Abstract
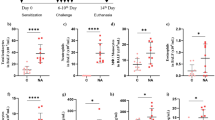
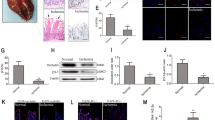
This study investigated the molecular mechanism by which bronchoalveolar lavage fluid exosomes (BALF-exo) alleviated acute lung injury (ALI). BALF-exo was isolated from mice. LPS was used to induce inflammation in rat alveolar macrophages (NR8383). NR8383 cell models were treated with BALF-exo or BALF-exo loaded with miR-223-3p mimics/inhibitors, or STK39 was overexpressed in NR8383 cells before LPS and BALF-exo treatment. These cells were subjected to apoptosis, autophagy, and inflammation assays. RNA immunoprecipitation and dual-luciferase reporter assay were conducted to verify the binding between miR-223-3p and STK39. LPS-induced ALI mouse models were treated with BALF-exo loaded with miR-223-3p mimics. miR-223-3p was lowly expressed in BALF-exo from LPS-treated mice. BALF-exo loaded with miR-223-3p mimics increased viability and autophagy and decreased apoptosis and inflammation in NR8383 cell models. Inhibition of miR-223-3p in BALF-exo or overexpression of STK39 in NR8383 cells repressed the therapeutic effect of BALF-exo in LPS-treated NR8383 cells. STK39 was a target gene of miR-223-3p. miR-223-3p shuttled by BALF-exo negatively regulated the expression of STK39 in NR8383 cells. BALF-exo loaded with miR-223-3p mimics also reduced lung injuries in ALI mice. In conclusion, miR-223-3p loaded in BALF-exo alleviates ALI by targeting STK39 in alveolar macrophages.






Similar content being viewed by others
References
Butt Y, Kurdowska A, Allen TC. Acute lung injury: a clinical and molecular review. Arch Pathol Lab Med. 2016;140:345–50.
Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315:788–800.
Mowery NT, Terzian WTH, Nelson AC. Acute lung injury. Curr Probl Surg. 2020;57:100777.
Hughes KT, Beasley MB. Pulmonary manifestations of acute lung injury: more than just diffuse alveolar damage. Arch Pathol Lab Med. 2017;141:916–22.
Fan EKY, Fan J. Regulation of alveolar macrophage death in acute lung inflammation. Respir Res. 2018;19:50.
Hou L, Yang Z, Wang Z, et al. NLRP3/ASC-mediated alveolar macrophage pyroptosis enhances HMGB1 secretion in acute lung injury induced by cardiopulmonary bypass. Lab Invest. 2018;98:1052–64.
Yang L, Zhang Z, Zhuo Y, et al. Resveratrol alleviates sepsis-induced acute lung injury by suppressing inflammation and apoptosis of alveolar macrophage cells. Am J Transl Res. 2018;10:1961–75.
D’Arcy MS. Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int. 2019;43:582–92.
Qian Q, Cao X, Wang B, et al. Endoplasmic reticulum stress potentiates the autophagy of alveolar macrophage to attenuate acute lung injury and airway inflammation. Cell Cycle. 2020;19:567–76.
Peng W, Peng F, Lou Y, et al. Autophagy alleviates mitochondrial DAMP-induced acute lung injury by inhibiting NLRP3 inflammasome. Life Sci. 2021;265:118833.
Carnino JM, Lee H, Jin Y. Isolation and characterization of extracellular vesicles from Broncho-alveolar lavage fluid: a review and comparison of different methods. Respir Res. 2019;20:240.
He C, Zheng S, Luo Y, Wang B. Exosome theranostics: biology and translational medicine. Theranostics. 2018;8:237–55.
Ferguson SW, Nguyen J. Exosomes as therapeutics: the implications of molecular composition and exosomal heterogeneity. J Control Release. 2016;228:179–90.
Lu TX, Rothenberg ME. MicroRNA. J Allergy Clin Immunol. 2018;141:1202–7.
Ju M, Liu B, He H, et al. MicroRNA-27a alleviates LPS-induced acute lung injury in mice via inhibiting in fl ammation and apoptosis through modulating TLR4/MyD88/NF-kappaB pathway. Cell Cycle. 2018;17:2001–18.
Xie W, Lu Q, Wang K, et al. miR-34b-5p inhibition attenuates lung inflammation and apoptosis in an LPS-induced acute lung injury mouse model by targeting progranulin. J Cell Physiol. 2018;233:6615–31.
Yi X, Wei X, Lv H, et al. Exosomes derived from microRNA-30b-3p-overexpressing mesenchymal stem cells protect against lipopolysaccharide-induced acute lung injury by inhibiting SAA3. Exp Cell Res. 2019;383:111454.
Gallolu Kankanamalage S, Lee AY, Wichaidit C, et al. WNK1 is an unexpected autophagy inhibitor. Autophagy. 2017;13:969–70.
Lu Z, Shen J, Chen X, et al. Propofol upregulates MicroRNA-30b to inhibit excessive autophagy and apoptosis and attenuates ischemia/reperfusion injury in vitro and in patients. Oxid Med Cell Longev. 2022;2022:2109891.
Matthay MA, McAuley DF, Ware LB. Clinical trials in acute respiratory distress syndrome: challenges and opportunities. Lancet Respir Med. 2017;5:524–34.
Yang B, Chen Y, Shi J. Exosome biochemistry and advanced nanotechnology for next-generation theranostic platforms. Adv Mater. 2019;31:e1802896.
Ye C, Li H, Bao M, Zhuo R, Jiang G, Wang W. Alveolar macrophage-derived exosomes modulate severity and outcome of acute lung injury. Aging (Albany NY). 2020;12:6120–8.
Zhou Y, Li P, Goodwin AJ, et al. Exosomes from endothelial progenitor cells improve outcomes of the lipopolysaccharide-induced acute lung injury. Crit Care. 2019;23:44.
Jiang K, Yang J, Guo S, Zhao G, Wu H, Deng G. Peripheral circulating exosome-mediated delivery of miR-155 as a novel mechanism for acute lung inflammation. Mol Ther. 2019;27:1758–71.
Wei X, Yi X, Lv H, et al. MicroRNA-377-3p released by mesenchymal stem cell exosomes ameliorates lipopolysaccharide-induced acute lung injury by targeting RPTOR to induce autophagy. Cell Death Dis. 2020;11:657.
Ji Q, Xu X, Song Q, et al. miR-223-3p Inhibits Human Osteosarcoma Metastasis and Progression by Directly Targeting CDH6. Mol Ther. 2018;26:1299–312.
Han LL, Zhou XJ, Li FJ, et al. MiR-223-3p promotes the growth and invasion of neuroblastoma cell via targeting FOXO1. Eur Rev Med Pharmacol Sci. 2019;23:8984–90.
Zhu Y, Li K, Yan L, He Y, Wang L, Sheng L. miR-223-3p promotes cell proliferation and invasion by targeting Arid1a in gastric cancer. Acta Biochim Biophys Sin (Shanghai). 2020;52:150–9.
Zhang D, Lee H, Wang X, et al. A potential role of microvesicle-containing miR-223/142 in lung inflammation. Thorax. 2019;74:865–74.
Kim GD, Ng HP, Patel N, Mahabeleshwar GH. Kruppel-like factor 6 and miR-223 signaling axis regulates macrophage-mediated inflammation. FASEB J. 2019;33:10902–15.
Wu J, Niu P, Zhao Y, et al. Impact of miR-223-3p and miR-2909 on inflammatory factors IL-6, IL-1ss, and TNF-alpha, and the TLR4/TLR2/NF-kappaB/STAT3 signaling pathway induced by lipopolysaccharide in human adipose stem cells. PLoS ONE. 2019;14:e0212063.
Zhou Y, Zhang T, Yan Y, et al. MicroRNA-223–3p regulates allergic in fl ammation by targeting INPP4A. Braz J Otorhinolaryngol. 2020;87:591–600.
Fava C, Danese E, Montagnana M, et al. Serine/threonine kinase 39 is a candidate gene for primary hypertension especially in women: results from two cohort studies in Swedes. J Hypertens. 2011;29:484–91.
Shin DJ, Lee SH, Park S, Jang Y. Association between serine/threonine kinase 39 gene polymorphism, hypertension, and other cardiovascular risk factors in Koreans. Korean Circ J. 2013;43:13–22.
Zhao H, Qi Y, Wang Y, et al. Interactive contribution of serine/threonine kinase 39 gene multiple polymorphisms to hypertension among northeastern Han Chinese. Sci Rep. 2014;4:5116.
Huang T, Zhou Y, Cao Y, Tao J, Zhou ZH, Hang DH. STK39, overexpressed in osteosarcoma, regulates osteosarcoma cell invasion and proliferation. Oncol Lett. 2017;14:4599–604.
Li C, Wang A, Chen Y, Liu Y, Zhang H, Zhou J. MicroRNA2995p inhibits cell metastasis in breast cancer by directly targeting serine/threonine kinase 39. Oncol Rep. 2020;43:1221–33.
Zhao Q, Zhu Y, Liu L, et al. STK39 blockage by RNA interference inhibits the proliferation and induces the apoptosis of renal cell carcinoma. Onco Targets Ther. 2018;11:1511–9.
Vergadi E, Ieronymaki E, Lyroni K, Vaporidi K, Tsatsanis C. Akt signaling pathway in macrophage activation and M1/M2 polarization. J Immunol. 2017;198:1006–14.
Hong J, Mo S, Gong F, et al. lncRNA-SNHG14 plays a role in acute lung injury induced by lipopolysaccharide through regulating autophagy via miR-223-3p/Foxo3a. Mediat Inflamm. 2021;2021:7890288.
Tan HY, Qing B, Luo XM, Liang HX. Downregulation of miR-223 promotes HMGB2 expression and induces oxidative stress to activate JNK and promote autophagy in an in vitro model of acute lung injury. J Inflamm (Lond). 2021;18:29.
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
HN and LHX conceived the ideas. HN and LHX designed the experiments. THY, DXY and LHX performed the experiments. LHX and THY analyzed the data. HN and THY provided critical materials. DXY and SL wrote the manuscript. HN supervised the study. HN, LHX and QB revised the manuscript. All the authors have read and approved the final version for publication.
Corresponding author
Ethics declarations
Conflict of interests
This study declares no conflict of interests.
Ethical approval
All animal experiments were approved by the Animal Care and Use Committee of The Second Xiangya Hospital of Central South University and abided by the guidelines for laboratory animal care and use issued by the National Institutes of Health. Additionally, all the experiments were carried out with the best efforts to reduce the pain and number of experimental animals.
Consent to participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, N., Tan, H., Deng, X. et al. MiR-223-3p-loaded exosomes from bronchoalveolar lavage fluid promote alveolar macrophage autophagy and reduce acute lung injury by inhibiting the expression of STK39. Human Cell 35, 1736–1751 (2022). https://doi.org/10.1007/s13577-022-00762-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-022-00762-w