Abstract
Key message
In the context of increasing droughts related to climate change, our results showed that heavy thinning and/or very low initial planting density can increase CO 2 assimilation rate in needles, and may be used as a short-term management strategy for loblolly pine plantation across sites prone to drought.
Context
The dry summer of 2013 provided us an opportunity to understand the CO2 assimilation rate and stomatal conductance after density manipulation treatments using the dual isotope (δ13C and δ18O) signatures in needles of planted loblolly pine (Pinus taeda L.) trees in the southeastern USA.
Aims
To our knowledge, this is the first study using the dual isotope approach to examine the physiological response of loblolly pine trees, one of the most widely planted tree species in the world, to stand density manipulation treatments (i.e., thinning intensity and planting density).
Methods
In 2001–2003, trees were planted with five different planting densities, 494, 1111, 1729, 2346, and 2964 trees ha−1 at three sites. In 2009–2011, two thinning treatments (none and moderate thinning) were applied in the 1111 trees ha−1 plots, whereas three treatments (none, light and heavy thinning) were applied in the 1729 trees ha−1 plots. Response variables (specific leaf area (SLA), foliar N, δ13C and δ18O) were measured in February 2014.
Results
SLA was lower, while δ18O was higher in the 494 trees ha−1 plots than the 2964 trees ha−1 plots without thinning. In plots planted to 1729 trees ha−1 SLA was lower, while δ13C and δ18O were higher following heavy thinning than in the unthinned control. These responses plus increased crown length, DBH, and height following heavy thinning may reflect an increased tree-level CO2 assimilation rate.
Conclusion
Our results showed that heavy thinning and/or very low initial planting density can be used as a short-term management strategy for loblolly pine plantation across sites prone to drought.
Similar content being viewed by others
1 Introduction
Drought has been widely reported as a major causal factor of reduced forest productivity (Allen et al. 2010). Forest management options such as manipulating planting density and density reductions with thinning are often proposed to increase drought tolerance (e.g., Sohn et al. 2016). Loblolly pine (Pinus taeda L.) is the most widely planted tree species across the southeastern United States (Brender et al. 1981; Baker and Balmer 1983), with nearly 15 million ha of even-aged plantations and an expected increase in plantation area in the future (Huggett et al. 2013). The natural range of the species stretches across the southeastern United States, which experiences periodic extreme drought conditions (Maxwell and Soulé 2009). These droughts are projected to continue or may even increase in frequency in the future (Seager et al. 2009), and it is still unclear if commercial thinning or other density manipulation strategies are useful silvicultural options to minimize the impacts of increasing drought events expected under climate change.
Tree-level physiological responses to thinning are not consistently discussed in the literature (e.g., Ginn et al. 1991; Sheriff 1996; Tang et al. 1999; McDowell et al. 2003; Sword Sayer et al. 2004; Moreno-Gutiérrez et al. 2011). Thinning has been found effective in decreasing specific leaf area (Giuggiola et al. 2013), decreasing inter-tree competition for above and below ground resources (light, water and nutrients) (Bréda et al. 1995; Selig et al. 2008), increasing foliar N (Di Matteo et al. 2010; Moreno-Gutiérrez et al. 2011), increasing photosynthesis rates (A, CO2 assimilation) (Tang et al. 1999), and decreasing tree mortality (Elkin et al. 2015; Sohn et al. 2016). The increase in A with thinning has been associated with a reduction in tree water stress since less trees are using the available water (Bréda et al. 1995). However, canopy opening associated with thinning also increases soil temperature (Matsushima and Chang 2007), and therefore, may increase soil evaporation (Lévesque et al. 2014).
Other studies show an opposite response with thinning increasing water stress and tree dieback (Lagergren et al. 2008; Brooks and Mitchell 2011; Bose et al. 2014). This is possibly due to abrupt changes in stand microclimate conditions such as increases in within-stand radiation, air flow in the canopy, evaporative demand and wind speed (Proe et al. 2001; McDowell et al. 2006). Theoretically, an increasing evaporative demand may cause partial or complete cavitation of the xylem (Adams et al. 2017), eventually reducing water supply to the leaves, therefore reducing the stomatal aperture (g s ) and could limit the A (Bladon et al. 2007). The inconsistent responses to thinning make it difficult to evaluate the effectiveness of a thinning treatment for improving the performance of trees in drought prone forests (Moreno-Gutiérrez et al. 2011).
The stable carbon isotope ratio (δ13C) is a powerful tool and has successfully been used to evaluate the physiological mechanisms associated with tree growth after commercial thinning (Moreno-Gutiérrez et al. 2011; Giuggiola et al. 2015), which represents the ratio, CO2 concentration in the leaf intercellular space (c i )/CO2 concentration in the atmosphere (c a ) (Farquhar et al. 1989). Therefore, the δ13C can be used to interpret changes in intrinsic water use efficiency, defined as the ratio of net photosynthetic rate (A) and stomatal conductance (gs) (Farquhar et al. 1982; Farquhar et al. 1989). An increasing δ13C could be due to either (i) reduced g s at a constant rate of A or (ii) increased rate of A at a constant g s (Farquhar et al. 1982; Farquhar et al. 1989).
Some researchers have suggested using the oxygen isotope (δ18O) along with δ13C (Scheidegger et al. 2000; Grams et al. 2007), since δ18O is specifically related to isotopic signature of source water, which includes isotopic composition of the soil water and the leaf water enrichment in response to transpiration (Dawson et al. 2002; Roden and Siegwolf 2012). The δ18O of plant tissue is proportional to water vapor pressure in the atmosphere (e a ) and water vapor pressure inside the leaf intercellular space (e i ) (Farquhar and Lloyd 1993; Barbour 2007). This indicates that δ18O has an inverse relationship with g s, when evaporative demand in the environment and source water δ18O remain constant (Farquhar et al. 2007). Therefore, sites with greater evaporative demand may have higher δ18O in plant materials. Since δ18O is unaffected by A, it can distinguish the independent effect of g s and A on δ13C (Farquhar et al. 1982; Farquhar et al. 2007; Roden and Farquhar 2012). However, recent studies reported operational limitations associated with the dual isotope approach, especially the assumption that the environmental influences on source water δ18O, atmospheric vapor δ18O, ambient humidity, and leaf temperature must be remained constant over the monitoring period and must be constant across treatments examined (Roden and Siegwolf 2012; Roden and Farquhar 2012).
Several studies have used simultaneous measurements of δ13C and δ18O to explain tree-level physiological responses to stand density manipulation treatments in drought prone sites in Europe (e.g., Grams et al. 2007; Moreno-Gutiérrez et al. 2011) as well as in North America (e.g., McDowell et al. 2003; Powers et al. 2008), but a study has yet to be conducted in loblolly pine plantations. The knowledge gap is important considering the projection of climate-change associated increased drought frequency and the intensive management of loblolly pine in the southeastern USA.
The objective of this study was to evaluate the effects of planting density and thinning intensity on specific leaf area (SLA), foliar N, foliar δ13C and δ18O in loblolly pine trees in the Upper West Gulf Coastal Plain of southeastern Arkansas and northeastern Louisiana, USA in 10–12 year stands, thinned 2–4 years prior to sampling. We hypothesized that (i) trees in higher density treatments usually receive less light, therefore, SLA increase with the increasing planting density, and decrease with the increasing thinning intensity (Hager and Sterba 1985; Giuggiola et al. 2013), (ii) thinning will increase resource (light, water and nutrients) availability to remaining trees, effectively causing an increase in the rate of A and g s and photosynthesis. Therefore, we expected no difference in foliar δ13C due to the equal changes in g s and A, and δ18O will be lower due to the increased g s in treatments with low plant density (due to thinning or low initial planting density) than treatments with high plant density (McDowell et al. 2003; Sohn et al. 2014), however, g s often has a minor effect on δ18O in needles and the changes in foliar δ18O are primarily driven by transpiration or the evaporative demand in the atmosphere as well as from mixing of the leaf water with xylem water (Kahmen et al. 2008; Roden and Siegwolf 2012; Bögelein et al. 2017), and (iii) low planting density and thinning will increase foliar N content (López-Serrano et al. 2005), and that enriched foliar N will have a positive association with the increased δ13C in low planting density and/or thinning treatment (i.e., an increased foliar N will increase the photosynthesis rate) (Warren et al. 2001; Moreno-Gutiérrez et al. 2011).
2 Methods
2.1 Study area
Samples were collected from three sites in the Upper West Gulf Coastal Plain of southeastern Arkansas and northeastern Louisiana, USA: Drew County, Arkansas (33° 32′ N, 91° 37′ W), Ashley County, Arkansas (33° 15′ N, 91° 46′ W), and Union Parish, Louisiana (33° 0′19, 92° 24′). Soils at the Drew and Ashley sites are classified as the Henry series (Coarse-silty, mixed, active, thermic typic fragiaqualfs), poorly drained, with > 50 cm depth to a restrictive layer. Soils at the Union site are classified as the Sacul series (fine, mixed, active, thermic aquic hapludults), moderately well drained, and < 50 cm depth to a restrictive layer (Web Soil Survey 2014). Elevations are 47, 60, and 67 m above sea level at the Ashley, Drew, and Union sites, respectively. The sites are located in an area of humid subtropical climate, with a mean annual temperature of 17.5 °C and mean annual precipitation of 1358 mm (NOAA: http://www.ncdc.noaa.gov) in Monticello, AR (33° 38′ N 91° 45′ W). July and August are the hottest and driest months of the year, with average maximum temperature of 33.3 °C and average monthly precipitation of 82 mm. Moderate to severe drought conditions were common across the region in the summer (2013) before foliage was sampled for isotope analysis. For example, the total precipitation from July to August of 2013 was 79 mm while the long-term (1980–2014) total precipitation was 168 mm (http://droughtmonitor.unl.edu).
2.2 Experimental design
2.2.1 Planting density
The three study sites (Drew, Ashley and Union) were established in 2001, 2002 and 2003, respectively. At each study site, one replicate of five planting density treatments was installed including 494, 1111, 1729, 2346, and 2964 trees ha−1. At the Drew and Ashley sites, soils were bedded, followed by planting and a broadcast application of sulfometuron methyl herbicide and directed applications of glyphosate herbicide, as needed, to maintain weed-free conditions for the first year. At the Union site, imazapyr and glyphosate herbicides were used to prepare the site, followed by planting and follow-up direct applications of glyphosate to maintain weed free conditions for the first year. All study sites were planted with loblolly pine using the best open-pollinated family for that location as determined by the landowner. Each planting spot was double planted to ensure survival, with one seedling cut in the autumn following planting. Fertilization included 44.8 kg ha−1 N + 56.0 kg ha−1 P during year 1, and 224.1 kg ha−1 N + 22.4 kg ha−1 P during year 10. All three sites were treated for Nantucket pine tip moth (Rhyacionia frustrana) with tebufenozide and permethrin during the first 2 years, spraying each tree monthly between March and October.
2.2.2 Thinning intensity
Additional treatment plots were established at each site to examine the effects of different thinning intensities. Thinning treatments were applied in plots with two planting densities: 1111 and 1729 trees ha−1 (Table 1). The timing of thinning was selected to correspond to when the stands reached a stand density index, a composite density metric that incorporates average tree diameter and number of trees per hectare (i.e., Reineke 1933), of approximately 612. This SDI value corresponds to 55% (the threshold where significant mortality starts to occur from self-thinning) of the maximum stand density index of 1111. The 1111 trees ha−1 plots were thinned to 494 trees ha−1 (55% density reduction), while the 1729 trees ha−1 plots were thinned to either 1111 trees ha−1 (36% density reduction) or 494 trees per ha−1 (71% density reduction). For consistency, we refer to post-thin density reductions of 36, 55, and 71% as “light”, “moderate”, and “heavy” thinning, respectively. The applied thinning removed smaller, less competitive trees and trees with defects such as stem fusiform rust (Cronartium fusiforme) infections, forking, sweep or crook, while also considering tree spacing. At the Drew site, the plots planted to 1729 trees ha−1 were thinned in spring 2009, while the plots planted to 1111 trees ha−1 were thinned in spring 2010. At the Ashley site and Union sites, 1729 trees ha−1 plots were thinned in spring 2010 and the 1111 trees ha−1 plot were thinned in spring 2011 (Table 1).
The study has a total of eight treatments (five planting densities and three thinning intensities), replicated once at each site. The size of treatment plot varied from 0.09-ha for the plots planted to 2964 trees ha−1 to 0.22-ha for the plots planted to 494 trees ha−1.
2.3 Data collection and isotope analysis
Heights and diameters at breast height (dbh) were measured on trees with at least 1.4 m height in early 2013 on the Drew (Age 12) and Union (Age 10) sites and in early 2014 on the Ashley site (age 12). Height was measured to the nearest 0.3 m and diameter to the nearest 0.25 cm. Heights were measured on all trees for the 494 trees ha−1 planting density, every other tree for the 1111 trees ha−1 planting density and every third tree for the 1729, 2346, and 2964 trees ha−1 planting densities. Height to the base of the live crown was measured on all trees that were measured for total height. Tree density per ha and basal area per ha were calculated applying the plot specific per ha expansion factor with surviving trees and plot basal area, respectively.
Foliage samples were collected from three randomly selected trees in each plot in February 2014. We have a total eight plots (each for a treatment) replicated across three sites, which makes a total 72 trees (3 trees*8 treatments/plots*3 sites = 72). Collected samples were sun-exposed leaves located at the top-middle part of the crown. Only the last flush of needles formed in 2013 were sampled. Samples were placed in plastic bags and stored on ice in the field. Once back at the lab, foliage samples were placed in a freezer until processed. One hundred needles from each sample were scanned on a flat-bed scanner with a backlight to estimate projected (one-sided) leaf area with the WinSeedle software (Regent Instruments). The scanned needles were then dried in a convection oven at 65 °C for 72 h and weighed to the nearest 1 mg. These data were used to estimate specific leaf area (projected leaf area per unit of dry mass) by tree. After weighing, dried foliage samples were ground in a Wiley mill to pass through a 20-mesh screen and then pulverized into a fine powder using a sterilized mortar and pestle. Samples for δ13C analysis were weighed to 0.7 mg, placed in tin capsules, while δ18O samples were weighed to 0.3 mg and placed in silver capsules.
Foliage samples were analyzed by the Stable Isotope Facility at the University of Arkansas. δ13C composition and N concentration were analyzed with an elemental analyzer by combusting samples in oxygen at 1100 °C, then passed through oxidation and reduction tubes and peaks were separated on a chromatography column. δ18O was analyzed with a high temperature conversion elemental analyzer. Samples were combusted at 1300 °C in a reducing environment. Isotope ratios were expressed in delta notation (‰; parts per thousand), where the isotopic composition of the bulk leaf material was expressed relative to internationally accepted reference standards (Dawson et al. 2002). The standard for δ13C was PeeDee Belemnite and the standard for δ18O was the Vienna-standard mean ocean water. Area-based foliar N content was calculated by multiplying foliar N concentration by the reciprocal of specific leaf area to obtain units of g m−2. We used the Carlo Erba NC2500 interfaced to a Delta Plus IRMS (Thermoscie Bremen Germany) for the δ13C and N concentration, and TCEA (Thermo Chemolysis Elemental Analyzer) was interface to a Delta Plus XP for the δ18O analysis, with the precisions of 0.15 and 0.30 per mil for δ13C and δ18O, respectively.
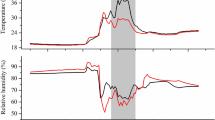
As we do not have direct measurement of source water δ18O of the three study sites, micro-climatic factors driving water availability including average daily temperature (°C), total annual precipitation (mm) and average daily solar radiation (W m−2) were extracted from ~ 1 km gridded rasters of Daymet Earth Data (https://daymet.ornl.gov/), and relative humidity in the air was extracted (%) from PRISM Climate Data (http://oldprismmap.nacse.org/) using coordinates of the three study sites. We extracted these data for the period from 1990 to 2013 (the last growth year prior to sample collection) to test the difference across three sites. The results of the one-way ANOVA showed that the three sites were similar in terms of average daily temperature (°C), total annual precipitation (mm), average daily solar radiation (W m−2) and average growing season (April–October) relative humidity (%) (Fig. 1).
2.4 Statistical analysis
All statistical analysis were performed in R v.3.2.2 (R Development Core Team 2014). Linear mixed effect models (Pinheiro et al. 2014) were used to evaluate the effect of planting density and thinning intensity on the response variables. The response variables were specific leaf area, foliar N content, foliar δ13C and δ18O. The effects of planting density and thinning intensity were analyzed separately as thinning treatments were implemented in only two planting density treatments (1111 stems ha−1 and 1729 stems ha−1). Sites (n = 3) were treated as random factors. Both planting density and thinning intensity were considered as discrete variables each with five levels. The planting density of 2964 trees ha−1 was considered as reference level and compared with four other levels of planting density (494, 1111, 1729 and 2346 trees ha−1). Differences between thinning intensity were tested by means of orthogonal contrasts. Four contrasts were created and executed into the model using the function “contrasts” of R (R Development Core Team 2014). The four contrasts were (1) planting density of 1111 trees ha−1: control vs moderate thinning, (2) planting density of 1729 trees ha−1: control vs light thinning, (3) planting density of 1729 trees ha−1: control vs heavy thinning, and (4) moderate thinning at 1111 trees ha−1 planting density vs heavy thinning at 1729 trees ha−1 planting density. We visually verified the assumptions of normality of residuals and homogeneity of the variances. Mean values ± 95% confidence intervals were presented in the figures. We also tested the linear correlation between foliar δ13C and foliar N content using the linear mixed effect models while incorporating random effects associated with sites.
3 Results
The average dbh and the crown length were negatively related to stand density (by planting/thinning). Thinning had a substantial effect on tree size (dbh and height), average dbh increased by 10, 12, and 32% in the light, moderate, and heavy thinning treatments, respectively. Average tree height was similar across treatments (Table 2). For trees from which needle samples were collected, average tree height was similar across treatments and average dbh was greater for treatments with lower density (Table 3). Planting density significantly affected the SLA and foliar δ18O, but not the foliar N concentration, and foliar δ13C (Table 4). SLA was significantly lower, while foliar δ18O was significantly higher in the planting density of 494 trees ha−1 than in the planting density of 2964 trees ha−1 (Table 4, and Fig. 2A and B).
Effect of planting density on specific leaf area and δ18O in loblolly pine (Pinus taeda L.) trees. Note. Error bar represents mean ± 95% confidence interval. Non-significant results (Table 3) are not presented. In both figures, 2964 trees ha−1 stand density treatment was considered as the reference in the analysis, and was significantly different from 494 trees ha−1, and differences with other density treatments were non-significant
Thinning intensity significantly affected the SLA, foliar δ13C, and δ18O, but not the foliar N content (Table 4). In the initial planting density treatment of 1729 trees ha−1, SLA was significantly lower, while foliar δ13C and δ18O were significantly higher in heavy thinning (71% density removal) than in the unthinned treatment (Table 4, Fig. 3A, B and C). Moderate thinning (55% density removal) at 1111 trees ha−1 planting density and light thinning (36% removal) at 1729 trees ha−1 planting density exhibited no difference (Table 4). Foliar N content had a positive linear association with foliar δ13C (R2 = 0.24, p = 0.01) (Fig. 4).
Effect of thinning treatments on specific leaf area, foliar δ13C and δ18O in loblolly pine (Pinus taeda L.) trees. Note. Error bar represents mean ± 95% confidence interval, PD = planting density, moderate = 55% density removal, heavy = 71% density removal, and light = 36% density removal. Non-significant results (Table 3) are not presented. C1, C2, C3 and C4 are four contrasts used in the analysis, gray and black represent significant and non-significant differences, respectively
4 Discussion
The results of our study indicated that both approaches to reduce stand density, the reduced planting density and increased thinning intensities stimulated growth (i.e., increased crown length, dbh, and height) in residual loblolly pine, increased δ18O in needles, and slightly increased δ13C in needles. The increased tree growth and increased δ13C and δ18O in needles may suggest an increased tree-level CO2 assimilation rate with a reduced or unchanged stomatal conductance (Scheidegger et al. 2000). The increased δ18O in needles is probably due to 18O enriched source water (Giuggiola et al. 2015), which resulted from an increased evaporative demand through increasing penetration of solar radiation, increasing temperature and wind speed after heavy thinning (Farquhar et al. 2007).
Despite the important findings that heavy thinning increased tree-level δ13C, the analysis has some key limitations. There was no direct measurement of xylem or soil water δ18O and/or gas exchange, which is necessary to assure that pre-evaporated water was not enriched in δ18O (Roden and Siegwolf 2012). The needle temperature may vary across treatments if the treatments are associated with different solar radiation input and humidity. The differences in needle temperature might affect the outcomes of δ18O ratio. In addition, leaf boundary layer may be changed by the very different planting densities and thinning intensities, and may also affect the isotope ratios (Roden and Siegwolf 2012; Gessler et al. 2014). Light and soil water availability were not directly measured, which are important variables driving photosynthesis and transpiration. A final limitation is that the time since thinning varies from 2 to 4 years across three study sites, which may contribute to unexplained variance by our model.
The three study sites were similar in terms of major micro-climatic factors (temperature, precipitation, relative humidity and solar radiation) that potentially influence water availability and relative humidity (Fig. 1), and the distances among treatment plots within a site were minimal. Roden and Siegwolf (2012) demonstrated two prerequisites of the dual isotope approach including (i) the mean relative air humidity during the growing season should not be high (< 80%), and (ii) only isohydric tree species with tight stomatal control to changes in vapor pressure deficit should be studied. The average growing season (April–October) relative humidity of our three study sites ranged from 63.8–64.1%, while loblolly pine is a drought avoider species with strong stomatal control (Teskey et al. 1987; Tang et al. 1999; Domec et al. 2009). Therefore, these two assumptions of the dual isotope approach were met.
4.1 Specific leaf area
We hypothesized that specific leaf area (SLA) would decrease with increasing thinning intensity and decreasing planting density. However, only heavy thinning (71% density removal) relative to the unthinned—1729 trees ha−1, and only the lowest planting density (494 trees ha−1) relative to 2964 trees ha−1 exhibited reduced SLA. Morphologically, leaves with lower SLA have greater mass for a given unit of leaf area (Wilson et al. 1999). Maximum photosynthetic rates decrease as SLA decreases across multiple plant life forms (Reich et al. 1998), but the decrease in photosynthesis at the leaf level may be offset by the increase in total tree leaf area associated with lower stand density (Shelburne et al. 1993), and thus total tree photosynthesis may increase. Although tree leaf area was not measured in this study, the heavy thinning and the low planting density treatment (494 trees ha−1) increased crown length (Table 2), suggesting increased light availability to individual tree crowns and greater surface area for light interception.
4.2 Foliar δ13C and δ18O
An elevated δ13C in needles can result from either an increase in CO2 assimilation rate at a constant stomatal conductance or from a decrease in stomatal conductance with a constant CO2 assimilation rate (Scheidegger et al. 2000). Our results showed that the average crown length and average dbh were higher in trees of low planting density 494 trees ha−1 relative to trees of higher planting densities, and trees of heavy thinning treatment than trees of the unthinned—1729 trees ha−1 controls (Table 2). A larger crown length and a larger tree size may indicate an increased CO2 assimilation rate through increased photosynthesis by trees of 494 trees ha−1 planting density than higher planting densities, and by trees of heavy thinning than trees of unthinned—1729 trees ha−1 controls. Therefore, the heavy thinning treatment probably increased the light availability for residual trees, and concomitantly increased the rate of photosynthesis (may be indicated by an increased δ13C in needles).
The increased δ18O in needles might be due to an increased soil evaporation (Roden and Siegwolf 2012; Gessler et al. 2014), which was also reported by Giuggiola et al. (2015), while working with a heavy thinning treatment (similar to the heavy thinning treatment of our study) in xeric sites of Switzerland. The higher δ18O in the heavy thinned treatment could be due to an increased atmospheric evaporative demand by allowing greater penetration of solar radiation and wind into the stands. An increased evaporative demand after heavy thinning has also been reported for Pinus nigra in central Spain (Martín-Benito et al. 2010), Pseudotsuga menziesii in Vancouver Island, Canada (Brooks and Mitchell 2011), and Populus tremuloides in boreal Canada (Bladon et al. 2007). In drought prone sites, stand microclimate can be drier and warmer (e.g., Rambo and North 2009; Ma et al. 2010), which may cause a higher air and soil temperature and a lower air humidity. This warming increases water evaporation from soil, and results in 18O enrichment of soil water as 16O diffuses more than the 18O isotope (Farquhar et al. 2007; Sarris et al. 2013). Therefore, 18O enriched source water may result in 18O enriched xylem water of trees, leading to a concomitant increase of foliar δ18O (18O/16O).
On the other hand, if heavy thinning increases soil water availability, trees may have a greater accessibility to shallower water sources that are relatively more 18O enriched (Ehleringer et al. 1993; Giuggiola et al. 2015). Therefore, similar to the previous explanation, 18O enriched source water will result in an elevated foliar δ18O. Therefore, the increased foliar δ18O in the heavy thinning treatment could be due to an increased evaporative demand, or to an increased availability of shallow water, or a combination of them (Farquhar et al. 2007; Roden and Siegwolf 2012; Gessler et al. 2014).
The variable physiological responses to density manipulation treatments have been widely reported (see the review of Giuggiola et al. 2015), and a range of tree-, species-, and site-level factors modulate the magnitude and the pattern of responses. For example, McDowell et al. (2003) reported a reduced δ13C due to an elevated stomatal conductance relative to CO2 assimilation rate in Pinus ponderosa stands after thinning. In contrast, while working on Pinus radiata in a thinning experiment, Warren et al. (2001) reported an increased δ13C due to an increased CO2 assimilation rate that resulted from an elevated foliar N. Other studies reported no significant effect of thinning on residual tree δ13C (e.g., Sala et al. 2005; McDowell et al. 2006).
4.3 Foliar N concentration and δ13C
A strong relationship between leaf N content and net photosynthetic capacity has been widely reported (e.g., Reich et al. 1995; Wright et al. 2004). We found a positive linear relationship (although weak) between foliar δ13C and foliar N content, suggesting CO2 assimilation rate during photosynthesis was affected by foliar N content, and that was independent of density manipulation treatment types. Warren et al. (2001) reported that a greater foliar N content generally results in a greater net CO2 assimilation rate during photosynthesis, and a corresponding greater δ13C or intrinsic water use efficiency (WUEi). We did not detect a statistically significant difference among treatments in terms of foliar N content, therefore, greater δ13C/WUEi in heavy thinning relative to unthinned control was probably not due to foliar N content.
5 Management implications
Initial planting density and thinning intensity are important silvicultural tools for forest landowners managing loblolly pine across the southeastern US. Preferred planting density and/or final density after thinning is derived from a combination of economic and biologic factors, but little information is available on the physiology and in particular water-use efficiency effects in this key management decision. Our results showed that the heavy thinning (71% of total density) treatment resulted in a higher δ13C relative to the unthinned—1729 trees ha−1 control. This outcome may indicate an increased photosynthesis, which we hypothesized and is widely reported in the literature (e.g., McDowell et al. 2003; Giuggiola et al. 2015). Therefore, heavy thinning and/or very low initial planting density can be used as a short-term management strategy to increase drought resistance for loblolly pine plantation across sites prone to drought.
References
Adams HD, Zeppel MJB, Anderegg WRL, Hartmann H, Landhäusser SM, Tissue DT, Huxman TE, Hudson PJ, Franz TE, Allen CD, Anderegg LDL, Barron-Gafford GA, Beerling DJ, Breshears DD, Brodribb TJ, Bugmann H, Cobb RC, Collins AD, Dickman LT, Duan H, Ewers BE, Galiano L, Galvez DA, Garcia-Forner N, Gaylord ML, Germino MJ, Gessler A, Hacke UG, Hakamada R, Hector A, Jenkins MW, Kane JM, Kolb TE, Law DJ, Lewis JD, Limousin J-M, Love DM, Macalady AK, Martínez-Vilalta J, Mencuccini M, Mitchell PJ, Muss JD, O’Brien MJ, O’Grady AP, Pangle RE, Pinkard EA, Piper FI, Plaut JA, Pockman WT, Quirk J, Reinhardt K, Ripullone F, Ryan MG, Sala A, Sevanto S, Sperry JS, Vargas R, Vennetier M, Way DA, Xu C, Yepez EA, McDowell NG (2017) A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nat. Ecol Evol 1(9):1285–1291. https://doi.org/10.1038/s41559-017-0248-x
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EH, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim J-H, Allard G, Running SW, Semerci A, Cobb N (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259(4):660–684. https://doi.org/10.1016/j.foreco.2009.09.001
Baker JB, WE Balmer (1983) Loblolly pine. Agriculture Handbook, Ed by Burns, RM USDA, Forest Service Washington, DC, USA:148
Barbour MM (2007) Stable oxygen isotope composition of plant tissue: a review. Funct Plant Biol 34(2):83–94. https://doi.org/10.1071/FP06228
Bladon KD, Silins U, Landhäusser SM, Messier C, Lieffers VJ (2007) Carbon isotope discrimination and water stress in trembling aspen following variable retention harvesting. Tree Physiol 27(7):1065–1071. https://doi.org/10.1093/treephys/27.7.1065
Bögelein R, Thomas FM, Kahmen A (2017) Leaf water 18O and 2H enrichment along vertical canopy profiles in a broadleaved and a conifer forest tree. Plant Cell Environ 40(7):1086–1103. https://doi.org/10.1111/pce.12895
Bose AK, AS Nelson, M Kane, A Rigling (2017) Carbon and oxygen isotope data in needles of loblolly pine. [dataset] DOI: https://doi.org/10.6084/m9.figshare.5663362.v2, URL: https://figshare.com/articles/Loblolly_pine_foliar_carbon_and_oxygen_data/5663362
Bose AK, Harvey BD, Brais S, Beaudet M, Leduc A (2014) Constraints to partial cutting in the boreal forest of Canada in the context of natural disturbance-based management: a review. Forestry 87(1):11–28. https://doi.org/10.1093/forestry/cpt047
Bréda N, Granier A, Aussenac G (1995) Effects of thinning on soil and tree water relations, transpiration and growth in an oak forest (Quercus petraea (Matt.) Liebl.) Tree Physiol 15(5):295–306. https://doi.org/10.1093/treephys/15.5.295
Brender EV, RP Belanger, BF Malac (1981) Loblolly pine. In: Choices in silviculture for American forests Washington, DC:Society of American Foresters
Brooks JR, Mitchell AK (2011) Interpreting tree responses to thinning and fertilization using tree-ring stable isotopes. New Phytol 190(3):770–782. https://doi.org/10.1111/j.1469-8137.2010.03627.x
Dawson TE, S Mambelli, AH Plamboeck, PH Templer, KP Tu (2002) Stable isotopes in plant ecology. Annual review of ecology and systematics:507–559
Di Matteo G, De Angelis P, Brugnoli E, Cherubini P, Scarascia-Mugnozza G (2010) Tree-ring Δ13C reveals the impact of past forest management on water-use efficiency in a Mediterranean oak coppice in Tuscany (Italy). Ann For Sci 67(5):510. https://doi.org/10.1051/forest/2010012
Domec J-C, Palmroth S, Ward E, Maier CA, Thérézien M, Oren R (2009) Acclimation of leaf hydraulic conductance and stomatal conductance of Pinus taeda (loblolly pine) to long-term growth in elevated CO2 (free-air CO2 enrichment) and N-fertilization. Plant Cell Environ 32(11):1500–1512. https://doi.org/10.1111/j.1365-3040.2009.02014.x
Ehleringer JR, Hall AE, Farquhar GD (1993) Water use in relation to productivity. In: Ehleringer JR, Hall AE, Farquhar GD (eds) In: Stable isotopes and plant carbon–water relations. Academic Press, New York, pp 3–8
Elkin C, Giuggiola A, Rigling A, Bugmann H (2015) Short- and long-term efficacy of forest thinning to mitigate drought impacts in mountain forests in the European Alps. Ecol Appl 25(4):1083–1098. https://doi.org/10.1890/14-0690.1
Farquhar GD, Cernusak LA, Barnes B (2007) Heavy water fractionation during transpiration. Plant Physiol 143(1):11–18. https://doi.org/10.1104/pp.106.093278
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Biol 40(1):503–537. https://doi.org/10.1146/annurev.pp.40.060189.002443
Farquhar GD, Lloyd J (1993) Carbon and oxygen isotope effects in the exchange of carbon dioxide between terrestrial plants and the atmosphere. In: Ehleringer JR, Hall AE, Farquhar GD (eds) Stable isotopes and plant carbon/water relations. Academic Press, New York, pp 49–79
Farquhar GD, O'leary M, Berry J (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Funct Plant Biol 9:121–137
Gessler A, Ferrio JP, Hommel R, Treydte K, Werner RA, Monson RK (2014) Stable isotopes in tree rings: towards a mechanistic understanding of isotope fractionation and mixing processes from the leaves to the wood. Tree Physiol 34(8):796–818. https://doi.org/10.1093/treephys/tpu040
Ginn SE, Seiler JR, Cazell BH, Kreh RE (1991) Physiological and growth responses of eight-year-old loblolly pine stands to thinning. For Sci 37:1030–1040
Giuggiola A, Bugmann H, Zingg A, Dobbertin M, Rigling A (2013) Reduction of stand density increases drought resistance in xeric Scots pine forests. For Ecol Manag 310:827–835. https://doi.org/10.1016/j.foreco.2013.09.030
Giuggiola A, Ogée J, Rigling A, Gessler A, Bugmann H, Treydte K (2015) Improvement of water and light availability after thinning at a xeric site: which matters more? A dual isotope approach. New Phytol 210:108–121
Grams TEE, Kozovits AR, Haberle K-H, Matyssek R, Dawson TE (2007) Combining δ13C and δ18O analyses to unravel competition, CO2 and O3 effects on the physiological performance of different-aged trees. Plant Cell Environ 30(8):1023–1034. https://doi.org/10.1111/j.1365-3040.2007.01696.x
Hager H, Sterba H (1985) Specific leaf area and needle weight of Norway spruce (Picea abies) in stands of different densities. Can J For Res 15(2):389–392. https://doi.org/10.1139/x85-062
Huggett R, DN Wear, R Li, J Coulston, S Liu (2013) Forecasts of forest conditions. Southern Forest Futures Project: Technical Report
Kahmen A, Simonin K, Tu KP, Merchant A, Callister A, Siegwolf R, Dawson TE, Arndt SK (2008) Effects of environmental parameters, leaf physiological properties and leaf water relations on leaf water δ18O enrichment in different Eucalyptus species. Plant Cell Environ 31(6):738–751. https://doi.org/10.1111/j.1365-3040.2008.01784.x
Lagergren F, Lankreijer H, Kucera J, Cienciala E, Mölder M, Lindroth A (2008) Thinning effects on pine-spruce forest transpiration in central Sweden. For Ecol Manag 255(7):2312–2323. https://doi.org/10.1016/j.foreco.2007.12.047
Lévesque M, Siegwolf R, Saurer M, Eilmann B, Rigling A (2014) Increased water-use efficiency does not lead to enhanced tree growth under xeric and mesic conditions. New Phytol 203(1):94–109. https://doi.org/10.1111/nph.12772
López-Serrano FR, de las Heras J, González-Ochoa AI, García-Morote FA (2005) Effects of silvicultural treatments and seasonal patterns on foliar nutrients in young post-fire Pinus halepensis forest stands. For Ecol Manag 210(1-3):321–336. https://doi.org/10.1016/j.foreco.2005.02.042
Ma S, Concilio A, Oakley B, North M, Chen J (2010) Spatial variability in microclimate in a mixed-conifer forest before and after thinning and burning treatments. For Ecol Manag 259(5):904–915. https://doi.org/10.1016/j.foreco.2009.11.030
Martín-Benito D, Del Río M, Heinrich I, Helle G, Cañellas I (2010) Response of climate-growth relationships and water use efficiency to thinning in a Pinus nigra afforestation. For Ecol Manag 259(5):967–975. https://doi.org/10.1016/j.foreco.2009.12.001
Matsushima M, Chang SX (2007) Effects of understory removal, N fertilization, and litter layer removal on soil N cycling in a 13-year-old white spruce plantation infested with Canada bluejoint grass. Plant Soil 292(1-2):243–258. https://doi.org/10.1007/s11104-007-9220-x
Maxwell JT, PT Soulé (2009) United States drought of 2007: historical perspectives. Climate research (Open Access for articles 4 years old and older) 38:95
McDowell N, Brooks J, Fitzgerald S, Bond B (2003) Carbon isotope discrimination and growth response of old Pinus ponderosa trees to stand density reductions. Plant Cell Environ 26(4):631–644. https://doi.org/10.1046/j.1365-3040.2003.00999.x
McDowell NG, Adams HD, Bailey JD, Hess M, Kolb TE (2006) Homeostatic maintenance of ponderosa pine gas exchange in response to stand density changes. Ecol Appl 16(3):1164–1182. https://doi.org/10.1890/1051-0761(2006)016[1164:HMOPPG]2.0.CO;2
Moreno-Gutiérrez C, Barbera GG, Nicolas E, De Luis M, Castillo VM, Martinez-Fernandez F, Querejeta JI (2011) Leaf δ18O of remaining trees is affected by thinning intensity in a semiarid pine forest. Plant Cell Environ 34(6):1009–1019. https://doi.org/10.1111/j.1365-3040.2011.02300.x
Pinheiro J, D Bates, S DebRoy, D Sarkar (2014) nlme: linear and nonlinear mixed effects models. R package version 3.1–117. Available at https://cran.rproject.org/web/packages/nlme/index.html
Powers MD, Pregitzer KS, Palik BJ (2008) δ13C and δ18O trends across overstory environments in whole foliage and cellulose of three Pinus species. J Am Soc Mass Spectrom 19(9):1330–1335. https://doi.org/10.1016/j.jasms.2008.06.010
Proe M, Griffiths J, McKay H (2001) Effect of whole-tree harvesting on microclimate during establishment of second rotation forestry. Agric For Meteorol 110(2):141–154. https://doi.org/10.1016/S0168-1923(01)00285-4
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at www.r-project.org
Rambo TR, North MP (2009) Canopy microclimate response to pattern and density of thinning in a Sierra Nevada forest. For Ecol Manag 257(2):435–442. https://doi.org/10.1016/j.foreco.2008.09.029
Reich P, Ellsworth D, Walters M (1998) Leaf structure (specific leaf area) modulates photosynthesis–nitrogen relations: evidence from within and across species and functional groups. Funct Ecol 12(6):948–958. https://doi.org/10.1046/j.1365-2435.1998.00274.x
Reich P, Walters M, Kloeppel B, Ellsworth D (1995) Different photosynthesis-nitrogen relations in deciduous hardwood and evergreen coniferous tree species. Oecologia 104(1):24–30. https://doi.org/10.1007/BF00365558
Reineke LH (1933) Perfecting a stand-density index for even-aged stands. J Agric Res 46:627–638
Roden J, Siegwolf R (2012) Is the dual-isotope conceptual model fully operational? Tree Physiol 32(10):1179–1182. https://doi.org/10.1093/treephys/tps099
Roden JS, Farquhar GD (2012) A controlled test of the dual-isotope approach for the interpretation of stable carbon and oxygen isotope ratio variation in tree rings. Tree Physiol 32(4):490–503. https://doi.org/10.1093/treephys/tps019
Sala A, Peters GD, McIntyre LR, Harrington MG (2005) Physiological responses of ponderosa pine in western Montana to thinning, prescribed fire and burning season. Tree Physiol 25(3):339–348. https://doi.org/10.1093/treephys/25.3.339
Sarris D, Siegwolf R, Körner C (2013) Inter-and intra-annual stable carbon and oxygen isotope signals in response to drought in Mediterranean pines. Agric For Meteorol 168:59–68. https://doi.org/10.1016/j.agrformet.2012.08.007
Scheidegger Y, Saurer M, Bahn M, Siegwolf R (2000) Linking stable oxygen and carbon isotopes with stomatal conductance and photosynthetic capacity: a conceptual model. Oecologia 125(3):350–357. https://doi.org/10.1007/s004420000466
Seager R, Tzanova A, Nakamura J (2009) Drought in the southeastern United States: causes, variability over the last millennium, and the potential for future hydroclimate change. J Clim 22(19):5021–5045. https://doi.org/10.1175/2009JCLI2683.1
Selig MF, Seiler JR, Tyree MC (2008) Soil carbon and CO2 efflux as influenced by the thinning of loblolly pine (Pinus taeda L.) plantations on the piedmont of Virginia. For Sci 54:58–66
Shelburne V, Hedden R, Allen R (1993) The effect of site, stand density, and sapwood permeability on the relationship between leaf area and sapwood area in loblolly pine (Pinus taeda L.) For Ecol Manag 58(3-4):193–209. https://doi.org/10.1016/0378-1127(93)90145-D
Sheriff DW (1996) Responses of carbon gain and growth of Pinus radiata stands to thinning and fertilizing. Tree Physiol 16(6):527–536. https://doi.org/10.1093/treephys/16.6.527
Sohn JA, Brooks JR, Bauhus J, Kohler M, Kolb TE, McDowell NG (2014) Unthinned slow-growing ponderosa pine (Pinus ponderosa) trees contain muted isotopic signals in tree rings as compared to thinned trees. Trees 28(4):1035–1051. https://doi.org/10.1007/s00468-014-1016-z
Sohn JA, Saha S, Bauhus J (2016) Potential of forest thinning to mitigate drought stress: a meta-analysis. For Ecol Manag 380:261–273. https://doi.org/10.1016/j.foreco.2016.07.046
Sword Sayer MA, Goelz JCG, Chambers JL, Tang Z, Dean TJ, Haywood JD, Leduc DJ (2004) Long-term trends in loblolly pine productivity and stand characteristics in response to thinning and fertilization in the West Gulf region. For Ecol Manag 192(1):71–96. https://doi.org/10.1016/j.foreco.2004.01.006
Tang Z, Chambers JL, Guddanti S, Barmett JP (1999) Thinning, fertilization, and crown position interact to control physiological responses of loblolly pine. Tree Physiol 19(2):87–94. https://doi.org/10.1093/treephys/19.2.87
Teskey R, Bongarten B, Cregg B, Dougherty P, Hennessey T (1987) Physiology and genetics of tree growth response to moisture and temperature stress: an examination of the characteristics of loblolly pine (Pinus taeda L.) Tree Physiol 3(1):41–61. https://doi.org/10.1093/treephys/3.1.41
Warren CR, McGrath JF, Adams MA (2001) Water availability and carbon isotope discrimination in conifers. Oecologia 127(4):476–486. https://doi.org/10.1007/s004420000609
Web Soil Survey, 2014. https://websoilsurvey.sc.egov.usda.gov/App/HomePage.htm, Accessed December 2014
Wilson PJ, Thompson K, Hodgson JG (1999) Specific leaf area and leaf dry matter content as alternative predictors of plant strategies. New Phytol 143(1):155–162. https://doi.org/10.1046/j.1469-8137.1999.00427.x
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavender-Bares J, Chapin T, Cornelissen JH, Diemer M (2004) The worldwide leaf economics spectrum. Nature 428(6985):821–827. https://doi.org/10.1038/nature02403
Acknowledgements
We are greatly appreciative to the editor and the two reviewers of the Annals of Forest Science for numerous helpful suggestions. The first author acknowledges the funding received through the National Science Foundation Center for Advanced Forestry Systems and the University of Maine. We thank the Plantation Management Research Cooperative at the University of Georgia for treatment implementation and tree measurements. Funding for sample collected and isotope analysis was provided by the University of Arkansas, Monticello to Andrew Nelson. The first author thanks Drs. Matthias Saurer and Marco Lehmann of WSL Swiss Federal Institute for Forest, Snow and Landscape Research for sharing a discussion on interpretation of the stable isotope approach.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Data accessibility
A data file named ‘Carbon and oxygen isotope data in needles of loblolly pine’ and an instruction file are available from figshare Digital Data Repository (https://figshare.com/articles/Loblolly_pine_foliar_carbon_and_oxygen_data/5663362) (Bose et al. 2017).
Additional information
Handling Editor: Erwin Dreyer
Contribution of the co-authors
A.K.B. analyzed the data, and led the writing of the manuscript, A.S.N. and M.K. designed the experiment, collected the data, and contributed in writing, and A.R. contributed in interpretation of the dual isotope approach and writing of the manuscript.
Rights and permissions
About this article
Cite this article
Bose, A.K., Nelson, A.S., Kane, M. et al. Density reduction in loblolly pine (Pinus taeda L.) stands to increase tree C assimilation: an approach with the dual δ13C and δ18O isotope signatures in needles. Annals of Forest Science 75, 8 (2018). https://doi.org/10.1007/s13595-017-0687-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13595-017-0687-1