104-z.
Paoli A. Ketogenic diet for obesity: friend or foe? Int J Environ Res Public Health. 2014;11:2092–107. Available from: https://www.mdpi.com/1660-4601/11/2/2092
Paoli A, Bosco G, Camporesi EM, Mangar D. Ketosis, ketogenic diet and food intake control: a complex relationship. Front Psychol [Internet]. 2015;6. Available from: https://www.frontiersin.org/journals/psychology/articles/10.3389/fpsyg.2015.00027
Dowis K, Banga S. The Potential health benefits of the ketogenic diet: a narrative review. Nutrients Switzerland. 2021;13(5):1654.
Trimboli P, Castellana M, Bellido D, Casanueva FF. Confusion in the nomenclature of ketogenic diets blurs evidence. Rev Endocr Metab Disord Germany. 2020;21(1):1–3.
Paoli A, Gorini S, Caprio M. The dark side of the spoon - glucose, ketones and COVID-19: a possible role for ketogenic diet? J Transl Med. 2020;18:441. https://doi.org/10.1186/s12967-020-02600-9.
Youm Y-H, Nguyen KY, Grant RW, Goldberg EL, Bodogai M, Kim D, et al. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome–mediated inflammatory disease. Nat Med. 2015;21:263–9. https://doi.org/10.1038/nm.3804.
•• Ashtary-Larky D, Bagheri R, Bavi H, Baker JS, Moro T, Mancin L, et al. Ketogenic diets, physical activity and body composition: a review. Br J Nutr England. 2022;127:1898–920. Summarizes the state of the art regarding the effects of the ketogenic diet on body composition in two different populations, athletes and general individuals.
Krebs HA. The regulation of the release of ketone bodies by the liver. Adv Enzyme Regul England. 1966;4:339–54.
French EK, Donihi AC, Korytkowski MT. Diabetic ketoacidosis and hyperosmolar hyperglycemic syndrome: review of acute decompensated diabetes in adult patients. BMJ. 2019;365:l1114. Available from: http://www.bmj.com/content/365/bmj.l1114.abstract
Paoli A, Tinsley GM, Mattson MP, De Vivo I, Dhawan R, Moro T. Common and divergent molecular mechanisms of fasting and ketogenic diets. Trends Endocrinol Metab. 2023; Available from: https://www.sciencedirect.com/science/article/pii/S104327602300214X
Ashtary-Larky D, Bagheri R, Asbaghi O, Tinsley GM, Kooti W, Abbasnezhad A, et al. Effects of resistance training combined with a ketogenic diet on body composition: a systematic review and meta-analysis. Crit Rev Food Sci Nutr United States. 2022;62:5717–32.
Coratella G, Campa F, Matias C, Toselli S, Koury J, Andreoli A, et al. Generalized bioelectric impedance-based equations underestimate body fluids in athletes. Scand J Med Sci Sport. 2021;31:2123–32.
Silva AM, Fields DA, Heymsfield SB, Sardinha LB. Body composition and power changes in elite judo athletes. Int J Sports Med Germany. 2010;31:737–41.
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann Intern Med United States. 2018;169:467–73.
Paoli A, Cenci L, Fancelli M, Parmagnani A, Fratter A, Cucchi A, et al. Ketogenic diet and phytoextracts. Comparison of the efficacy of mediterranean, zone and tisanoreica diet on some health risk factors. Agro Food Ind Hi Tech. 2010;21:24–9.
Bruci A, Tuccinardi D, Tozzi R, Balena A, Santucci S, Frontani R, et al. Very low-calorie ketogenic diet: a safe and effective tool for weight loss in patients with obesity and mild kidney failure. Nutrients Switzerland. 2020;12(2):333.
Buechert M, Lange T, Deibert P, Urbain P. In vivo fat quantification: monitoring effects of a 6-week non-energy-restricted ketogenic diet in healthy adults using MRI. ADP and BIA Nutrients Switzerland. 2020;12(1):244.
Lodi A, Zarantonello L, Bisiacchi PS, Cenci L, Paoli A. Ketonemia and glycemia affect appetite levels and executive functions in overweight females during two ketogenic diets. Obesity (Silver Spring) United States. 2020;28:1868–77.
Kämmerer U, Klement RJ, Joos FT, Sütterlin M, Reuss-borst M. low carb and ketogenic diets increase quality of life, physical performance, body composition, and metabolic health of women with breast cancer. Nutrients Switzerland. 2021;13(3):1029.
Paoli A, Mancin L, Caprio M, Monti E, Narici MV, Cenci L, et al. Effects of 30 days of ketogenic diet on body composition, muscle strength, muscle area, metabolism, and performance in semi-professional soccer players. J Int Soc Sports Nutr. 2021;18:62.
Paoli A, Cenci L, Pompei P, Sahin N, Bianco A, Neri M, et al. Effects of two months of very low carbohydrate ketogenic diet on body composition, muscle strength, muscle area, and blood parameters in competitive natural body builders. Nutrients Switzerland. 2021;13(2):374.
• Barrea L, Muscogiuri G, Aprano S, Vetrani C, de Alteriis G, Varcamonti L, et al. Phase angle as an easy diagnostic tool for the nutritionist in the evaluation of inflammatory changes during the active stage of a very low-calorie ketogenic diet. Int J Obes (Lond) England. 2022;46:1591–7. Clarifies the potential use of phase angle as an ecological tool for nutritionists interested in monitoring the change of inflammatory biomarkers during a ketogenic diet.
Barrea L, de Alteriis G, Muscogiuri G, Vetrani C, Verde L, Camajani E, et al. Impact of a very low-calorie ketogenic diet (VLCKD) on changes in handgrip strength in women with obesity. Nutrients Switzerland. 2022;14(19):4213.
Camajani E, Feraco A, Basciani S, Gnessi L, Barrea L, Armani A, et al. VLCKD in Combination with physical exercise preserves skeletal muscle mass in sarcopenic obesity after severe COVID-19 disease: a case report. Healthc Basel, Switzerland. 2022;10(3):573.
Camajani E, Feraco A, Proietti S, Basciani S, Barrea L, Armani A, et al. Very low calorie ketogenic diet combined with physical interval training for preserving muscle mass during weight loss in sarcopenic obesity: A pilot study. Front Nutr Switzerland. 2022;9:955024.
Paoli A, Cenci L, Grimaldi KA. Effect of ketogenic Mediterranean diet with phytoextracts and low carbohydrates/high-protein meals on weight, cardiovascular risk factors, body composition and diet compliance in Italian council employees. Nutr J England. 2011;10:112.
Di Rosa C, Lattanzi G, Spiezia C, Imperia E, Piccirilli S, Beato I, et al. Mediterranean diet versus very low-calorie ketogenic diet: effects of reaching 5% body weight loss on body composition in subjects with overweight and with obesity-a cohort study. Int J Environ Res Public Health Switzerland. 2022;19(20):1340.
Mancin L, Amatori S, Caprio M, Sattin E, Bertoldi L, Cenci L, et al. Effect of 30 days of ketogenic Mediterranean diet with phytoextracts on athletes’ gut microbiome composition. Front Nutr Switzerland. 2022;9:979651.
Valente M, Garbo R, Filippi F, Antonutti A, Ceccarini V, Tereshko Y, et al. Migraine prevention through ketogenic diet: more than body mass composition changes. J Clin Med Switzerland. 2022;11(17):4946.
Barrea L, Verde L, Santangeli P, Lucà S, Docimo A, Savastano S, et al. Very low-calorie ketogenic diet (VLCKD): an antihypertensive nutritional approach. J Transl Med England. 2023;21:128.
Barrea L, Verde L, Di Lorenzo C, Savastano S, Colao A, Muscogiuri G. Can the ketogenic diet improve our dreams? Effect of very low-calorie ketogenic diet (VLCKD) on sleep quality. J Transl Med England. 2023;21:479.
De Nucci S, Bonfiglio C, Donvito R, Di Chito M, Cerabino N, Rinaldi R, et al. Effects of an eight week very low-calorie ketogenic diet (VLCKD) on white blood cell and platelet counts in relation to metabolic dysfunction-associated steatotic liver disease (MASLD) in subjects with overweight and obesity. Nutrients Switzerland. 2023;15(20):4468.
Merlino G, Tereshko Y, Pez S, Dal Bello S, Pittino A, Di Lorenzo C, et al. Sleep of migraine patients is ameliorated by ketogenic diet, independently of pain control. Sleep Med Netherlands. 2023;107:196–201.
Pandurevic S, Mancini I, Mitselman D, Magagnoli M, Teglia R, Fazzeri R, et al. Efficacy of very low-calorie ketogenic diet with the Pronokal® method in obese women with polycystic ovary syndrome: a 16-week randomized controlled trial. Endocr Connect. England; 2023;12.
Rinaldi R, De Nucci S, Castellana F, Di Chito M, Giannuzzi V, Shahini E, et al. The effects of eight weeks’ very low-calorie ketogenic diet (VLCKD) on liver health in subjects affected by overweight and obesity. Nutrients Switzerland. 2023;15(4):825.
Tereshko Y, Dal Bello S, Di Lorenzo C, Pez S, Pittino A, Sartor R, et al. 2:1 ketogenic diet and low-glycemic-index diet for the treatment of chronic and episodic migraine: a single-center real-life retrospective study. J Headache Pain England. 2023;24:95.
Paoli A, Moro T, Bosco G, Bianco A, Grimaldi KA, Camporesi E, et al. Effects of n-3 polyunsaturated fatty acids (ω-3) supplementation on some cardiovascular risk factors with a ketogenic Mediterranean diet. Mar Drugs Switzerland. 2015;13:996–1009.
Tereshko Y, Dal Bello S, Di Lorenzo C, Pittino A, Filippi F, Belgrado E, et al. The effect of three different ketogenic diet protocols on migraine and fatigue in chronic and high-frequency episodic migraine: a pilot study. Nutrients Switzerland. 2023;15(20):4334.
Verde L, Barrea L, Docimo A, Savastano S, Colao A, Muscogiuri G. Chronotype as a predictor of weight loss and body composition improvements in women with overweight or obesity undergoing a very low-calorie ketogenic diet (VLCKD). Clin Nutr England. 2023;42:1106–14.
Verde L, Dalamaga M, Capó X, Annunziata G, Hassapidou M, Docimo A, et al. The antioxidant potential of the mediterranean diet as a predictor of weight loss after a very low-calorie ketogenic diet (VLCKD) in women with overweight and obesity. Antioxidants (Basel, Switzerland). 2022;12(1):18.
Vetrani C, Verde L, Savastano S, Colao A, Muscogiuri G, Barrea L. Supplementation with medium-chain fatty acids increases body weight loss during very low-calorie ketogenic diet: a retrospective analysis in a real-life setting. J Transl Med England. 2023;21:29.
Rubini A, Bosco G, Lodi A, Cenci L, Parmagnani A, Grimaldi K, et al. Effects of twenty days of the ketogenic diet on metabolic and respiratory parameters in healthy subjects. Lung United States. 2015;193:939–45.
Urbain P, Strom L, Morawski L, Wehrle A, Deibert P, Bertz H. Impact of a 6-week non-energy-restricted ketogenic diet on physical fitness, body composition and biochemical parameters in healthy adults. Nutr Metab (Lond) England. 2017;14:17.
Cannataro R, Perri M, Gallelli L, Caroleo MC, De Sarro G, Cione E. Ketogenic Diet Acts on Body Remodeling and MicroRNAs Expression Profile. MicroRNA (Shariqah, United Arab Emirates). United Arab Emirates. 2019;8:116–26.
Ferraris C, Guglielmetti M, Pasca L, De Giorgis V, Ferraro OE, Brambilla I, et al. Impact of the ketogenic diet on linear growth in children: a single-center retrospective analysis of 34 cases. Nutrients Switzerland. 2019;11(7):1442.
Perticone M, Maio R, Sciacqua A, Suraci E, Pinto A, Pujia R, et al. Ketogenic diet-induced weight loss is associated with an increase in vitamin d levels in obese adults. Molecules. Switzerland; 2019;24.
Romano L, Marchetti M, Gualtieri P, Di Renzo L, Belcastro M, De Santis G Lou, et al. Effects of a personalized VLCKD on body composition and resting energy expenditure in the reversal of diabetes to prevent complications. Nutrients. Switzerland; 2019;11.
Yancy WSJ, Olsen MK, Guyton JR, Bakst RP, Westman EC. A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: a randomized, controlled trial. Ann Intern Med United States. 2004;140:769–77.
Johnston CS, Tjonn SL, Swan PD, White A, Hutchins H, Sears B. Ketogenic low-carbohydrate diets have no metabolic advantage over nonketogenic low-carbohydrate diets. Am J Clin Nutr United States. 2006;83:1055–61.
Ministrini S, Calzini L, Nulli Migliola E, Ricci MA, Roscini AR, Siepi D, et al. Lysosomal acid lipase as a molecular target of the very low carbohydrate ketogenic diet in morbidly obese patients: the potential effects on liver steatosis and cardiovascular risk factors. J Clin Med. Switzerland; 2019;8.
Mohorko N, Černelič-Bizjak M, Poklar-Vatovec T, Grom G, Kenig S, Petelin A, et al. Weight loss, improved physical performance, cognitive function, eating behavior, and metabolic profile in a 12-week ketogenic diet in obese adults. Nutr Res United States. 2019;62:64–77.
Prins PJ, Noakes TD, Welton GL, Haley SJ, Esbenshade NJ, Atwell AD, et al. High rates of fat oxidation induced by a low-carbohydrate, high-fat diet, do not impair 5-km running performance in competitive recreational athletes. J Sports Sci Med Turkey. 2019;18:738–50.
Hadizadeh M, Gan W, Mohafez H, Sugajima Y. Impact of ketogenic diet on body composition during resistance training among untrained individuals. Open Sports Sci J. 2020;13:114–9.
Hagihara K, Kajimoto K, Osaga S, Nagai N, Shimosegawa E, Nakata H, et al. Promising effect of a new ketogenic diet regimen in patients with advanced cancer. Nutrients. Switzerland; 2020;12.
Khodabakhshi A, Akbari ME, Mirzaei HR, Mehrad-Majd H, Kalamian M, Davoodi SH. Feasibility, safety, and beneficial effects of mct-based ketogenic diet for breast cancer treatment: a randomized controlled trial study. Nutr Cancer United States. 2020;72:627–34.
Klement RJ, Champ CE, Kämmerer U, Koebrunner PS, Krage K, Schäfer G, et al. Impact of a ketogenic diet intervention during radiotherapy on body composition: III-final results of the KETOCOMP study for breast cancer patients. Breast Cancer Res England. 2020;22:94.
Klement RJ, Schäfer G, Sweeney RA. A ketogenic diet exerts beneficial effects on body composition of cancer patients during radiotherapy: An interim analysis of the KETOCOMP study. J Tradit Complement Med Netherlands. 2020;10:180–7.
Cincione RI, Losavio F, Ciolli F, Valenzano A, Cibelli G, Messina G, et al. Effects of mixed of a ketogenic diet in overweight and obese women with polycystic ovary syndrome. Int J Environ Res Public Health. Switzerland; 2021;18.
Foppiani A, De Amicis R, Lessa C, Leone A, Ravella S, Ciusani E, et al. Isocaloric ketogenic diet in adults with high-grade gliomas: a prospective metabolic study. Nutr Cancer United States. 2021;73:1004–14.
White AM, Johnston CS, Swan PD, Tjonn SL, Sears B. Blood ketones are directly related to fatigue and perceived effort during exercise in overweight adults adhering to low-carbohydrate diets for weight loss: a pilot study. J Am Diet Assoc United States. 2007;107:1792–6.
Klement RJ, Koebrunner PS, Meyer D, Kanzler S, Sweeney RA. Impact of a ketogenic diet intervention during radiotherapy on body composition: IV. Final results of the KETOCOMP study for rectal cancer patients. Clin Nutr England. 2021;40:4674–84.
Li J, Bai W-P, Jiang B, Bai L-R, Gu B, Yan S-X, et al. Ketogenic diet in women with polycystic ovary syndrome and liver dysfunction who are obese: A randomized, open-label, parallel-group, controlled pilot trial. J Obstet Gynaecol Res Australia. 2021;47:1145–52.
Schiavo L, De Stefano g, persico f, gargiulo s, di spirito f, griguolo g, et al. a randomized, controlled trial comparing the impact of a low-calorie ketogenic vs a standard low-calorie diet on fat-free mass in patients receiving an elipse™ Intragastric balloon treatment. Obes Surg United States. 2021;31:1514–23.
Vidić V, Ilić V, Toskić L, Janković N, Ugarković D. Effects of calorie restricted low carbohydrate high fat ketogenic vs. non-ketogenic diet on strength, body-composition, hormonal and lipid profile in trained middle-aged men. Clin Nutr England. 2021;40:1495–502.
Al Aamri KS, Alrawahi AH, Al Busaidi N, Al Githi MS, Al Jabri K, Al Balushi F, et al. The effect of low-carbohydrate ketogenic diet in the management of obesity compared with low caloric, low-fat diet. Clin Nutr ESPEN England. 2022;49:522–8.
Cincione RI, Messina A, Cibelli G, Messina G, Polito R, Losavio F, et al. Italian ketogenic mediterranean diet in overweight and obese patients with prediabetes or type 2 diabetes. Nutrients. Switzerland; 2022;14.
Klement RJ, Sweeney RA. Impact of a ketogenic diet intervention during radiotherapy on body composition: V. Final results of the KETOCOMP study for head and neck cancer patients. Strahlentherapie und Onkol Organ der Dtsch Rontgengesellschaft [et al]. Germany. 2022;198:981–93.
Sørlie V, De Soysa AK, Hyldmo ÅA, Retterstøl K, Martins C, Nymo S. Effect of a ketogenic diet on pain and quality of life in patients with lipedema: The LIPODIET pilot study. Obes Sci Pract United States. 2022;8:483–93.
Valinejad A, Khodaei K. Does exercise during a ketogenic diet effectively alter appetite sensation, appetite-regulating hormones, and body composition? Exp Biol Med (Maywood). England. 2022;247:1898–906.
Yang M, Bai W, Jiang B, Wang Z, Wang X, Sun Y, et al. Effects of a ketogenic diet in women with PCOS with different uric acid concentrations: a prospective cohort study. Reprod Biomed Online Netherlands. 2022;45:391–400.
Partsalaki I, Karvela A, Spiliotis BE. Metabolic impact of a ketogenic diet compared to a hypocaloric diet in obese children and adolescents. J Pediatr Endocrinol Metab Germany. 2012;25:697–704.
Cincione IR, Graziadio C, Marino F, Vetrani C, Losavio F, Savastano S, et al. Short-time effects of ketogenic diet or modestly hypocaloric Mediterranean diet on overweight and obese women with polycystic ovary syndrome. J Endocrinol Invest Italy. 2023;46:769–77.
Elsahoryi NA, Alkurd RA, Subih H, Musharbash R. Effect of low-calorie ketogenic vs low-carbohydrate diets on body composition and other biomarkers of overweight/obese women: An 8 weeks randomised controlled trial. Obes Med. 2023;41:100496. Available from: https://www.sciencedirect.com/science/article/pii/S2451847623000209
Kysel P, Haluzíková D, Pleyerová I, Řezníčková K, Laňková I, Lacinová Z, et al. Different effects of cyclical ketogenic vs. nutritionally balanced reduction diet on serum concentrations of myokines in healthy young males undergoing combined resistance/aerobic training. Nutrients. Switzerland; 2023;15.
Rondanelli M, Patelli Z, Gasparri C, Mansueto F, Ferraris C, Nichetti M. Very low calorie ketogenic diet and common rheumatic disorders. In: A case report. J. Clin. cases. United States; 2023. p. 1985–91.
Vinciguerra F, Longhitano S, Carrubba N, Piazza L, Di Stefano C, Arpi ML, et al. Efficacy, feasibility and tolerability of ketogenic diet for the treatment of poor response to bariatric surgery. J Endocrinol Invest Italy. 2023;46:1807–14.
Zajac A, Poprzecki S, Maszczyk A, Czuba M, Michalczyk M, Zydek G. The effects of a ketogenic diet on exercise metabolism and physical performance in off-road cyclists. Nutrients Switzerland. 2014;6:2493–508.
Klement RJ, Sweeney RA. Impact of a ketogenic diet intervention during radiotherapy on body composition: I. Initial clinical experience with six prospectively studied patients. BMC Res Notes. England; 2016;9:143.
Gomez-Arbelaez D, Bellido D, Castro AI, Ordoñez-Mayan L, Carreira J, Galban C, et al. Body composition changes after very-low-calorie ketogenic diet in obesity evaluated by 3 standardized methods. J Clin Endocrinol Metab United States. 2017;102:488–98.
Gomez-Arbelaez D, Crujeiras AB, Castro AI, Martinez-Olmos MA, Canton A, Ordoñez-Mayan L, et al. Resting metabolic rate of obese patients under very low calorie ketogenic diet. Nutr Metab (Lond) England. 2018;15:18.
D’Abbondanza M, Ministrini S, Pucci G, Nulli Migliola E, Martorelli E-E, Gandolfo V, et al. Very low-carbohydrate ketogenic diet for the treatment of severe obesity and associated non-alcoholic fatty liver disease: the role of sex differences. Nutrients. Switzerland; 2020;12.
Pilone V, Tramontano S, Renzulli M, Romano M, Cobellis L, Berselli T, et al. Metabolic effects, safety, and acceptability of very low-calorie ketogenic dietetic scheme on candidates for bariatric surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg United States. 2018;14:1013–9.
Nazarewicz RR, Ziolkowski W, Vaccaro PS, Ghafourifar P. Effect of short-term ketogenic diet on redox status of human blood. Rejuvenation Res United States. 2007;10:435–40.
Rhyu H-S, Cho S-Y. The effect of weight loss by ketogenic diet on the body composition, performance-related physical fitness factors and cytokines of Taekwondo athletes. J Exerc Rehabil Korea (South). 2014;10:326–31.
Buscemi S, Buscemi C, Corleo D, De Pergola G, Caldarella R, Meli F, et al. Obesity and circulating levels of vitamin d before and after weight loss induced by a very low-calorie ketogenic diet. Nutrients. Switzerland; 2021;13.
Houtkooper LB, Going SB, Lohman TG, Roche AF, Van Loan M. Bioelectrical impedance estimation of fat-free body mass in children and youth: a cross-validation study. J Appl Physiol United States. 1992;72:366–73.
Sun SS, Chumlea WC, Heymsfield SB, Lukaski HC, Schoeller D, Friedl K, et al. Development of bioelectrical impedance analysis prediction equations for body composition with the use of a multicomponent model for use in epidemiologic surveys. Am J Clin Nutr United States. 2003;77:331–40.
Lukaski HC, Bolonchuk WW, Hall CB, Siders WA. Validation of tetrapolar bioelectrical impedance method to assess human body composition. J Appl Physiol United States. 1986;60:1327–32.
Van Loan M, Mayclin P. Bioelectrical impedance analysis: is it a reliable estimator of lean body mass and total body water? Hum Biol. 1987;69:299–309.
Segal KR, Van Loan M, Fitzgerald PI, Hodgdon JA, Van Itallie TB. Lean body mass estimation by bioelectrical impedance analysis: a four-site cross-validation study. Am J Clin Nutr United States. 1988;47:7–14.
Gray DS, Bray GA, Gemayel N, Kaplan K. Effect of obesity on bioelectrical impedance. Am J Clin Nutr United States. 1989;50:255–60.
Deurenberg P, Weststrate JA, Hautvast JG. Changes in fat-free mass during weight loss measured by bioelectrical impedance and by densitometry. Am J Clin Nutr United States. 1989;49:33–6.
Kushner RF, Kunigk A, Alspaugh M, Andronis PT, Leitch CA, Schoeller DA. Validation of bioelectrical-impedance analysis as a measurement of change in body composition in obesity. Am J Clin Nutr United States. 1990;52:219–23.
Campa F, Toselli S, Mazzilli M, Gobbo LA, Coratella G. Assessment of body composition in athletes: a narrative review of available methods with special reference to quantitative and qualitative bioimpedance analysis. Nutrients. 2021;13:1620.
Lukaski H, Raymond-Pope CJ. New frontiers of body composition in sport. Germany: Int J Sports Med; 2021.
Saragat B, Buffa R, Mereu E, De Rui M, Coin A, Sergi G, et al. Specific bioelectrical impedance vector reference values for assessing body composition in the Italian elderly. Exp Gerontol England. 2014;50:52–6.
Piccoli A, Pillon L, Dumler F. Impedance vector distribution by sex, race, body mass index, and age in the United States: Standard reference intervals as bivariate Z scores. Nutrition United States. 2002;18:153–67.
Campa F, Bongiovanni T, Rossi A, Cerullo G, Casolo A, Martera G, et al. Athletic bioimpedance-based equations underestimate fat free mass components in male elite soccer players: development and validation of new soccer-specific predictive models. J Transl Med England. 2023;21:912.
Matias CN, Campa F, Santos DA, Lukaski H, Sardinha LB, Silva AM. Fat-free mass bioelectrical impedance analysis predictive equation for athletes using a 4-compartment model. Int J Sports Med Germany. 2021;42:27–32.
Matias CN, Santos DA, Júdice PB, Magalhães JP, Minderico CS, Fields DA, et al. Estimation of total body water and extracellular water with bioimpedance in athletes: A need for athlete-specific prediction models. Clin Nutr. 2016;35:468–74.
Sardinha LB, Correia IR, Magalhães JP, Júdice PB, Silva AM, Hetherington-Rauth M. Development and validation of BIA prediction equations of upper and lower limb lean soft tissue in athletes. Eur J Clin Nutr England. 2020;74:1646–52.
Sardinha LB, Rosa GB, Hetherington-Rauth M, Correia IR, Magalhães JP, Silva AM, et al. Development and validation of bioelectrical impedance prediction equations estimating regional lean soft tissue mass in middle-aged adults. Eur J Clin Nutr. 2023;77:202–11.
Kotler DP, Burastero S, Wang J, Pierson RN Jr. Prediction of body cell mass, fat-free mass, and total body water with bioelectrical impedance analysis: effects of race, sex, and disease. Am J Clin Nutr. 1996;64:489S-497S. https://doi.org/10.1093/ajcn/64.3.489S.
Janssen I, Heymsfield SB, Wang ZM, Ross R. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J Appl Physiol United States. 2000;89:81–8.
Toselli S, Campa F, Matias CN, de Alencar Silva BS, Dos Santos VR, Maietta Latessa P, et al. Predictive equation for assessing appendicular lean soft tissue mass using bioelectric impedance analysis in older adults: Effect of body fat distribution. Exp Gerontol England. 2021;150:111393.
Sergi G, De Rui M, Veronese N, Bolzetta F, Berton L, Carraro S, et al. Assessing appendicular skeletal muscle mass with bioelectrical impedance analysis in free-living Caucasian older adults. Clin Nutr. 2015;34:667–73. Available from: https://www.sciencedirect.com/science/article/pii/S0261561414001861
Cohn SH, Vaswani AN, Yasumura S, Yuen K, Ellis KJ. Assessment of cellular mass and lean body mass by noninvasive nuclear techniques. J Lab Clin Med United States. 1985;105:305–11.
•• Silva AM, Campa F, Stagi S, Gobbo LA, Buffa R, Toselli S, et al. The bioelectrical impedance analysis (BIA) international database: aims, scope, and call for data. Eur J Clin Nutr. England; 2023. An international database where users can access and potentially share bioelectrical data measured using various technologies, frequencies, and in diverse populations.
Acknowledgements
We thank the corresponding authors of the studies included in the review for providing the data where not directly available.
Funding
Open access funding provided by Università degli Studi di Padova within the CRUI-CARE Agreement. No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
AP and FC: Conceptualization and Methodology, Writing- Original draft preparation, Writing- Reviewing and Editing.
Corresponding author
Ethics declarations
Human and Animal Rights and Informed Consent
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for Publication
This manuscript does not contain any individual person’s data.
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Paoli, A., Campa, F. Problems and Opportunities in the use of Bioelectrical Impedance Analysis for Assessing Body Composition During Ketogenic Diets: A Scoping Review. Curr Obes Rep 13, 496–509 (2024). https://doi.org/10.1007/s13679-024-00573-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13679-024-00573-0