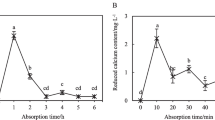
Abstract
To reveal the molecular mechanism in the accumulation of chlorogenic acids (CGAs) in peach (Prunus persica) fruit during growth and development, CGA contents in the flesh of the three peach cultivars ‘Ruiguang 18’, ‘Heiyoutao’, and ‘Beijingyixianhong’ were determined. The expression levels of CGA metabolism-related genes were analyzed based on transcriptome data (RNA-seq). These candidate genes were then screened and real-time fluorescent quantitative PCR (qRT-PCR) was performed to verify their expression. The results showed that the content of total CGAs, 5-O-caffeoylquinic acid and 3-O-caffeoylquinic acid, in the flesh of ‘Ruiguang 18’ exhibited a decreasing trend during fruit development, and there was a great drop at maturity stage (P < 0.05). The three contents in ‘Heiyoutao’ increased first at stage S2 (P < 0.05) and then decreased significantly (P < 0.05). In ‘Beijingyixianhong’, they stayed stable in the early stages, then total CGAs and 3-O-caffeoylquinic acid decreased significantly at the maturity stage (P < 0.05). RNA-seq transcriptome data analysis and qRT-PCR expression analysis showed that the accumulation of CGAs in fruit flesh was mainly affected by the expression of Prupe.3G100800 (PpHCT) and Prupe.3G107300 (Pp4CL), and their expression levels were highly consistent with total CGA content. Thus, we concluded that Prupe.3G100800 (PpHCT) and Prupe.3G107300 (Pp4CL) are the key genes for CGAs synthesis in peach flesh.





Similar content being viewed by others
References
Abdelghafar A, Burrell R, Reighard G, Gasic K (2018) Antioxidant capacity and bioactive compounds accumulation in peach breeding germplasm. J Am Pomol Soc 72:40–69
Awad MA, Jager PK (2001) Flavonoid and chlorogenic acid changes in skin of 'elstar' and 'jonagold' apples during development and ripening. Sci Hortic 90:69–83
Chen ZX, Liu GH, Liu YQ, Xian ZQ, Tang N (2017) Overexpression of the LmHQT1 gene increases chlorogenic acid production in Lonicera macranthoides Hand-Mazz. Acta Physiol Plant 39:27
Comino C, Hehn A, Moglia A, Menin B, Bourguad F, Lanteri S, Portis E (2009) The isolation and mapping of a novel hydroxycinnamoyltransferase in the globe artichoke chlorogenic acid pathway. BMC Plant Biol 9:30
Comino C, Lanteri S, Portis E, Acquadro A, Romani A, Hehn A, Larbat R, Bourgaud F (2007) Isolation and functional characterization of a cDNA coding a hydroxycinnamoyl transferase involved in phenylpropanoid biosynthesis in Cynara cardunculus L. BMC Plant Biol 7:14
Chang S, Tan C, Frankel NF, Barrett DM (2000) Low-density lipoprotein antioxidant activity of phenolic compounds and polyphenol oxidase activity in selected clingstone peach cultivars. J Agric Food Chem 48:147–151
Cui T, Nakamura K, Ma L, Li JZ, Kayahara H (2005) Analyses of arbutin and chlorogenic acid, the major phenolic constituents in oriental pear. J Agric Food Chem 53:3882–3887
Florea L, Song L, Salzberg SL (2013) Thousands of exon skipping events differentiate among splicing patterns in sixteen human tissues. F1000 Res 2:188
Hammerschmidt R (2014) Chlorogenic acid: a versatile defense compound. Physiol Mol Plant Pathol 88:iii–iv
He JG, Chang YD, Guan JF, Ge WY, Zhao Z (2017) Changes of chlorogenic acid content and its synthesis-associated genes expression in Xuehua pear fruit during development. J Integr Agric 16:471–477
Hoffmann L, Besseau S, Geoffroy P, Ritzenthaler C, Meyer D, Lapierre C, Pollet B, Legrand M (2004) Silencing of hydroxycinnamoyl-coenzyme A shikimate/quinate hydroxycinnamoyl transferase Affects phenylpropanoid biosynthesis. Plant Cell 16:1446–1465
Jaiswal R, Heiko M, Anja M, Karar MGE, Kuhnert N (2014) Identification and characterization of chlorogenic acids, chlorogenic acid glycosides and flavonoids from Lonicera henryi L. (Caprifoliaceae) leaves by LC-MSn. Phytochemistry 108:252–263
Jiao WX, Li XX, Wang XM, Cao JK, Jiang WB (2018) Chlorogenic acid induces resistance against, Penicillium expansum, in peach fruit by activating the salicylic acid signaling pathway. Food Chem 260:274–282
Jung S, Lee T, Cheng CH, Buble K, Zheng P, Yu J, Humann J, Ficklin SP, Gasic K, Scott K, Frank M, Ru S, Hough H, Evans K, Peace C, Olmstead M, DeVetter LW, McFerson J, Coe M, Wegrzyn JL, Staton ME, Abbott AG, Main D (2019) 15 years of GDR: new data and functionality in the Genome Database for Rosaceae. Nucleic Acids Res 47:D1137–D1145
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Lombardo VA, Osorio S, Borsani J, Lauxmann MA, Bustamante CA, Budde CO (2011) Metabolic profiling during peach fruit development and ripening reveals the metabolic networks that underpin each developmental stage. Plant Physiol 157:1696–1710
Lu JF, Liu SY, Lu W, Xi WP (2017) Phenolic profiles and antioxidant activity of fruit pilp from different type of peaches. Sci Agric Sin 50:3205–3214
Martinez G, Regente M, Jacobi S, Rio M, Del Pinedo M, Delacanal L (2017) Chlorogenic acid is a fungicide active against phytopathogenic fungi. Pestic Biochem Physiol 140:30–35
Mudau SP, Steenkamp PA, Piater LA, Palma M, De Tucci M, Madala NE, Dubery IA (2018) Metabolomics-guided investigations of unintended effects of the expression of the hydroxycinnamoyl quinate hydroxycinnamoyl transferase (hqt1) gene from Cynara cardunculus var. scolymus in Nicotiana tabacum cell cultures. Plant Physiol Biochem 127:287–298
Naveed M, Hejazi V, Abbas M, Kamboh AA, Khan GJ, Shumzaid M, Ahmad F, Babazadeh D, Xia FF, Modarresi GF, Li WH, Zhou XH (2018) Chlorogenic acid (CGA): a pharmacological review and call for further research. Biomed Pharmacother 97:67–74
Niggeweg R, Michael AJ, Martin C (2004) Engineering plants with increased levels of the antioxidant chlorogenic acid. Nat Biotechnol 22:746–754
Orazem P, Mikulic-Petkovsek M, Stampar F, Hudina M (2013) Changes during the last ripening stage in pomological and biochemical parameters of the ‘Redhaven’ peach cultivar grafted on different rootstocks. Sci Hortic 160:326–334
Shadle G, Chen F, Reddy MS, Jackson L, Nakashima J, Dixon RA (2007) Down-regulation of hydroxycinnamoyl CoA: Shikimate hydroxycinnamoyl transferase in transgenic alfalfa affects lignification, development and forage quality. Phytochemistry 68:1521–1529
Sonnante G, Damore R, Blanco E, Pierri CL, Palma M, De Luo J, Tucci M, Martin C (2010) Novel hydroxycinnamoyl-coenzyme A quinate transferase genes from artichoke are involved in the synthesis of chlorogenic acid. Plant Physiol 153:1224–1238
Tong ZG, Gao ZH, Wang F, Zhou J, Zhang Z (2009) Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol Biol 10:1–3
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, Baren MJ, Van Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515
Tomas-Barberan FA, Gil MI, Cremin P, Waterhouse AL, Hess-Pierce B, Kader AA (2001) HPLC-DAD-ESIMS Analysis of phenolic compounds in nectarines, peaches, and plums. J Agric Food Chem 49:4748–4760
Villarino M, Sandin-Espana P, Melgarejo P, De Cal A (2011) High chlorogenic and neochlorogenic acid levels in immature peaches reduce Monilinia laxa infection by interfering with fungal melanin biosynthesis. J Agric Food Chem 59:3205–3213
Vitus IO, Juan JB, Josep U, Yolanda G (2019) Breeding strategies for identifying superior peach genotypes resistant to brown rot. Sci Hortic 246:1028–1036
Yan J, Song ZZ, Cai ZX, Shen ZJ, Ma RJ, Yu ML (2018) Proanthocyanidin accumulation in peach fruit with three types of flesh color Jiangsu. Agric J 34:651–656
Yan J, Cai ZX, Shen ZJ, Zhang BB, Qian W, Yu ML (2014a) Determination and comparison of 10 phenolic compounds in peach with three types of flesh color. Acta Horticult Sin 41:319–328
Yan J, Shen ZJ, Cai ZX, Yu ML (2014b) Advances of study on phenolic compounds in peach fruit. J Fruit Sci 31:477–485
Zhang BB, Shen ZJ, Ma RJ, Jiang H, Yan J, Yu ML (2018a) Antioxidant capacity evaluation of peach fruit based on flesh individual phenol and total phenol content. Acta Horticult Sin 45:121–132
Zhang BB, Shen ZJ, Ma RJ, Yan J, Yu ML (2018b) Grey relational analysis and fuzzy synthetic discrimination of antioxidant components in peach fruit. Arch Biol Sci 70:449–458
Zheng BB, Zhao L, Jiang XH, Cherono S, Liu JJ, Ogutu C, Ntini C, Zhang XJ, Han YP (2020) Assessment of organic acid accumulation and its related genes in peach. Food Chem 334:127567
Zhou L, Kang GD, Wang Q, Zhang LY, Zhang CX, Tian Y, Cong PH (2013) Analysis of chlorogenic acid content in Apple Fruit during development. China Fruits 3:36–38
Zhu P, Miao XL, Chen Y (2016) Degradation kinetics of chlorogenic acid, cryptochlorogenic acid, and neochlorogenic acid at neutral and alkaline pH values. Acta Pharm Sin 51:122–126
Acknowledgements
This work was supported by funds of Natural Science Foundation of Jiangsu Province (BK20200278); China Agriculture Research System (CARS-30); Species Conservation Project of Ministry of Agriculture and Rural Affair (19190156); National Crop Germplasm Resources Infrastructure in China (NHGRC2020-NH16).
Author information
Authors and Affiliations
Contributions
JY provided the experimental ideas and designed the research; SG, MZ, BZ, and ZC performed the experiments and the analyzed data. JY and ZS drafted the manuscript. ZS, RM, and MY revised the paper. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that they have no conflict of interest.
Additional information
Communicated by Heakeun Yun.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yan, J., Su, Z., Guo, S. et al. Chlorogenic acid accumulation and related gene expression in peach fruit. Hortic. Environ. Biotechnol. 63, 403–411 (2022). https://doi.org/10.1007/s13580-021-00399-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-021-00399-y