Abstract
Purpose of Review
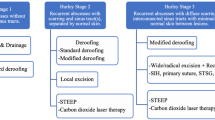
Hidradenitis suppurativa is a chronic inflammatory skin condition marked by significant structural changes such as nodules, abscesses, and subcutaneous tunnels, often accompanied by drainage and pain. Medical treatment alone usually does not suffice for optimal disease control. Therefore, surgical interventions, particularly for moderate-to-severe cases with tunnel formation, are a crucial adjunct to medical therapy. This paper aims to outline a practical approach to deroofing in hidradenitis suppurativa, covering the necessary materials, surgical approach, wound care, common complications, and frequent patient questions.
Recent Findings
Classically, wide excision of the entire anatomical area affected by hidradenitis suppurativa is performed in the operating room under general anesthesia. As a tissue-sparing alternative, deroofing—excision of tunnels—can be performed in-office under local anesthesia. This method allows for targeted application to either single lesions or entire anatomic regions, preserving the base of the cavity. Post-surgical wounds are typically left to heal by secondary intention.
Summary
Deroofing is a safe, in-office procedure for the management of hidradenitis suppurativa, with minimal rates of post-surgical complications. It is generally well-tolerated and associated with high patient satisfaction. When combined with proper medical treatment, deroofing can help patients achieve local disease control. Optimizing post-operative wound care is paramount to facilitate patient recovery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hidradenitis suppurativa (HS) is a complex disease characterized by both structural skin changes and persistent inflammation, necessitating a multifaceted approach to management [1]. With advancements in medical treatment, including antibiotics, hormonal therapy, and biologics, an integrated approach to HS management has developed. Combining surgical management with medical therapy allows clinicians to address both the structural manifestations of HS in addition to the underlying inflammation driving the disease.
Surgical interventions have emerged as a vital component in managing hidradenitis suppurativa (HS), a unique characteristic when contrasted with other chronic inflammatory skin conditions. Surgical treatment can address the distinct types of lesions that characterize HS, including inflammatory nodules, tunnels, abscesses, and scars [2, 3•]. Depending on the size and extent of the lesion, HS surgeries can be performed either in-office under local anesthesia or in an operating room. Surgical approaches vary in terms of technique, extent (lesional or regional), and intent (partial treatment or definitive) [3•]. Simple approaches like incision and drainage can provide immediate relief by draining painful abscesses while deroofing and excision can offer definitive treatment options.
Deroofing is a surgical approach that targets chronic HS changes in the skin and aims to replace areas that are chronically draining or painful with a quiescent scar. The consensus definition for deroofing is “removal of all or the large majority of skin overlying a hidradenitis cavity, with the base left intact following debridement of the base” [4]. Here, the term “cavity” is broad, including lesions such as abscesses and tunnels. The procedure can be conducted using various methods—scalpel, laser, or electrosurgical instruments—including skin-tissue sparing excision with electrosurgical peeling (STEEP) instrumentation [3•, 4]. Similarly, debridement can be achieved via manual friction (such as gauze rubbed on the base), curettage, grattage, laser vaporization, or other techniques [3•, 4]. The intent of deroofing may vary, as complete removal of the entire lesion or affected area may not always be feasible or desirable to the patient in real-world practice [3•, 4]. This means deroofing can target single lesions or entire anatomic regions and be partial (leaving adjacent affected skin behind) or complete (leaving no known cavity or portion of a cavity behind) [5]. Importantly, deroofing leaves the base of the cavity intact [5]. This paper aims to outline a practical approach to deroofing in hidradenitis suppurativa, covering the necessary materials, surgical approach, common complications, and frequent patient queries.
Setting Up for Deroofing
Required Instruments
A comprehensive surgical kit for deroofing could include the following instruments, which are summarized in Table 1:
Double-ended surgical probe
The primary tool used in this procedure is a blunt, straight, and malleable surgical probe to identify the extent and tracking of hidradenitis cavities like tunnels. An example is the McKesson Miltex #10–10-NS (#537373), measuring around 6 to 7 inches in length. If this specific probe is unavailable, acceptable alternatives for smaller/shorter cavities include a blunt hyfrecator tip or a pair of blunt dissecting or tissue supercut scissors with a curved design. These alternatives are typically suitable for small areas of deroofing and may not be effective for large tunnels or extensive lesions. The crucial feature of these instruments is their blunt end, designed to minimize tissue damage during exploration.
Toothed forceps
Due to the fibrotic nature of the affected skin, toothed forceps are necessary. They provide a reliable grip allowing thickened skin to be effectively manipulated.
Sharp scissors
Cutting through fibrotic tissue requires sharp tissue scissors. Super Cut scissors are an ideal choice because of the unique blade design that combines a razor edge and a scissor edge. Other alternatives include Curved Stevens Tenotomy scissors or Curved Metzenbaum scissors, both of which are well-suited for the precision required for deroofing.
Scalpel
A scalpel, with either a #15 or #10 blade, is needed to incise down to the depth of the tunnel. This ensures a thorough exploration and deroofing of the affected area.
Curette
A curette is needed to debride the base of the lesion. This helps with maximizing the removal of diseased tissue and therefore minimizing recurrence.
Optional Set-up
Tumescent lidocaine anesthesia (TLA)
For larger cases, tumescent lidocaine may be used to provide local anesthesia. This reduces patient discomfort during the procedure and enables a more thorough exploration and treatment of extensive disease. TLA has several benefits, including a longer-lasting effect than regular subcutaneous lidocaine anesthesia and has been associated with a reduced risk of operative bleeding [6]. Additionally, when larger areas are being surgically treated, the use of TLA allows for optimal pain control with lower doses of lidocaine, avoiding the risk of dose-dependent lidocaine systemic toxicity. TLA is typically prepared as follows: 100 mL of 1% lidocaine with epinephrine 1:100,000 and 10 mL of sodium bicarbonate are added to a 1000 mL bag of 0.9% normal saline (NSS) [7]. This results in 1110 mL of the TLA solution with a 0.09% concentration of lidocaine. These volumes can be adapted to allow the use of available agents (for example, 50 mL of 2% lidocaine with epinephrine). Importantly, TLA solutions should be prepared on the day of surgery and marked with safety labels warning against their inadvertent use as intravenous (IV) NSS.
Surgical Approach for Deroofing
-
1.
Start the procedure by administering a local anesthetic around the affected region. This typically covers an area of 2–5 cm (about 1.97 in) beyond the visually identified lesions or can extend to encompass any neighboring dimples or areas of skin alteration.
-
2.
Identify the typical appearance of the tract’s outlet, which is often characterized by a distinct formation on the skin’s surface such as erythema surrounding friable tissue or an indentation in the skin, as shown in Fig. 1.
-
3.
As shown in Fig. 2, the following steps are then performed. First, probe the affected area circumferentially—a complete 360°. This exploration allows for a thorough understanding of the surrounding cavities in terms of number, dimensions, and depth.
-
4.
Next, insert the probe into a tunnel then make an incision along and down to the level of the probe. This results in the creation of two skin flaps, representing the roof of the tunnel.
-
5.
The next step involves removing the previously formed skin flaps. Using the flat angle of scissors, gently slope the skin at the edges. This action results in a smoother topography of the skin, encouraging granulation by minimizing step-off depth from the surrounding skin to the healing wound base.
-
6.
Perform a debridement to eliminate the base of the lesion. This can be achieved with a curette. An essential component of this surgical intervention involves the extraction of the lining within the tract. This material often appears gelatinous and serosanguinous. It is crucial to remove these elements to ensure a thorough debridement of the tract [8]. This action is performed down to the level of the dermis and may involve some superficial fat.
-
7.
Finally, probe the treated area once more to ensure there are no undetected connecting cavities. The thoroughness of this last step significantly impacts the long-term success of the surgical intervention.
Pearls for Deroofing
Patient Communication
Maintaining open communication with patients is crucial when performing surgical deroofing. It is important to establish the limits of what can be addressed in a single visit and inform patients about what they can expect during and after the procedure. In-depth discussions about wound healing and pain management should take place, ensuring that patients are fully informed about the post-procedure expectations, including potential discomfort, wound care, and anticipated recovery time. To help patients develop a better understanding of the healing process and set expectations for treatment of larger areas, starting with a smaller procedure such as a complete lesional deroofing during the first deroofing session can be beneficial.
Detection and Exploration of Lesions
During the procedure, it is essential to detect and explore all lesions thoroughly. Applying gentle pressure and manipulating the lesion by pushing and squeezing can reveal hidden tracts that may not be easily detectable by probing alone. Initial lidocaine injections can be used to outline the extent of the lesions and aid in the detection of hidden tunnels by watching where the lidocaine drains when injected into the cavity area. It is important to delineate the entirety of the affected area while maintaining gentle probing to avoid the creation of false tunnels due to excessive probing force. Special attention should be given to lesions located in the axilla and inguinal folds due to their potential complexity and depth. Recognize that some tunnels may be deeper than what can be safely explored in an outpatient setting. In such cases, ultrasound guidance may be required [9].
Surgical Technique
During debridement, it is advisable to achieve tapered or sloped wound edges to avoid a step-off from the surrounding skin. The angle of your scissors can assist in achieving this. While rough debridement is sufficient, more challenging areas that require careful and precise tissue removal may necessitate the use of a hyfrecator. Additionally, the use of a hyfrecator may be beneficial for creating a smoother wound edge and preventing premature re-epithelialization, which allows for optimal healing of the wound bed.
Post-Deroofing Wound Care
Wound care post-deroofing surgery can be challenging, mainly due to the difficulty of placing and maintaining bandages in intertriginous areas. Nonetheless, optimal wound care is of paramount importance to prevent the complications that may arise from the disruption of the body’s protective barrier. Additionally, proper wound care ensures faster and better healing. For instance, maintaining a moist environment around a healing wound has been shown to decrease inflammation [10], and therefore, promote faster re-epithelialization. The use of occlusive dressings can facilitate the maintenance of such an environment when dealing with post-surgical wounds.
Specifically, for patients dealing with wound care after deroofing surgery, we recommend dressing the area with petrolatum and a non-stick gauze, such as Tefla™. The latter are inexpensive and readily available. The concomitant use of petrolatum might decrease the risk of dressing adherence to the wound bed upon drying of the wound’s natural exudate. As woven gauze dressings have a low absorptive capacity, the use of a secondary dressing, such as a foam dressing, might be recommended to decrease exudate leakage and subsequent discomfort, clothes staining, or smell. Tape or film dressings can be used to hold both the primary and secondary dressings in place. Film dressings offer the advantage of acting as a protective barrier against water, allowing the patient to take a shower.
The use of combination dressings, such as bordered foam dressings is a reasonable alternative. Typically, bordered foam dressings harbor a silicone-based adhesive border, which tends to be soft and therefore associated with minimal pain and granulation bed disruption upon removal. Their use in post-operative wounds following orthopedic surgery has been associated with decreased peri-wound blistering [11]. Data on their use following HS surgery remains scarce.
Generally, data on optimal post-deroofing wound care remains limited. In the absence of comprehensive HS-specific guidelines for post-surgical wound care, a Delphi study generated expert consensus and recommendations concerning appropriate wound care guidelines for routine and post-surgical HS wounds [12•].
Common Complications of Deroofing
While surgical deroofing is a highly effective treatment, it is not without potential complications. Most notably, patients can experience bleeding and pain post-operatively [13]. Pain is generally tolerable [14] and patient-reported outcomes reveal that it may be less intense than the pain associated with disease flare [14]. The use of over-the-counter analgesics is generally sufficient in our clinical experience.
Although infections following this surgical procedure are infrequent [13, 15], they can pose significant challenges when they do occur. Therefore, strict adherence to post-operative care instructions and close monitoring of the surgical site is essential.
Dressing the wound can sometimes present difficulties, due to the variable nature of the lesion sites and the need for careful bandaging to promote proper healing and prevent further complications.
An additional complication, though typically manageable, is the development of hypergranulation. This condition, characterized by an overgrowth of granulation tissue at the site of a healing wound, can be effectively treated with topical applications of silver nitrate. Furthermore, decreasing occlusion—or the coverage of the wound—can also contribute to the resolution of hypergranulation. The use of topical corticosteroids has shown efficacy for the resolution of hypergranulation in post-surgical wounds [16, 17] and burn wounds [18]. Studies reporting on the efficacy of steroids for the treatments of hypergranulation have however used agents at varying potencies and for different durations. Therefore, there are currently no specific recommendations on the choice of the agent and treatment duration. We suggest starting with a lower potency agent before escalating to stronger therapy.
Finally, an important post-operative complication is disease recurrence in surgically treated areas. A meta-analysis estimated this risk at 27.0% (95% CI, 23.0–31.0%) following an analysis of 22 articles reporting on surgical outcomes for the treatment of HS [19]. However, due to the poor-quality evidence and potential improper reporting of the results as per the authors, the reported rate is not generalizable. It remains, nonetheless, important to counsel patients on the possibility of disease recurrence following deroofing. In addition, it is generally thought that disease recurrence is typically milder, affecting smaller areas and easily dealt with via additional deroofing. Optimization of medical management in the pre- and post-operative periods is crucial to mitigating the risk of recurrence.
Frequently Asked Patient Questions (Table 2)
What is the Level of Discomfort Associated With the Surgical Procedure?
Typically, mild to moderate pain can be expected within the first 24 h following the procedure [13]. This discomfort is typically comparable to that experienced during an HS flare [14]. However, pain gradually subsides, and by 6 weeks post-operation, patients usually report no residual pain [13]. Additionally, the majority of patients are typically satisfied and recommend the procedure [23].
Is Medication Discontinuation Required Prior to Surgery?
Generally, the answer is no. Stopping medication can often lead to poorly controlled disease, which may pose a higher risk of surgical complications than the potential side effects of the medications themselves [20, 21]. In the phase 4 SHARPS trial, the efficacy and safety of adalimumab in combination with wide-excision surgery followed by secondary intention healing was investigated [20]. Treatment-emergent outcomes, including post-operative wound infection, complication, and hemorrhage, were similar in both patient groups receiving adalimumab or placebo [20]. However, those on adalimumab therapy achieved significantly more HS clinical response across all body regions [20]. This data supported the efficacy of adalimumab in conjunction with surgery and refuted the need to discontinue this therapy perioperatively [20]. These findings are applicable to patients undergoing deroofing surgery as wounds are generally smaller and more superficial than those secondary to wide excision.
What is the Expected Healing Time for this Surgical Procedure?
The healing time depends on the size of the area treated. Larger areas can take between 3 to 8 weeks (about 2 months) to heal fully [13]. As shown in Fig. 3, granulation and contraction of the wound size can occur quickly with excellent cosmesis without the use of sutures.
What is the Anticipated Extent of Scarring?
Scars from the surgical deroofing procedure usually shrink during the healing process. They tend to be around 25 to 30% of the initial size of the treated area [8, 13]. Most patients are typically satisfied with the appearance of their healed wounds and scars [22].
Is a Complete Cure Expected After the Procedure?
Managing expectations regarding the goal of the treatment can be difficult. Complete lesional or regional surgical deroofing aims to replace active HS with a scar. While this significantly reduces the burden of the disease, recurrences may occur. However, these are typically smaller and can be managed effectively with additional deroofing procedures. While not a complete cure, surgical deroofing reduces the disease’s impact and can improve quality of life [22, 23].
Conclusions
Surgical deroofing has emerged as a critical tool in managing hidradenitis suppurativa, resulting in high patient satisfaction due to its effectiveness in reducing recurrence and disease-related morbidity. It forms part of a broader, multidisciplinary treatment approach which addresses the physical manifestations of HS and its underlying pathophysiological mechanisms.
A comprehensive management strategy should incorporate medical therapies in addition to surgical deroofing. This procedure is particularly relevant for cases where HS proves persistent or recurrent despite optimizing medical therapy. In case of disease recurrence, smaller local procedures may be used as part of the ongoing management plan.
In conclusion, the surgical deroofing technique significantly broadens the spectrum of available treatment options and can be used to deliver more personalized and effective care for patients with HS.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82(5):1045–58.
Frew JW, et al. Global harmonization of morphological definitions in hidradenitis suppurativa for a proposed glossary. JAMA Dermatol. 2021;157(4):449–55.
• Bermudez NM, et al. Hidradenitis suppurativa: surgery, lasers, and emerging techniques. Surg Technol Intern. 2023;42:sti42/1659-sti42/1659. This paper on hidradenitis suppurativa (HS) is significant for its comprehensive exploration of the disease, emphasizing its prevalence, impact on quality of life, and the current management paradigms. The document meticulously details various surgical interventions, ranging from established techniques like intralesional corticosteroids, incision and drainage, punch debridement, and deroofing, to emerging methods such as Skin-Tissue-Sparing Excision with Electrosurgical Peeling (STEEP), lasers, cryoinsufflation, and antibiofilm therapy. Comparative analyses and summary tables provide valuable insights for clinicians, while the paper stresses the importance of integrating surgical approaches with medical management for optimal outcomes. Additionally, the document underscores the need for further research, especially randomized controlled trials, to enhance our understanding of HS and refine treatment strategies. Overall, this paper serves as a comprehensive resource for healthcare professionals involved in addressing the complexities of hidradenitis suppurativa.
Saunte DML, Jemec GBE. Hidradenitis suppurativa: advances in diagnosis and treatment. JAMA. 2017;318(20):2019–32.
Bui H, et al. Surgical procedural definitions for hidradenitis suppurativa developed by expert Delphi consensus. JAMA Dermatol. 2023;159(4):441–7.
Roerden A, et al. Benefits, safety and side effects of tumescent local anesthesia in dermatologic surgery in infants. JDDG: Journal der Deutschen Dermatologischen Gesellschaft. 2021;19(3):352–357.
Klein JA. Anesthetic formulation of tumescent solutions. Dermatol Clin. 1999;17(4):751–9.
Danby FW. Commentary: unroofing for hidradenitis suppurativa, why and how. J Am Acad Dermatol. 2010;63(3):481 e1–e3.
Wortsman X, Jemec G. A 3D ultrasound study of sinus tract formation in hidradenitis suppurativa. Dermatol Online J. 2013;19(6).
Junker JP, et al. Clinical impact upon wound healing and inflammation in moist, wet, and dry environments. Adv Wound Care. 2013;2(7):348–56.
Bredow J, et al. Clinical trial to evaluate the performance of a flexible self-adherent absorbent dressing coated with a soft silicone layer compared to a standard wound dressing after orthopedic or spinal surgery: study protocol for a randomized controlled trial. Trials. 2015;16(1):1–5.
• Chopra D, et al. Wound care for patients with hidradenitis suppurativa: recommendations of an international panel of experts. J Am Acad Dermatol. 2023;89(6):1289–1292. This paper addresses the challenges of wound care in hidradenitis suppurativa (HS), emphasizing the lack of specific guidelines. The study, conducted through an e-Delphi approach with a multidisciplinary expert panel, provides comprehensive recommendations for routine and post-surgical wound care. The consensus statements cover various aspects, including cleanser selection, dressing types, and frequency of changes. The significance lies in the absence of existing HS-specific guidelines, making these expert recommendations valuable for clinicians and patients. The study underscores the substantial burden of wound care in HS and highlights the need for further research to optimize strategies and enhance patient outcomes in this condition.
Dahmen RA, et al. Deroofing followed by thorough sinus tract excision: a modified surgical approach for hidradenitis suppurativa. JDDG: Journal der Deutschen Dermatologischen Gesellschaft. 2019;17(7):698–702.
Ravi S, et al. Patient impressions and outcomes after clinic-based hidradenitis suppurativa surgery. JAMA Dermatol. 2022;158(2):132–41.
van Hattem S, et al. Surgical treatment of sinuses by deroofing in hidradenitis suppurativa. Dermatol Surg. 2012;38(3):494–7.
McShane DB, Bellet JS. Treatment of hypergranulation tissue with high potency topical corticosteroids in children. Pediatr Dermatol. 2012;29(5):675–8.
Saikaly SK, Saikaly LE, Ramos-Caro FA. Treatment of postoperative hypergranulation tissue with topical corticosteroids: a case report and review of the literature. Dermatol Ther. 2021;34(2): e14836.
Jaeger M, et al. Treatment of hypergranulation tissue in burn wounds with topical steroid dressings: a case series. Intern Med Case Rep J. 2016;241–245.
Mehdizadeh A, et al. Recurrence of hidradenitis suppurativa after surgical management: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;73(5):S70–7.
Bechara FG, et al. Efficacy and safety of adalimumab in conjunction with surgery in moderate to severe hidradenitis suppurativa: the SHARPS randomized clinical trial. JAMA Surg. 2021;156(11):1001–9.
Alikhan A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: Part I: Diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81(1):76–90.
Kohorst JJ, et al. Patient satisfaction and quality of life following surgery for hidradenitis suppurativa. Dermatol Surg. 2017;43(1):125–33.
van der Zee HH, Prens EP, Boer J. Deroofing: a tissue-saving surgical technique for the treatment of mild to moderate hidradenitis suppurativa lesions. J Am Acad Dermatol. 2010;63(3):475–80.
Funding
There were no sources of financial assistance to conduct the study/analysis described in the manuscript and/or used to assist with the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
F.D. M.Y. and D.S. wrote the main manuscript text. F.D. and D.S. prepaired figures 1-4. All authors reviewed the mansucript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dzuali, F., Yaghi, M. & Saylor, D.K. Deroofing: A Practical Guide for the Dermatologist. Curr Derm Rep 13, 1–7 (2024). https://doi.org/10.1007/s13671-024-00419-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13671-024-00419-x