Abstract
Twelve indolyl diketopiperazines 1–12 were isolated from the mycelia culture of Aspergillus penicilliodes Speg., a dominant microorganism from the post fermentation process of ripe Pu-er tea. Their structures were elucidated by extensive spectroscopic methods. Among them, trypostatins C (1) and D (2) featuring with a rare methyl vinyl ketone side chain at C-2 are new compounds, while 3 and 4 were obtained for the first time from nature source. The isolates 3–12 did not show obvious cytotoxicities against five human cancer cell lines at a concentration of 40 μM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Diketopiperazines (DKPs) with a scaffold of 2,5-diketopiperazine formed via the condensation of two amino acid residues. Their conformationally constrained six-membered rings made DKPs an increasing attractive pharmacophore in medicinal chemistry, with a broad pharmacological activity spectrum, e.g., antibacterial [1, 2], antifungal [3, 4], antiviral [5], anticancer [6, 7], immunosuppressive [8], neuro-protection [9], and anti-hyperglycemic [10] activities. To date, DKPs have been commonly found in the marine-derived microorganisms and endophytic fungi. The natural ones with anticancer activities were mainly isolated from Aspergillus and Penicillium genera [11]. Among which, (−)-phenylahistin (halimide) obtained from A. ustus NSC-F038, was a representative DKP with a prenylated chain. Its synthetic analogue, NPI-2358, had been approved for the phase-II clinic trial for the treatment of lymphoma or solid lung cancer as a vascular disrupting agent [12].
Aspergillus penicilliodes Speg. was isolated previously from the post-fermentation process of ripe Pu-er tea by our group [13]. In the research of the characteristic components responsible for health improvement of ripe Pu-er tea, the strain KIBA0502 of A. penicilliodes was studied chemically for its metabolites. This led to the identification of 12 indolyl diketopiperazines. Trypostatins C (1) and D (2) featuring with a rare methyl vinyl ketone side chain at C-2 are new compounds, while 3 and 4 were obtained for the first time from nature source. Their structures were determined by extensive spectroscopic analysis and by comparison with literature values. Most of the isolates were tested for their cytotoxicities against five human cancer cell lines.
2 Results and Discussion
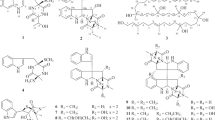
The ethyl acetate extract of the mycelia culture of A. penicilliodes (strain KIBA0502) isolated previously from the fermentative stacks of Pu-er tea was applied to repeated column chromatography over silica gel and Sephadex LH-20, followed with semi-preparative HPLC, to afford 12 indolyl diketopiperazines (1–12) (Fig. 1). Two of them, 1 and 2, are new compounds.
Tryprostatin C (1) was obtained as a yellow amorphous powder. Its molecular formula was determined to be C21H23N3O4 by the HRESIMS (m/z 404.1587 [M+Na]+). The 13C NMR and DEPT spectra of 1 displayed the occurrence of 21 carbon signals assignable to one ketone (δC 199.6), two amide carbonyl (δC 169.4, 165.3), one methoxy (δC 55.7), one methyl (δC 32.0), four methylenes (δC 45.6, 28.5, 25.9, 22.7), two nitrogen-bearing (δC 55.6, 59.3) and two olefinic (δC 129.5, 119.3) methines, and eight aromatic carbons arising from a tri-substituted indolyl unit. The 1H NMR spectrum of 1 showed the presence of one cis-coupled double bond (δH 6.76, 6.09, each 1H, d, J = 12.0 Hz, H-18, H-19), a set of ABX coupled aromatic protons [δH 7.44 (d, J = 8.9 Hz, H-4), 6.80 (dd, J = 2.2, 8.9 Hz, H-5), 6.89 (d, J = 2.2 Hz, H-7)] attributable to a C-6 substituted indolyl unit, two down-field shifted methines [δH 4.23 (dd, J = 11.6, 4.2 Hz, H-9), 3.99 (dd, J = 7.4, 7.5 Hz, H-12)] assignable to a 2,5-diketopiperazine skeleton, one methoxy (δH 3.81, s) and one methyl (δH 2.33, s). From the well-defined coupling sequences and spatial relationships, three sub-structural fragments could be constructed for 1, consisting of one 2,3,6-trisubstituted indole, one 2,5-diketopiperazineand one methyl vinyl ketone moieties. These were subsequently reinforced by correlative interpretation of the 2D NMR spectra (Fig. 2), which allowed the unambiguous assignment of all the 1H and 13C NMR signals. The 1H-1H COSY correlations constructed obviously three partial structures of –C(4)H–C(5)H–, –C(8)H–C(9)H–NH– and –C(12)H–C(13)H2–C(14)H2–C(15)H2– in 1. In the HMBC spectrum, correlation of the methoxy protons at δH 3.81 with C-6 (δC 159.7) indicated its location on C-6. The HMBC correlations of H2-8 (δH 3.01, 3.78) with C-2 (δC 131.7), C-3 (δC 117.7), C-3a (δC 122.0), C-9 (δC 55.6), C-17 (δC 165.3) revealed the indole and 2,5-diketopiperazine units were linked with each other through a methylene bridge of C-8. Correlations of δH 2.33 (H3-21) with C-20 (δC 199.6) and C-19 (δC 119.3), and both δH 6.76 (H-18) and 6.09 (H-19) with C-20 (δC 199.6) confirmed the presence of methyl vinyl ketone partial structure, which were determined to be connected to the indolyl C-2 by the HMBC correlations of H-19 (δH 6.09) with C-2 (δC 131.7) and H-18 (δH 6.76) with C-3 (δC 117.7). Comparison of the NMR data of 1 with those of tryprostatin A [14] manifested that 1 had a similar indolyl diketopiperazine skeleton, except for the presence of a methyl vinyl ketone side chain in 1, instead of an isoprenyl group in tryprostatin A. The relative configuration of 1 was established by ROESY experiment. In which, correlation of H-9 with H-12 indicated both protons located at the same side. Based on comparison of its specific rotation with that of tryprostatin A [15], the absolute stereochemistry at C-9 and C-12 in 1 was assigned to be both as S configurations, which are the same to those of the other known 2,5-diketopiperazine-type compounds. On the basis of the above evidences, the structure of tryprostatin C (1) was deduced as shown in Fig. 1.
Compound 2 obtained as yellow amorphous powder, had a molecular formula of C21H23N3O4, as determined by the HRESIMS (m/z 404.1581 [M+Na]+), which was same to that of 1. The 1D NMR spectroscopic data were almost the same with those of 1, suggesting that 2 had the same indole diketopiperazine skeleton. The only different between 2 and 1 was the appearance of a trans-coupled double bond [(δH 7.48, 6.36, each 1H, d J = 16.0 Hz, H-18, H-19)] in 2, instead of the cis form in 1. The 2D NMR spectra further revealed that structure of 2 as shown in Fig. 1. The configuration of 2 was deduced to be the same as that of 1, according to the ROESY correlation of H-9 with H-12, and further confirmed by the specific rotation. Therefore, the absolute configuration of tryprostatin D (2) was established to be 9S,12S.
It is noted, at the room temperature, compound 2 changed rapidly to 1 in a methanol or chloroform solutions. The cis conformation of double bond in 1 is obviously more stable than the trans form in 2. This is opposite to the most cases that trans-coupled double bonds are more stable than the cis ones. It might be the formation of an intramolecular hydrogen bond between 1-NH and the C-20 ketone that caused the C-18/C-19 double bond to be cis form in 1.
Compounds 3–12 were 10 known indolyl diketopiperazines. They were identified as methyl(S)-{2-((3R,4aS,9aR)-hydroxy-2-oxo-3-(4-oxoquinazolin-3(4H)-yl)-2,3,4,4a, 9,9a-hexahydro-1H-pyrido[2,3-b]indol-1-yl)propanoate}(3), methyl(S)-{2-((3R,4aR, 9aS)-4a-hydroxy-2-oxo-3-(4-oxoquinazolin-3(4H)-yl)-2,3,4,4a,9,9a-hexahydro-1H- pyrido [2, 3] indol-1-yl)propanoate}(4) [16, 17], chaetominine (5) [18, 19], brevianamide F (6) [20], 6-methoxyspirotryprostatin B (7) [21], verruculogen (8) [22], fumitremorgin C (9) [23], cyclotryprostatin A (10), cyclotryprostatin B (11) and cyclotryprostatin C (12) [24], respectively, by comparison of their spectroscopic data with literature values, and single-crystal X-ray diffraction in the case of 5. All of the isolates were obtained from A. penicilliodes for the first time.
The known compounds 3–12 were evaluated for their cytotoxic activities against human myeloid leukemia HL-60, hepatocellular carcinoma SMMC7721, lung cancer A-549, breast cancer MCF-7, and colon cancer SW480 cell lines. However, no compounds showed obvious activity at a concentration of 40 μM.
3 Experimental
3.1 General Experimental Procedures
Optical rotations were measured with a P-1020 polarimeter (JASCO, Tokyo, Japan). UV spectra were recorded on a Shimadzu UV2401A ultraviolet–visible spectrophoto-meter. HREIMS spectra were run on a Waters Autospec Premier P776. NMR spectra were measured in CD3OD and recorded on a Bruker DRX-600 spectrometer, using TMS as an internal standard. Chemical shifts were reported in units of δ (ppm) and coupling constants (J) were expressed in Hz. Column chromatography (CC) were carried out silica gel (200–300 mesh, Qingdao Haiyang Chemical Co. Ltd., Qingdao, China), Sephadex LH-20 (25–100 μm, GE Healthcare Bio-Science AB). An Agilent series 1260 (Agilent Technologies) were used for semi-preparative HPLC with an Agilent ZORBAX SB-C18 column (5 μm, 250 mm × 9.4 mm). TLC was performed on pre-coated TLC plates (0.2–0.25 mm thickness, GF254 Silica gel, Qingdao Hailang Chemical Co. Ltd., Qingdao, China) with compounds visualized by spraying with anisaldehyde-sulfuric acid reagent and careful heating.
3.2 Fungal Material
Aspergillus penicilliodes Speg. was isolated previously by our group from the fermentative stacks of Pu-er tea produced at Puer specific tea factory in Puer city, Yunnan Province, China, and deposited in China general microbiological culture collection center (CGMCC), Institute of Microbiology, Chinese Academy of Sciences [13]. A loop of spores from a colony growing on potato dextrose agars was inoculated into potato dextrose seed broth (20 g glucose, 200 g potato, 4 g peptone, 0.5 g MgSO4, 1.0 g KH2PO4, and 1000 mL distilled water) for 3 days in the dark at 28 °C, 140 r/min. The seed culture (10 mL) was inoculated into 100 × 500 mL Erlenmeyer flasks, each containing 100 g of wheat immersed in waterand sterilized. Fermentation was conducted under stationary condition for 20 days in the dark at 28 °C.
3.3 Extraction and Isolation
The mycelia culture was extracted three times with CHCl3–MeOH (1:1 v/v). After concentrated in vacuo, the resultant extract was suspended into H2O and partitioned successively with petroleum ether (4× 3 L), EtOAc (4× 3 L), and n-BuOH (4× 3 L). The EtOAc fraction (40.5 g) was subjected to a silica gel column, eluting with CHCl3–MeOH (100:0, 98:2, 95:5, 90:10, 85:15, 80:20 v/v) to afford six fractions (Fr. 1-6), which were combined based on TLC analyses. Further separation of Fr.3 (11.0 g) by silica gel CC, using CHCl3–MeOH (100, 100:1, 100:2 v/v) as the eluent afforded 12 sub-fractions (subfr.3-1-subfr.3-12). Subfr.3-3 (30 mg) and subfr.3-5 (0.5 g) were separately purified by semi-preparative HPLC (70% MeOH–H2O) to give 1 (2.7 mg) and 2 (1.3 mg), and 3 (3.2 mg), 4 (5.0 mg) and 5 (80 mg), respectively. Fr.4 (8.0 g) was applied to Sephadex LH-20 CC, eluting with MeOH–CHCl3 (1:1) to afford five sub-fractions (subfr.4-1-subfr.4-5). Subfr.4-3 and Subfr.4-6 were separately subjected to silica gel CC, eluting with a gradient of increasing acetone (0–100%) in petroleum ether to afford subfr. 4-3-1-subfr.4-3-7 and subfr.4-6-1-subfr.4-6-8, respectively. Semi-preparative HPLC (60% aq. MeOH) purification of subfr.4-3-2 (60 mg) and subfr. 4-6-5 (150 mg) afforded 6 (7.0 mg), and 9 (8.2 mg), 10 (23 mg) and 11 (3.0 mg), respectively. Fr.5 (5.5 g) was separated into eightsub-fractions (subfr.5-1-subfr.5-8) by Sephadex LH-20 CC (MeOH-CHCl3, 1:1). Subfr.5-4 (62 mg) was purified by semi- preparative HPLC (50% aq. MeOH) to yield 7 (6.4 mg), 8 (2.0 mg) and 12 (5.0 mg).
3.4 Spectroscopic Data
3.4.1 Trypostatin C (1)
Yellow amorphous powder; [α] 20D −24.5 (c 0.27, CHCl3); UV (MeOH) λmax (log ε): 202 (4.44), 219 (4.13), 398 (3.86) nm; 1H and 13C NMR data see Table 1; ESI–MS (positive ion mode): m/z 404 [M+Na]+; HRESIMS (positive ion mode): m/z 404.1586 [M+Na]+, (calcd. for C20H37NO6Na, 404.1586).
3.4.2 Trypostatin D (2)
Yellow amorphous powder; [α] 20D −61.7 (c 0.13, CHCl3); UV (MeOH) λmax (log ε): 201 (4.37), 217(4.18), 400 (3.94) nm; 1H and 13C NMR data see Table 1; ESI–MS (positive ion mode): m/z 404 [M+Na]+; HRESIMS (positive ion mode): m/z 404.1581 [M+Na]+, (calcd. for C22H39NO6Na, 404.1586).
3.5 Cytotoxicity Assay
Five human cancer cell lines, myeloid leukemia HL-60, hepatocellular carcinoma SMMC7721, lung cancer A-549 cells, breast cancer MCF-7, and colon cancer SW480, were used in the cytotoxic assay. All the cells were cultured in RPMI-1640 or DMEM medium (Hyclone, USA), supplemented with 10% fetal bovine serum (Hyclone, USA).The cytotoxicassay was performed according to the MTS method in 96-well microplates. Briefly, adherent cells (100 μL) was seeded into each well of 96-well cell culture plates and allowed to adhere for 24 h before drug addition, while suspended cells were seeded just before drug addition, each tumor cell line was exposed to the test compound dissolved in DMSO in triplicates for 48 h at 37 °C, with DDP and taxol (Sigma, USA) as positive controls. After the incubation, 20 μL MTS and 100 μL medium was added to each well after removal of 100 μL medium, and the incubation continued for 4 h at 37 °C. The optical density was measured at 492 nm using a Multiskan FC plate reader (Thermo Scientific, USA).
4 Conclusion
In this study, 12 indole diketopiperazines 1–12 were identified for the first time from A. penicilliodes. Among them, 1 and 2 are new compounds, while 3 and 4 are new natural products. So far, the natural diketopiperazines with anti-cancer activities were mainly isolated from Aspergillus and Penicillium genera, with prenylated side chain as the most representative structural fragment, accounting for the majority of this class of compounds. This is the first time to have obtained indole diketo-piperazines featuring with a methyl vinyl ketone side chain at C-2.
References
M.P. Carvalho, W.R. Abraham, Curr. Med. Chem. 19, 3564–3577 (2012)
H. Kohn, W. Widger, Curr. Drug Targets Infect. Disord. 5, 273–295 (2005)
R. Musetti, R. Polizzotto, A. Vecchione, Micron. 38, 643–650 (2007)
K. Strom, J. Sjogren, A. Broberg, Appl. Environ. Microbiol. 68, 4322–4327 (2002)
P.L. Rodriguez, L. Carrasco, J. Virol. 6, 1971–1976 (1992)
A. Loevezijn, J.D. Allen, A.H. Schinkel, G.J. Koomen, Bioorg. Med. Chem. Lett. 11, 29–32 (2001)
H. Kanzaki, S. Yanagisawa, T. Nitoda, Biosci. Biotechnol. Biochem. 68, 2341–2345 (2004)
P. Waring, J. Beaver, Gen. Pharmacol. 27, 1311–1316 (1996)
C. Cornacchia, I. Cacciatore, L. Baldassarre, Mini-Rev. Med. Chem. 12, 2–12 (2012)
M.K. Song, I.K. Hwang, Exp. Biol. Med. 228, 1338–1345 (2003)
Q.X. Li, X.F. Shi, Z. Huang, X.P. Tian, F.Z. Wang, J. Trop. Oceanogr. 32, 35–47 (2013)
K. Kanoh, S. Kohno, T. Asari, T. Harada, J. Katada, M. Muramatsu, H. Kawashima, H. Sekiya, I. Uno, Bioorg. Med. Chem. Lett. 7, 2847–2852 (1997)
K.K. Chen, H.T. Zhu, D. Wang, Y.J. Zhang, C.R. Yang, Acta Bot. Yunnanica 28, 123–126 (2006)
C.B. Cui, H. Kakeya, G. Okada, R. Onose, H. Osada, J. Antibiot. 49, 527–533 (1996)
C.B. Cui, H. Kakeya, H. Osada, J. Antibiot. 49, 534–540 (1996)
S.P. Luo, Q.L. Peng, C.P. Xu, A.E. Wang, P.Q. Huang, Chin. J. Chem. 32, 757–770 (2014)
Z.Y. Mao, H. Geng, T.T. Zhang, Y.P. Ruan, J.L. Ye, P.Q. Huang, Org. Chem. Front. 3, 24–37 (2016)
R.H. Jiao, S. Xu, J.Y. Liu, H.M. Ge, H. Ding, C. Xu, H.L. Zhu, R.X. Tan, Org. Lett. 8, 5709–5712 (2006)
Q.L. Peng, S.P. Luo, X.E. Xia, L.X. Liu, P.Q. Huang, Chem. Commun. 50, 1986–1988 (2014)
A.J. Birch, R.A. Russell, Tetrahedron 28, 2999–3008 (1972)
C.B. Cui, H. Kakeya, H. Osada, Tetrahedron. 52, 12651–12666 (1996)
J. Fayos, D. Lokensgard, J. Clardy, R.J. Cole, J.W. Kirksey, J. Am. Chem. Soc. 96, 6785–6787 (1974)
W.R. Abraham, H.A. Arfmann, Phytochemistry. 29, 1025–1026 (1990)
C.B. Cui, H. Kakeya, H. Osada, Tetrahedron. 53, 59–72 (1997)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21672223). The authors are grateful to the staffs of the analytical and bioactivity screening groups at State Key Laboratory of Phytochemistry and Plant Resources in West China, KIB, CAS, for measuring the spectroscopic data and bioactivities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13659_2018_156_MOESM1_ESM.docx
1D and 2D NMR, ESIMS, HRESIMS, and UV spectra of 1–2, and X-ray molecular structure of 5 are available as Supporting Information (SI). Supplementary material 1 (DOCX 80171 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, H., Zhu, HT., Wang, D. et al. Two New Indolyl Diketopiperazines, Trypostatins C and D from Aspergillus penicilliodes Speg.. Nat. Prod. Bioprospect. 8, 107–111 (2018). https://doi.org/10.1007/s13659-018-0156-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13659-018-0156-z