Abstract
Purpose of Review
An unexpected and sudden outbreak of a novel infection known as a coronavirus (COVID-19) has imposed important problems to global well-being and economy. Based upon current researches, this virus is spreading from one human to another through respiratory droplets, i.e. cough and sneeze. Till now, there has not been any specific treatment found for this virus. Hence, there is a critical need to discover alternative techniques to cope with the current scenario.
Recent Findings
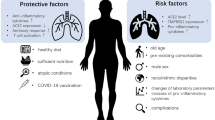
This review conducted an online search for prevention of coronavirus infection with the help of nutritional interventions. It has been observed that the effect of the virus is mostly on the individual with low immunity, individual affected with diseases like diabetes, and individual using any immune-suppressed drug or having past history of major surgeries or severe medical conditions.
Summary
Therefore, consuming foods which boost immunity helps in preventing respiratory-related disorder or suppressing diseases-related problems, which could be helpful in controlling the spread of this virus. In conclusion, it has been suggested that before the beginning of generalised treatments and interventions in each infected patient, nutritional status should be evaluated, as it can help in creating a specific nutrition intervention for the infected individual.
Similar content being viewed by others
Background
Sudden outbreak of a novel and alarming contagious primary atypical (viral) pneumonia was first observed in Wuhan, China, in December 2019 and spread globally resulting in a pandemic situation. Screening of this virus led to the identification of zoonotic coronavirus, similar to severe acute respiratory syndrome (SARS) and middle east respiratory syndrome (MERS) coronavirus. Researchers coined this disease as coronavirus disease 2019 (COVID-19) caused by the novel virus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. This disease mainly affects respiratory organs causing pneumonia and multi-organ failure, though it starts with common cold symptoms with shortness of breath [2•]. According to the data chart, as of 23rd March 2020, the rate of deaths per number of diagnosed cases found to be 4.4%, although according to age group and other health problems, it ranges from 0.2–15%. The World Health Organization (WHO) declared the 2019–2020 outbreak of the virus as a Public Health Emergency of International Concern (PHEIC) on 30th January 2020 and a pandemic on 11th March 2020. Based upon current researches, this virus is spreading from one human to another through respiratory droplets, i.e. cough and sneeze.
Coronavirus comes under the subfamily Orthocoronavirinae, in the family Coronaviridae [3]. It is a common cold virus and considered to be the weakest in this family. Coronavirus is an enveloped positive-sense ribonucleic acid (RNA) virus, characterised by the club-like spikes projecting from the surface. Genetic sequencing of COVID-19 is a little bit difficult, as the virus show cytopathic effect. According to research, COVID-19 is closely related to the BatCoV RaTG13 sequence, although RaTG13 does not show the exact variant of the novel virus. They also suggested that this novel coronavirus is not motley in nature and that almost half of its genome of a distinct lineage is found within beta coronavirus [4].
COVID-19 is mostly affecting the lungs because it accesses host cell through angiotensin-converting enzyme 2 (ACE2) abundant in type II alveolar cells of lungs. This virus seems to have a particular structure that contains a special surface glycoprotein called spike (peplomer) to connect to ACE2 and to enter the host cell. Diagnosis of this virus is possible by using a standard method reverse transcription-polymerase chain reaction (RT-PCR) from a nasopharyngeal swab. But still, now, there is no definite vaccine, treatment, or, cure. It has been observed that the effect of the virus is mostly on the individual with low immunity, individual affected with diseases like diabetes, individual with cardiovascular disease (CVD), and individual using any immune-suppressed drug or having past history of major surgeries or severe medical conditions. Till now, there has not been any specific treatment found for this virus. Hence, there is a critical need to discover alternative techniques to cope with the current scenario. This review conducted an online search for prevention of coronavirus infection with the help of nutritional interventions. It is invalid to say that consumption of healthy foods or following the traditional methods will cure the effects of COVID-19. But, the inclusion of some nutrition interventions can surely act as immune-boosters, which can help us to avoid this type of fatal diseases and their consequences, as we know, prevention is always better than cure.
Role of Nutrition
Vitamin A
Vitamin A comes in the category of fat-soluble vitamins. It is the first vitamin which was recognised as fat-soluble. β-carotene is known as a plant-derived precursor of vitamin A. This vitamin plays a very crucial role in the body’s immune system, as many of the body’s defences against infection depend on its adequate intake. Due to this, the vitamin is known as “anti-infective”. Various facets of innate immunity along with barrier function are controlled by vitamin A and its metabolites. Vitamin A is present in the body in three active forms: retinal, retinol, and retinoic acid. Retinoic acid acts as a ligand and activates nuclear retinoic acid receptor (RAR), while unknown metabolites may activate the retinoid X receptor (RXR). Hence, retinoic acid (all-trans and 9-cis) plays vital roles in the regulation of the differentiation, maturation, and function of the innate immune system and cells. Innate immune cells consist of neutrophils and macrophages. Retinoic acid encourages an instantaneous response to pathogen incursion with the help of phagocytosis and activation of natural killer (NK) T cells, which link immune-regulatory functions by cytotoxic activity [5]. Retinoic acid can also reform the differentiation of dendritic cell precursors, which are specialised protectors of the immune system managing innate and adaptive immune responses [6]. Many researchers reported that incorporation of vitamin A in the diet helps in protecting the body against various life-threatening diseases and infections like human immunodeficiency virus (HIV), malaria, coronary heart diseases, and various lung-related disorders [7, 8]. Therefore, low vitamin A status can be directly correlated with the hindered function of immune cells. Hence, vitamin A deficiency is linked with altered barrier function, impaired immune responses, and increased sensitivity to a variety of infections. According to a study, mice having low vitamin A status were more prone to the entry of pathogens due to the breakdown of the gut barrier and altered mucus secretion. Furthermore, a kind of coronavirus infection called infectious bronchitis virus (IBV) has a more pronounced effect in chickens who were fed a diet which was slightly lacking in vitamin A as compared with the chickens who were fed a diet sufficient in vitamin A [9]. Thus, in consideration of the recent time, where no successful treatment is there for COVID-19, vitamin A may be one of the possible choices. Moreover, a recent study reported that individuals having vitamin A deficiency display histopathological alterations causing an increased risk of lung dysfunction and respiratory disease [10]. This is especially appropriate considering the effects that COVID-19 have on respiration and lung function. Dietary sources of vitamin A are given in Table 1.
Vitamin C
It is a water-soluble vitamin. It is also known as ascorbic acid or “no-scurvy acid”. It plays a very crucial role in collagen formation in connective tissues. Vitamin C is also known as a classical antioxidant, directly suppressing free radicals in the aqueous layer while oxidising itself to dehydroascorbic acid. Additionally, an increased dietary ascorbic acid intake has been directly proportional to lesser concentrations of C-reactive proteins, which are responsible for inflammation in the body [11]. However, vitamin C also plays the role of a cofactor for a variety of biosynthetic and gene regulatory enzymes, proposing immune-modulating effects. Various in vitro and pre-clinical trials have shown the barrier-enhancing properties of vitamin C. Moreover, vitamin C can also adjust gene expression in dermal fibroblasts, improving their multiplication and migration, which play predominant roles for tissue remodelling and are significant in wound healing [12, 13]. Vitamin C is also known to stimulate neutrophil apoptosis to protect the host’s tissue from vigorous damage and further supports in macrophage removal. Furthermore, ascorbic acid plays a vital role in the proliferation and maturation of T cells, which are lymphocyte immune cells that protect the body from various pathogens and cancer cells. Many researchers reported that taking adequate amount of vitamin C in diet boosts immunity and helps the individual to becomes less prone to diseases and infections. For example, Atherton et al. [14] had reported that the resistance and immunity of chick embryo and tracheal organs against coronavirus have increased by the ingestion of vitamin C. Vitamin C is also known as a weak anti-histamine agent, as it prevents and relieves the individual from various flu-like symptoms, for instance, sneezing, nasal congestion, rhinorrhoea, and sinus congestion [15]. Vitamin C has shown its positive effects on respiratory-related disorders also. With the help of three controlled clinical human trials, it was revealed that groups fed and supplemented with vitamin C showed a lower prevalence of pneumonia, which resulted from the fact that vitamin C might control and reduce the vulnerability to lower respiratory tract infections in some occasions [16]. Furthermore, in a double-blinded controlled trial with old-aged candidates, 200 mg/day of ascorbic acid for about 4 weeks enhanced respiratory conditions [17]. In another analysis of eight randomised control trials (RCTs) in 3135 children, vitamin C administration with 0.5–2 g/day did not inhibit upper respiratory tract infection but decreased the time span of infection by 1.6 days [18]. Many recent cases revealed that the novel COVID-19 leads to lower respiratory tract infections, and therefore, taking vitamin C could be an effective player in the treatment and prevention against this virus (Table 2). In support of this, according to a report by China and the USA, vitamin C supplementation has shown improvement in the conditions of patients suffering from COVID-19 [19]. It has also been reported that vitamin C has an inhibitory effect against various other viruses including influenza, HIV, rabies virus, avian RNA tumour viruses, herpesviruses and paramyxoviruses, parvovirus, and human lymphotropic virus type 1 (HTLV-1) [20]. Dietary sources of vitamin C are given in Table 1.
Vitamin D
It is one of the most crucial vitamins. It is not only a vitamin but also a hormone, which can be produced in our body by the exposure of sunlight. This vitamin plays a very important role in maintaining bone integrity. It is a fat-soluble vitamin that acts as a crucial player in regulating both innate and adaptive immune responses. The major source is obtained from ultraviolet-induced conversion of its precursor 7-dehydrocholesterol in the skin to cholecalciferol, vitamin D3 [21]. According to various studies, calcitriol an active form of vitamin D is activated by ultraviolet radiations, leading to a rise in the production of antiviral peptidase, increasing the oxidative rupture capacity of microphage, and stopping the excessive expression of proinflammatory cytokines, which are a type of signalling molecule that promotes inflammation [22, 23]. Based on a study, it has been demonstrated that vitamin D is also known to improve the innate immunity by upregulating the expression and excretion of dominant antimicrobial peptides in immune cells, which further improves mucosal defence against various viral infections [24]. In support of this, vitamin D has shown a beneficial effect in various other viral infections, such as adding vitamin D to conventional ribavirin therapy for treating patients with chronic hepatitis C virus (HCV) considerably improved the viral response [25]. Another study reported on a decreased risk of influenza and COVID-19 infection, typically due to connected inflammatory status and antimicrobial peptides and by regulating adaptive immunity. This is substantiated by a meta-analysis of RCTs with daily administration of vitamin D seeming to be the most efficacious plan of action [26]. Interestingly, vitamin D administration has also shown to encourage binding of the SARS-CoV-2 cell entry receptor ACE2 to AGTR1 (angiotensin II receptor type 1), thus decreasing the total number of virus particles that could attach to ACE2 and enter the cell [27]. Epidemiological studies have associated vitamin D deficiency to increased sensitivity to acute viral respiratory infections. Many recent reviews assessing various possible mechanisms recommended that vitamin D plays a significant role of the innate immune responses to viral respiratory infections including parainfluenza 1 and 2, respiratory syncytial virus (RSV), and influenza A and B [21]. According to reports by Tangpricha et al. [28], it was revealed that a greater number of cases with low levels of vitamin D were present when winter season was over or about to get over. Moreover, people who work from home or housebound and those who do night shifts at work may be more prone to vitamin D deficiency. Vitamin D-related deficiency disorders are majorly common among the elderly, as they have limited exposure to sunlight [29]. The first case of novel coronavirus was recognised in winter 2019, and the most affected were those belonging to middle-aged or old-aged population. Therefore, one of the causes of this can be insufficient vitamin D. Therefore, at this time, it is particularly important to increase dietary sources of vitamin D (Table 1), as people are subjected to “stay and work from home” alleviating strategies. Additionally, it was also investigated that the bovine coronavirus infection has resulted from decreased levels of vitamin D in calves [30]. Furthermore, a trial is presently initiated (www.clinicaltrials.gov: NCT04334005) with the target of evaluating the potential of vitamin D as an immune-regulating agent. Subjects will be observed to see if there is any improvement in health status in asymptomatic or non-severe symptoms patients infected with COVID-19 along with verifying if vitamin D could control patient deterioration.
Vitamin E
Vitamin E comes under the category of fat-soluble vitamin. Vitamin E includes both tocopherols and tocotrienols. Vitamin E acts as an antioxidant in reducing oxidative stress by binding to free radicals [31••]. This antioxidant activity of vitamin E plays an important role in the human body by helping to slow down cells from any damage. Vitamin E is also known to employ its immune-enhancing effects by scavenging oxygen species to decrease oxidative stress, and it may produce anti-inflammatory effects [32, 33]. Vitamin E also has the potential to protect polyunsaturated fatty acids (PUFAs) from oxidation and to modulate the production of reactive oxygen species (ROS) and reactive nitrogen species (RNS) in the cell membranes. Similarly, vitamin E exists in high concentrations in immune cells, which further protects these cells from any oxidative injury because of their high metabolic activity and PUFA contents [34, 35]. It has also been known that vitamin E regulates the growth and functions of dendritic cells, which are essential for interlacing innate and adaptive immune systems to manage immune response in the body [36, 37]. Vitamin E has been displayed its positive effect in improving T cell immune synapse development and begin T cell activation signals [38]. According to a study, vitamin E supplementation when given to mice suffering from influenza infection, it reduced lung-related pathology and death by the enhancement of T helper 1-type cytokine response, which is known to induce proinflammatory responses against intracellular parasites [39]. Vitamin E supplementation has when given to humans restored interleukin-2 (IL-2) production, which improves T cell proliferation and immune system overall functioning [40]. Increase in the myocardial injury of coxsackievirus B3 (a kind of RNA viruses) infection had been reported in mice which are suffering from vitamin E deficiency [41]. Based on an experimental study, it has also been reported that vitamin E deficiency leads to increased severity of any disease or virus [42]. Additionally, decreased levels of vitamins E and D resulted in bovine coronavirus infection in calves [30]. In particular, ageing is related to dysregulation of the immune system, making people susceptible to more oxidative stress and inflammation. This further results in increased occurrences of infections in the elderly. Thus, increasing dietary sources of vitamin E (Table 1) in the diet of an elderly may be advantageous for their immune function, may provide resistance to infection, and may decrease morbidity because of infections. As the elderly population is more prone to infection, it will be beneficial to investigate vitamin E for potential health benefits against COVID-19. Certainly, a combination of vitamins E and C has been suggested as the beneficial antioxidant therapy for gastric cardia complications of the novel coronavirus [43].
Omega-3 Polyunsaturated Fatty Acids
Omega-3 and omega-6 PUFAs play a crucial role as an arbitrator of inflammation and flexible immune responses [44•]. Long-chain PUFAs principally support anti-inflammatory and proinflammatory processes and effects. PUFAs are known as a precursor of resolvins and prostaglandins respectively [44•]. Begin et al. [45] had examined plasma lipid levels in patients with acquired immunodeficiency syndrome (AIDS) and had discovered a particular and explicit absence of the long-chain PUFAs of omega-3 in their diet, which are found in high quantity in fish oils. Moreover, a compound named protectin D1 derived from omega-3 PUFA acts as a mediator and is known to distinctively reduce the various virus replications, and hence prevent the infection from spreading. The mechanism by which protectin D1 works is via RNA export machinery. It has also been reported that various PUFAs have anti-hepatitis C virus properties [46]. Hence, inculcation of omega-3 and omega-6 PUFAs in diet could serve as a major therapeutic tool for this novel virus, COVID-19. Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) possess anti-inflammatory properties, resulting in the lower level of these inflammatory eicosanoids produced from arachidonic acid that inhibits NFκβ (nuclear factor kappa-light-chain-enhancer of activated B cells) signalling, leading to lesser production of inflammatory cytokines [47,48,49]. In a study, animal models with lung injury showed that administration of fish oil lessens pulmonary accumulation of neutrophils with reduced lung permeability, pulmonary oedema, and cardiopulmonary dysfunction [50]. Coronavirus mainly causes respiratory damage that affects the lung extensively. In this condition, several inflammatory responses (tumour necrosis factor-α, IL-1β, IL-6) occur in the host immune system to protect from coronavirus. This condition is manifested as acute respiratory distress syndrome (ARDS). In the trial of patients with ARDS with a higher dose of EPA and DHA, antioxidants and γ-linolenic acid showed satisfactory effects on respiratory damage [50]. Cyclooxygenase enzyme (COX-1/2) inhibitors were used in some viral infections viz., influenza A. In some previous studies, it had shown that SARS-CoV-1 can bind with COX-2 promotor with heightened expression. However, the role of nonsteroidal anti-inflammatory drugs (NSAIDs) in the case of COVID-19 symptoms is still cloudy to draw any conclusion. But selective inhibition of human microsomal prostaglandin E synthase 1 (mPGES-1) was believed to be a beneficial and viable therapeutic target for SARS-CoV-2. Although it is known that omega fatty acids are a potent candidate for the therapeutic interaction with COX enzymes [51], its role in SARS-CoV-2 infection needs more in-depth studies to use as human trials.
Iron
Iron is one of the most important minerals for every age group. Iron deficiency is directly linked with impairment of immunity, which further results in many viruses and infection [52]. People suffering from iron deficiency are more prone and at risk in developing acute respiratory tract infections [53•], which are a major symptom of COVID-19. Reduced level of iron results in thymus atrophy and affects the activity of naïve T lymphocytes like zinc. Impaired respiratory burst, natural killer cell activity, or decreased T lymphocyte proliferation can be an indication towards iron deficiency. According to a data, T lymphocyte proliferation decreased up to 50 to 60% due to low iron level. Several pre-cilinical trials also suggest that various infections like plasmodia, mycobacterial infections may be dependent on the iron intake of the host. Increased risk of malaria and infections like pneumonia has been found in tropical areas in children irrespective of any age group due to a higher level of iron. Injurious effects of iron intervention upon infection have several justifications that suggest that a higher level of iron can impair immune cell activity [50]. It affects the inflammation and also provides the iron-requiring microorganisms a suitable environment to grow. In that case, co-administration of n3 fatty acids showed the lessened effect of iron. Iron deficiency and beyond a particular threshold level dosage both can harm the immune system. Thus, it is much crucial to maintain an accurate iron level for better immunity. Therefore, iron is an important nutrient in mitigating the risk factors associated with this novel coronavirus. Major food sources of iron are presented in Table 1.
Zinc
Zinc comes under the category of trace minerals. It is required in a very small quantity by the body. It helps in the development and protection of immune cells. Zinc deficiency leads to a decrease in both active and passive immunity and increased proneness to infectious viruses and diseases [54]. Reports have shown that children suffering from zinc deficiency have shown reduced measles that caused morbidity and mortality when fed with zinc supplements [55]. Replication of various RNA-induced viruses can be effectively damaged by rising the dose of intracellular zinc plus ionophores agent of zinc called pyrithione [56]. Additionally, a collaboration of zinc with pyrithione hinders the process of SARS-CoV replication [56]. Recently, many studies provided intuitive data regarding the role of zinc in antiviral immunity and infection. RNA viruses like coronavirus need RNA polymerase to replicate. According to a study, zinc can inhibit RNA polymerase and play a central role in host defence against coronavirus [57]. There is also evidence of zinc ionophore pyrrolidine dithiocarbamate that inhibited the in vitro replication of influenza virus, and it is believed that it might be a potent inhibitor of replication of SARS-CoV in vitro [56]. Zinc binding metallothionein also seemed to act as a significant antiviral agent [58, 59]. The lower level of zinc in the body affects the bone marrow. It lessens the immune precursor cells and decreased productivity of the naïve B lymphocytes, leading to thymic atrophy that also affects T lymphocytes. Innate immunity along with phagocytosis, and other immune cell activities viz., IL-2 and IFN-γ (Interferon), production is compromised due to deficient level of zinc. It also maintains the discharge of neutrophil extracellular traps to arrest invading microbes [60]. Based upon the experimental data, it has been demonstrated that zinc supplement (30 mg/day) increased the proliferation of T lymphocytes in elderly care home residents in the USA [61]. Although the effect of supplementation for respiratory diseases is not clear yet, it lowers the probability of getting respiratory infections, such as common cold [62]. Though recommended dietary allowance (RDA) of zinc according to the National Institutes of Health Dietary Reference Intake (NIH DRI) is 8–11 mg/day in the case of adults (upper intake level threshold 40 mg/day), to control the proliferation of RNA viruses viz., coronavirus, 30–50 mg/day might be helpful [63]. Therefore, zinc may be an effective game changer in reducing and preventing coronavirus-related symptoms such as diarrhoea and respiratory-related disorders.
Importance of Dietary Supplements
Arginine
L-arginine, when reacted with the enzyme called nitrous oxide synthase, produces a compound known as nitrous oxide. This compound has beneficial effects on lung physiology and cardiovascular functions [64]. Therefore, supplementing arginine in diet could be beneficial in combating COVID-19. Amino acids viz., arginine, are the precursor of polyamines that play a significant role in the regulation of DNA (deoxyribonucleic acid) replication and cell division. For optimal antibody production, its activity is believed to be reliable on the level of proteins present in the system. It has been recorded that the supplementation of arginine showed a significantly increased response of T cell function as well as its number compared with control subjects [65].
Carnitine
Combination of carnitine and acylcarnitine has the chief role in β-oxidation. This combination is also important in modulating the mechanisms of the immune system and the nervous system. The biologically active form of carnitine is L-carnitine. L-carnitine plays an essential role in the production of energy and fatty acid metabolism. This can further improve and control the immune-modulatory functions in diabetic patients, neuropathic pain, and other liver and kidney-related diseases [66, 67]. Thus, carnitine could be a good option to prevent the harm caused by COVID-19.
Probiotics
Probiotics are world-famous in improving the gut health and thus improves digestion of nutrients in the body. Probiotics are microorganisms which when ingested into the host body confer a number of health benefits generally by improving and restoring the gut flora. Probiotics are broadly known to influence the immune system and maintain immune function balance. They are known to bring about these effects by virtue of their cell structures such as DNA, peptidoglycan, and flagellin. They may act directly or indirectly by modulating the gut microbiota. These probiotic microbes have the ability to inhibit pathogenic bacteria by releasing various metabolites. In addition to this, these organisms compete for the sites of adhesion and thus inhibits the pathogens from binding to the host epithelium [68]. Probiotic bacteria can also interact with the host’s gut-related immune tissues. It is through these communications that probiotics affect immune function, even at sites far away from the gut [69, 70]. A large number of experiments have analysed the effect of several probiotic microbes, either single or in combination, on immune response and function, and inflammatory and infection conditions in humans [71]. Recent studies also reported that probiotic supplementation improved the vaccination responses in individuals [72, 73]. Various systematic reviews and meta-analyses verified that probiotics improve antibody response to influenza vaccination in adult subjects [74, 75]. Many of the studies reported that probiotics supplementation results in decreased prevalence and intensity of respiratory tract infections [76,77,78,79]. The gut microbiota also known as live microbes in the digestive tracts of humans appear to be protective against respiratory infections, as its absence in mice leads to viral respiratory infection and impaired immune response [80, 81]. Hence, taking an appropriate dosage of probiotics during this pandemic could be helpful for combating the current situation and also for the future.
Immuno-enhancing Foods
-
Fruits: oranges/sweet lime, pineapple, fresh berries, papaya, kiwi, guava, and tomatoes
-
Vegetables: carrots/beetroot, spinach, cabbage, cauliflower, broccoli, eggplant/brinjal, and capsicum/bell peppers
-
Nuts: almonds (soaked overnight) and walnuts
-
Ginger
-
Garlic
-
Turmeric
-
Green tea
-
Lemon
-
Liquid: 2.5–3 l/day (plain water, coconut water, green tea, freshly homemade vitamin C fruit juices, milk, buttermilk)
Immunity-Boosting Recipes
Turmeric Sauerkraut
Instructions
Grate 1/2 head of fresh and washed cabbage. Add 1 and 1/2 teaspoons of salt and massage it well with the cabbage. This results in complete opening up of pores of cabbage, which enables the natural microbe from the cabbage itself to get inside the pores for improved fermentation. Add onion, turmeric, and garlic according to the taste. Keep it in an air-tight jar. Keep the turmeric sauerkraut down immovably and add a small amount of water. Put a cabbage leaf on top to stop oxidation. Let it ferment for 2–3 days.
Advantages
Boosts immunity and helps in making gut healthy, contains probiotics, anti-inflammatory benefits, and vitamin C-rich.
Ginger Peanut Soy Curry
Instructions
Soak the soybean in clean water for 30 min. Over medium heat, fry the mustard seeds in any healthy oil, preferably olive oil, for 2–3 min. Add and cook the onion till it becomes soft and brown. Add ginger, garlic, and salt. Cook this mixture for a few minutes till the fragrance starts coming. Then, add tomato paste and peanut butter (if desired) and mix it thoroughly. Boil it and lower the flame to a simmer. Cook uncovered for 15 min till it becomes a sauce. Add spinach and cook for 5 min till it becomes withered. Fresh coriander leaves can be topped for taste.
Advantages
Protein-rich, flu-fighting property, immunity-boosting recipe, antiviral and anti-bacterial properties, increase the circulation, and cleanse the blood.
Immunity Booster Custard
Instructions
Prepare coconut paste at home. Buy lactose-free yoghurt from the market. Yoghurt can also be prepared at home. If desired, almond milk can also be used in place of yoghurt. Peel the fresh pomegranate. Mix all the ingredients gently. Add chia seeds in the end.
Advantages
Helps in decreasing dehydration, omega-3 and omega-6-rich, and vitamin C-rich.
Conclusion
In this review, all the possible nutrition-related interventions, the role of different nutrients, and immunity-boosting recipes for prevention of novel coronavirus have been discussed. It has been found that these general interventions are very crucial to improve immunity against RNA viral infection. Inadequate nutrition often results in a weak immune response. Hence, the inclusion of some nutrition interventions can surely act as immune-boosters, which can help us to avoid this type of fatal diseases and their consequences, as we know, prevention is always better than cure. It has been suggested that the nutritional status of suspected or affected patients should be done before beginning any general treatments, so that specific nutrition intervention can be created for them.
Abbreviations
- SARS:
-
severe acute respiratory syndrome
- MERS:
-
Middle East respiratory syndrome
- COVID-19:
-
coronavirus disease 2019
- SARS-CoV-2:
-
severe acute respiratory syndrome coronavirus 2
- WHO:
-
World Health Organization
- PHEIC:
-
Public Health Emergency of International Concern
- RNA:
-
ribonucleic acid
- ACE2:
-
angiotensin-converting enzyme 2
- RT-PCR:
-
reverse transcription-polymerase chain reaction
- CVD:
-
cardiovascular disease
- RAR:
-
retinoic acid receptor
- RXR:
-
retinoid X receptor
- NK T cells:
-
natural killer T cells
- HIV:
-
human immunodeficiency virus
- IBV:
-
bronchitis virus
- RCTs:
-
randomised control trials
- AGTR1:
-
angiotensin II receptor type 1
- RSV:
-
respiratory syncytial virus
- PUFAs:
-
polyunsaturated fatty acids
- ROS:
-
reactive oxygen species
- RNS:
-
reactive nitrogen species
- IL:
-
interleukin
- AIDS:
-
acquired immunodeficiency syndrome
- EPA:
-
eicosapentaenoic acid
- DHA:
-
docosahexaenoic acid
- NFκβ:
-
nuclear factor kappa-light-chain-enhancer of activated B cells
- ARDS:
-
acute respiratory distress syndrome
- COX:
-
cyclooxygenase enzymes
- NSAIDs:
-
nonsteroidal anti-inflammatory drugs
- mPGES-1:
-
microsomal prostaglandin E synthase 1
- INF:
-
interferon
- RDA:
-
recommended dietary allowance
- DRI:
-
dietary reference intake
- NIH:
-
National institutes of Health
- DNA:
-
deoxyribonucleic acid
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Cinelli M, Quattrociocchi W, Galeazzi A, Valensise CM, Brugnoli E, Schmidt AL et al. The COVID-19 social media infodemic. arXiv preprint arXiv:2003.05004. 2020
• Hui DS, Azhar EI, Madani TA, Ntoumi F, Kock R, Dar O, et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—the latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis. 2020;91:264–6 This article gives a detailed study about the outbreak of the novel COVID-19 along with the reason behind it. An analytical study has been done to enlighten regarding SARS-COV, SARS-COV2, and other previous infections from coronavirus and their consequences.
De Groot RJ, Baker SC, Baric RS, Brown CS, Drosten C, Enjuanes L, et al. Commentary: Middle East respiratory syndrome coronavirus (MERS-CoV): announcement of the Coronavirus Study Group. J Virol. 2013;87(14):7790–2.
Paraskevis D, Kostaki EG, Magiorkinis G, Panayiotakopoulos G, Sourvinos G, Tsiodras S. Full-genome evolutionary analysis of the novel corona virus (2019-nCoV) rejects the hypothesis of emergence as a result of a recent recombination event. Infect Genet Evol. 2020;79:104212.
Chang HK, Hou WS. Retinoic acid modulates interferon-γ production by hepatic natural killer T cells via phosphatase 2A and the extracellular signal-regulated kinase pathway. J Interf Cytokine Res. 2015;35(3):200–12.
Worbs T, Hammerschmidt SI, Förster R. Dendritic cell migration in health and disease. Nat Rev Immunol. 2017;17(1):30–48.
Villamor E, Mbise R, Spiegelman D, Hertzmark E, Fataki M, Peterson KE, et al. Vitamin A supplements ameliorate the adverse effect of HIV-1, malaria, and diarrheal infections on child growth. Pediatrics. 2002;109(1):e6.
Jee J, Hoet AE, Azevedo MP, Vlasova AN, Loerch SC, Pickworth CL, et al. Effects of dietary vitamin A content on antibody responses of feedlot calves inoculated intramuscularly with an inactivated bovine coronavirus vaccine. Am J Vet Res. 2013;74(10):1353–62.
West CE, Sijtsma SR, Kouwenhoven B, Rombout JH, van der Zijpp AJ. Epithelia-damaging virus infections affect vitamin A status in chickens. J Nutr. 1992;122(2):333–9.
Timoneda J, Rodríguez-Fernández L, Zaragozá R, Marín MP, Cabezuelo MT, Torres L, et al. Vitamin A deficiency and the lung. Nutrients. 2018;10(9):1132.
Wannamethee SG, Lowe GD, Rumley A, Bruckdorfer KR, Whincup PH. Associations of vitamin C status, fruit and vegetable intakes, and markers of inflammation and hemostasis. Am J Clin Nutr. 2006;83(3):567–74.
Duarte TL, Cooke MS, Jones GD. Gene expression profiling reveals new protective roles for vitamin C in human skin cells. Free Radic Biol Med. 2009;46(1):78–87.
Mohammed BM, Fisher BJ, Kraskauskas D, Ward S, Wayne JS, Brophy DF, et al. Vitamin C promotes wound healing through novel pleiotropic mechanisms. Int Wound J. 2016;13(4):572–84.
Atherton JG, Kratzing CC, Fisher A. The effect of ascorbic acid on infection of chick-embryo ciliated tracheal organ cultures by coronavirus. Arch Virol. 1978;56(3):195–9.
Field CJ, Johnson IR, Schley PD. Nutrients and their role in host resistance to infection. J Leukoc Biol. 2002;71(1):16–32.
Hemilä H. Vitamin C intake and susceptibility to pneumonia. Pediatr Infect Dis J. 1997;16(9):836–7.
Hunt C, Chakravorty NK, Annan G, Habibzadeh N, Schorah CJ. The clinical effects of vitamin C supplementation in elderly hospitalised patients with acute respiratory infections. Int J Vitam Nutr Res. 1994;64(3):212–9.
Vorilhon P, Arpajou B, Roussel HV, Merlin E, Pereira B, Cabaillot A. Efficacy of vitamin C for the prevention and treatment of upper respiratory tract infection. A meta-analysis in children. Eur J Clin Pharmacol. 2019;75(3):303–11.
Rodríguez L, Cervantes E, Ortiz R. Malnutrition and gastrointestinal and respiratory infections in children: a public health problem. Int J Environ Res Public Health. 2011;8(4):1174–205.
Lallement A, Zandotti C, Brouqui P. Persistent parvovirus B19 viremia with chronic arthralgia treated with ascorbic acid: a case report. J Med Case Rep. 2015;9(1):1.
Zdrenghea MT, Makrinioti H, Bagacean C, Bush A, Johnston SL, Stanciu LA. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev Med Virol. 2017;27(1):e1909.
Palacios C, Gonzalez L. Is vitamin D deficiency a major global public health problem? J Steroid Biochem Mol Biol. 2014;144:138–45.
Wang TT, Dabbas B, Laperriere D, Bitton AJ, Soualhine H, Tavera-Mendoza LE, et al. Direct and indirect induction by 1, 25-dihydroxyvitamin D3 of the NOD2/CARD15-defensin β2 innate immune pathway defective in Crohn disease. J Biol Chem. 2010;285(4):2227–31.
Cannell JJ, Vieth R, Umhau JC, Holick MF, Grant WB, Madronich S, et al. Epidemic influenza and vitamin D. Epidemiol Infect. 2006;134(6):1129–40.
Abu-Mouch S, Fireman Z, Jarchovsky J, Zeina AR, Assy N. Vitamin D supplementation improves sustained virologic response in chronic hepatitis C (genotype 1)-naïve patients. World J Gastroenterol: WJG. 2011;17(47):5184–90.
Bergman P, Lindh ÅU, Björkhem-Bergman L, Lindh JD. Vitamin D and respiratory tract infections: a systematic review and meta-analysis of randomized controlled trials. PLoS One. 2013;8(6).
Iddir M, Brito A, Dingeo G, Fernandez Del Campo SS, Samouda H, La Frano MR, et al. Strengthening the immune system and reducing inflammation and oxidative stress through diet and nutrition: considerations during the COVID-19 crisis. Nutrients. 2020;12(6):1562.
Tangpricha V, Pearce EN, Chen TC, Holick MF. Vitamin D insufficiency among free-living healthy young adults. Am J Med. 2002;112(8):659–62.
Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80(6):1678S–88S.
Nonnecke BJ, McGill JL, Ridpath JF, Sacco RE, Lippolis JD, Reinhardt TA. Acute phase response elicited by experimental bovine diarrhea virus (BVDV) infection is associated with decreased vitamin D and E status of vitamin-replete preruminant calves. J Dairy Sci. 2014;97(9):5566–79.
•• Galmés S, Serra F, Palou A. Vitamin E metabolic effects and genetic variants: a challenge for precision nutrition in obesity and associated disturbances. Nutrients. 2018;10(12):1919 This study reveals that besides saving cells from oxidative damage as a potent antioxidant, vitamin E also acts as an immunomodulator. Intake of vitamin E can help to build healthy immune system.
Lee GY, Han SN. The role of vitamin E in immunity. Nutrients. 2018;10(11):1614.
Wu D, Meydani SN. Vitamin E, immunity, and infection. Nutrition, Immunity, and Infection, vol. 22. Boca Raton: CRC Press; 2017. p. 197–212.
Wu D, Lewis ED, Pae M, Meydani SN. Nutritional modulation of immune function: analysis of evidence, mechanisms, and clinical relevance. Front Immunol. 2019;9:3160.
Zabetakis I, Lordan R, Norton C, Tsoupras A. COVID-19: the inflammation link and the role of nutrition in potential mitigation. Nutrients. 2020;12(5):1466.
Buendía P, Ramírez R, Aljama P, Carracedo J. Klotho prevents translocation of NFκB. In Vitamins & Hormones 2016 Jan 1 (Vol. 101, pp. 119-150). Academic Press.
Xuan NT, Trang PT, Van Phong N, Toan NL, Bac ND, Nguyen VL, et al. Klotho sensitive regulation of dendritic cell functions by vitamin E. Biol Res. 2016;49(1):45.
Marko MG, Pang HJ, Ren Z, Azzi A, Huber BT, Bunnell SC, et al. Vitamin E reverses impaired linker for activation of T cells activation in T cells from aged C57BL/6 mice. J Nutr. 2009;139(6):1192–7.
Han SN, Wu D, Ha WK, Beharka A, Smith DE, Bender BS, et al. Vitamin E supplementation increases T helper 1 cytokine production in old mice infected with influenza virus. Immunology. 2000;100(4):487–93.
Lewis ED, Meydani SN, Wu D. Regulatory role of vitamin E in the immune system and inflammation. IUBMB Life. 2019;71(4):487–94.
Beck MA, Kolbeck PC, Rohr LH, Shi Q, Morris VC, Levander OA. Vitamin E deficiency intensifies the myocardial injury of coxsackievirus B3 infection of mice. J Nutr. 1994;124(3):345–58.
Beck MA. Increased virulence of coxsackievirus B3 in mice due to vitamin E or selenium deficiency. J Nutr. 1997;127(5):966S–70S.
Wang JZ, Zhang RY, Bai J. An anti-oxidative therapy for ameliorating cardiac injuries of critically ill COVID-19-infected patients. Int J Cardiol. 2020;6.
• Cai C, Koch B, Morikawa K, Suda G, Sakamoto N, Rueschenbaum S, et al. Macrophage-derived extracellular vesicles induce long-lasting immunity against hepatitis C virus which is blunted by polyunsaturated fatty acids. Front Immunol. 2018;9:723 According to this study, intake of PUFA in daily diet blunt the long-lasting immune effect against viral infections like hepatitis C virus. Extracellular cellular vesicles are considered to be a mediator of inter-cellular communication. Microphage-derived extracellular vesicles get affected due to PUFA.
Begin ME, Manku MS, Horrobin DF. Plasma fatty acid levels in patients with acquired immune deficiency syndrome and in controls. Prostaglandins Leukot Essent Fat Acids. 1989;37(2):135–7.
Leu GZ, Lin TY, Hsu JT. Anti-HCV activities of selective polyunsaturated fatty acids. Biochem Biophys Res Commun. 2004;318(1):275–80.
Calder PC. Marine omega-3 fatty acids and inflammatory processes: effects, mechanisms and clinical relevance. Biochimica et Biophysica Acta (BBA)-Molecular and Cell Biology of Lipids. 2015;1851(4):469–84.
Calder PC. Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochem Soc Trans. 2017;45(5):1105–15.
Smeitink J, Jiang X, Pecheritsyna S, Renkema H, van Maanen R, Beyrath J. Hypothesis: mPGES-1-derived prostaglandin E2, a so far missing link in COVID-19 pathophysiology?
Calder PC. Nutrition, immunity and Covid-19. BMJ Nutrition, Prevention & Health. 2020 May 20:bmjnph-2020.
Lordan R, Redfern S, Tsoupras A, Zabetakis I. Inflammation and cardiovascular disease: are marine phospholipids the answer? Food Funct. 2020;11(4):2861–85.
Wessling-Resnick M. Crossing the iron gate: why and how transferrin receptors mediate viral entry. Annu Rev Nutr. 2018;38:431–58.
• Jayaweera JA, Reyes M, Joseph A. Childhood iron deficiency anemia leads to recurrent respiratory tract infections and gastroenteritis. Sci Rep. 2019;9(1):1–8 In this article consequences of the iron deficiency have been mentioned. Iron deficient anaemia in childhood leads to respiratory tract infections. Proper intake of iron-rich food may help to prevent the respiratory infections.
Tuerk MJ, Fazel N. Zinc deficiency. Curr Opin Gastroenterol. 2009;25(2):136–43.
Awotiwon AA, Oduwole O, Sinha A, Okwundu CI. Zinc supplementation for the treatment of measles in children. Cochrane Database Syst Rev. 2017;6.
TeVelthuis AJ, van den Worm SH, Sims AC, Baric RS, Snijder EJ, van Hemert MJ. Zn2+ inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010;6(11).
Kaushik N, Subramani C, Anang S, Muthumohan R, Nayak B, Ranjith-Kumar CT, et al. Zinc salts block hepatitis E virus replication by inhibiting the activity of viral RNA-dependent RNA polymerase. J Virol. 2017;91(21):e00754–17.
Read SA, Obeid S, Ahlenstiel C, Ahlenstiel G. The role of zinc in antiviral immunity. Adv Nutr. 2019;10(4):696–710.
Subramanian Vignesh K, Deepe GS Jr. Metallothioneins: emerging modulators in immunity and infection. Int J Mol Sci. 2017;18(10):2197.
Hasan R, Rink L, Haase H. Zinc signals in neutrophil granulocytes are required for the formation of neutrophil extracellular traps. Innate Immun. 2013;19(3):253–64.
Barnett JB, Dao MC, Hamer DH, Kandel R, Brandeis G, Wu D, et al. Effect of zinc supplementation on serum zinc concentration and T cell proliferation in nursing home elderly: a randomized, double-blind, placebo-controlled trial. Am J Clin Nutr. 2016;103(3):942–51.
Gammoh NZ, Rink L. Zinc in infection and inflammation. Nutrients. 2017;9(6):624.
McCarty MF, DiNicolantonio JJ. Nutraceuticals have potential for boosting the type 1 interferon response to RNA viruses including influenza and coronavirus. Prog Cardiovasc Dis. 2020;12.
Melis M, Mastinu M, Arca M, Crnjar R, Barbarossa IT. Effect of chemical interaction between oleic acid and L-arginine on oral perception, as a function of polymorphisms of CD36 and OBPIIa and genetic ability to taste 6-n-propylthiouracil. PLoS One. 2018;13(3).
Kim SH, Roszik J, Grimm EA, Ekmekcioglu S. Impact of l-arginine metabolism on immune response and anticancer immunotherapy. Front Oncol. 2018;8:67.
Yuan S, Chu H, Chan JF, Ye ZW, Wen L, Yan B, et al. SREBP-dependent lipidomic reprogramming as a broad-spectrum antiviral target. Nat Commun. 2019;10(1):1–5.
Yates MK, Chatterjee P, Flint M, Arefeayne Y, Makuc D, Plavec J, et al. Probing the Effects of pyrimidine functional group switches on acyclic fleximer analogues for antiviral activity. Molecules. 2019;24(17):3184.
Hemarajata P, Versalovic J. Effects of probiotics on gut microbiota: mechanisms of intestinal immunomodulation and neuromodulation. Ther Adv Gastroenterol. 2013;6(1):39–51.
Thomas CM, Versalovic J. Probiotics-host communication: modulation of signaling pathways in the intestine. Gut Microbes. 2010;1(3):148–63.
Ahern PP, Maloy KJ. Understanding immune–microbiota interactions in the intestine. Immunology. 2020;159(1):4–14.
Lomax AR, Calder PC. Probiotics, immune function, infection and inflammation: a review of the evidence from studies conducted in humans. Curr Pharm Des. 2009;15(13):1428–518.
Boge T, Rémigy M, Vaudaine S, Tanguy J, Bourdet-Sicard R, Van Der Werf S. A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine. 2009;27(41):5677–84.
Rizzardini G, Eskesen D, Calder PC, Capetti A, Jespersen L, Clerici M. Evaluation of the immune benefits of two probiotic strains Bifidobacterium animalis ssp. lactis, BB-12® and Lactobacillus paracasei ssp. paracasei, L. casei 431® in an influenza vaccination model: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2012;107(6):876–84.
Lei WT, Shih PC, Liu SJ, Lin CY, Yeh TL. Effect of probiotics and prebiotics on immune response to influenza vaccination in adults: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2017;9(11):1175.
Yeh TL, Shih PC, Liu SJ, Lin CH, Liu JM, Lei WT, et al. The influence of prebiotic or probiotic supplementation on antibody titers after influenza vaccination: a systematic review and meta-analysis of randomized controlled trials. Drug Des Dev Ther. 2018;12:217.
Ozen M, Kocabas Sandal G, Dinleyici EC. Probiotics for the prevention of pediatric upper respiratory tract infections: a systematic review. Expert Opin Biol Ther. 2015;15(1):9–20.
Laursen RP, Hojsak I. Probiotics for respiratory tract infections in children attending day care centers—a systematic review. Eur J Pediatr. 2018;177(7):979–94.
Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;13:130(5).
Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning L, et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19). Front Immunol. 2020;11:827.
Clarke TB. Early innate immunity to bacterial infection in the lung is regulated systemically by the commensal microbiota via nod-like receptor ligands. Infect Immun. 2014;82(11):4596–606.
Zhang N, He QS. Commensal microbiome promotes resistance to local and systemic infections. Chin Med J. 2015;128(16):2250–5.
Chapple LA, Gan M, Louis R, Yaxley A, Murphy A, Yandell R. Nutrition-related outcomes and dietary intake in non–mechanically ventilated critically ill adult patients: a pilot observational descriptive study. Aust Critical Care. 2020;23.
Author information
Authors and Affiliations
Contributions
Savita Budhwar: conceptualization, validation, and supervision. Kashika Sethi: literature search, writing of original draft, writing of review, and editing. Manali Chakraborty: literature search, writing of original draft, writing of review, and editing.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Budhwar, S., Sethi, K. & Chakraborty, M. A Rapid Advice Guideline for the Prevention of Novel Coronavirus Through Nutritional Intervention. Curr Nutr Rep 9, 119–128 (2020). https://doi.org/10.1007/s13668-020-00325-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13668-020-00325-1