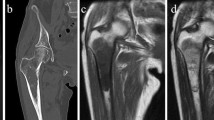
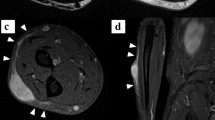
Abstract
Dedifferentiated chondrosarcoma is an aggressive mesenchymal tumor of the bone, and novel therapies are needed to improve its clinical outcomes. Patient-derived cell lines are essential tools for elucidating disease mechanisms associated with poor prognosis and for developing therapies. However, few lines and xenografts have been previously reported in dedifferentiated chondrosarcoma. We established a novel patient-derived dedifferentiated chondrosarcoma cell line, NCC-dCS1-C1. Primary dedifferentiated chondrosarcoma tissues were obtained at the time of surgery and subjected to primary tissue culture. The cell line was established and authenticated by assessing DNA microsatellite short tandem repeats. The cells maintained in monolayer cultures exhibited constant growth, spheroid formation capacity, and invasion ability. When the cells were implanted into mice, they exhibited histological features similar to those of the original tumor. Genomic analysis of single nucleotide polymorphisms showed aberrant genomic contents. The DNA sequencing revealed the absence of IDH1/2 mutations. The global targeted sequencing revealed that the cell line preserved homozygous deletion of CDKN2A and CREBBP. A proteomic study by mass spectrometry unveiled similar but distinct molecular backgrounds in the original tumor and the established cell line, suggesting that tumor cell functions might be altered during the establishment of the cell line. Using a screening approach, four anti-cancer drugs with anti-proliferative effects at a low concentration were identified. In conclusion, a novel dedifferentiated chondrosarcoma cell line, NCC-dCS1-C1, was successfully established from primary tumor tissues. The NCC-dCS1-C1 cell line will be a useful tool for investigations of the mechanisms underlying dedifferentiated chondrosarcomas.







Similar content being viewed by others
References
Dahlin DC, Beabout JW. Dedifferentiation of low-grade chondrosarcomas. Cancer. 1971;28:461–6.
Bruns J, Fiedler W, Werner M, Delling G. Dedifferentiated chondrosarcoma–a fatal disease. J Cancer Res Clin Oncol. 2005;131:333–9.
Capanna R, Bertoni F, Bettelli G, et al. Dedifferentiated chondrosarcoma. J Bone Jt Surg Am. 1988;70:60–9.
Staals EL, Bacchini P, Bertoni F. Dedifferentiated central chondrosarcoma. Cancer. 2006;106:2682–91.
Frassica FJ, Unni KK, Beabout JW, Sim FH. Dedifferentiated chondrosarcoma. A report of the clinicopathological features and treatment of seventy-eight cases. J Bone Jt Surg Am. 1986;68:1197–205.
Anract P, Tomeno B, Forest M. Dedifferentiated chondrosarcoma. A study of 13 clinical cases and review of the literature. Revue de chirurgie orthopedique et reparatrice de l’appareil moteur. 1994;80:669–80.
Mitchell AD, Ayoub K, Mangham DC, Grimer RJ, Carter SR, Tillman RM. Experience in the treatment of dedifferentiated chondrosarcoma. J Bone Jt Surg Br Vol. 2000;82:55–61.
Grimer RJ, Gosheger G, Taminiau A, et al. Dedifferentiated chondrosarcoma: prognostic factors and outcome from a European group. Eur J Cancer. 2007;43:2060–5.
Johnson S, Tetu B, Ayala AG, Chawla SP. Chondrosarcoma with additional mesenchymal component (dedifferentiated chondrosarcoma). I. A clinicopathologic study of 26 cases. Cancer. 1986;58:278–86.
Yokota K, Sakamoto A, Matsumoto Y, et al. Clinical outcome for patients with dedifferentiated chondrosarcoma: a report of 9 cases at a single institute. J Orthop Surg Res. 2012;7:38.
Gambarotti M, Righi A, Frisoni T, et al. Dedifferentiated chondrosarcoma with “adamantinoma-like” features: a case report and review of literature. Pathol Res Pract. 2017;213:698–701.
Ishida T, Kuwada Y, Motoi N, Oka T, Machinami R. Dedifferentiated chondrosarcoma of the rib with a malignant mesenchymomatous component: an autopsy case report. Pathol Int. 1997;47:397–403.
Jour G, Liu Y, Ricciotti R, Pritchard C, Hoch BL. Glandular differentiation in dedifferentiated chondrosarcoma: molecular evidence of a rare phenomenon. Hum Pathol. 2015;46:1398–404.
Sopta J, Dordevic A, Tulic G, Mijucic V. Dedifferentiated chondrosarcoma: our clinico-pathological experience and dilemmas in 25 cases. J Cancer Res Clin Oncol. 2008;134:147–52.
Zhang Y, Paz Mejia A, Temple HT, Trent J, Rosenberg AE. Squamous cell carcinoma arising in dedifferentiated chondrosarcoma proved by isocitrate dehydrogenase mutation analysis. Hum Pathol. 2014;45:1541–5.
Liu C, Xi Y, Li M, et al. Dedifferentiated chondrosarcoma: Radiological features, prognostic factors and survival statistics in 23 patients. PLoS One. 2017;12:e0173665.
Barretina J, Caponigro G, Stransky N, et al. The cancer cell line encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature. 2012;483:603–7.
Kudo N, Ogose A, Hotta T, et al. Establishment of novel human dedifferentiated chondrosarcoma cell line with osteoblastic differentiation. Virchows Arch. 2007;451:691–9.
van Oosterwijk JG, de Jong D, van Ruler MA, et al. Three new chondrosarcoma cell lines: one grade III conventional central chondrosarcoma and two dedifferentiated chondrosarcomas of bone. BMC Cancer. 2012;12:375.
Monderer D, Luseau A, Bellec A, et al. New chondrosarcoma cell lines and mouse models to study the link between chondrogenesis and chemoresistance. Lab Investig. 2013;93:1100–14.
Oyama R, Takahashi M, Yoshida A, et al. Generation of novel patient-derived CIC–DUX4 sarcoma xenografts and cell lines. Sci Rep. 2017;7:4712.
Tomayko MM, Reynolds CP. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother Pharmacol. 1989;24:148–54.
Kito F, Oyama R, Sakumoto M, et al. Establishment and characterization of novel patient-derived osteosarcoma xenograft and cell line. In vitro cellular developmental biology. Animal. 2018;54:528–36.
Hao S, Hong CS, Feng J, et al. Somatic IDH1 mutation in a pituitary adenoma of a patient with Maffucci syndrome. J Neurosurg. 2016;124:1562–7.
Moriya K, Kaneko MK, Liu X, et al. IDH2 and TP53 mutations are correlated with gliomagenesis in a patient with Maffucci syndrome. Cancer Sci. 2014;105:359–62.
Kato M, Nakamura H, Nagai M, et al. A computational tool to detect DNA alterations tailored to formalin-fixed paraffin-embedded samples in cancer clinical sequencing. Genome Med. 2018;10:44.
ANNOVAR. functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010;38:e164. https://doi.org/10.1093/nar/gkq603. Epub 2010 Jul 3.
The COSMIC. (Catalogue of Somatic Mutations in Cancer) database and website. Br J Cancer. 2004;91:355–8. https://doi.org/10.1038/sj.bjc.6601894.
Diagnostic utility of IDH. 1/2 mutations to distinguish dedifferentiated chondrosarcoma from undifferentiated pleomorphic sarcoma of bone. Hum Pathol. 2017;65:239–246. https://doi.org/10.1016/j.humpath.2017.05.015 (Epub 17 May 25).
Byrne AT, Alferez DG, Amant F, et al. Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat Rev Cancer. 2017;17:254–68.
Verweij J, Pinedo HM. Mitomycin C. mechanism of action, usefulness and limitations. Anticancer Drugs. 1990;1:5–13.
Mazerski J, Martelli S, Borowski E. The geometry of intercalation complex of antitumor mitoxantrone and ametantrone with DNA: molecular dynamics simulations. Acta Biochim Pol. 1998;45:1–11.
Huang WS, Metcalf CA, Sundaramoorthi R, et al. Discovery of 3-[2-(imidazo[1,2-b]pyridazin-3-yl)ethynyl]-4-methyl-N-{4-[(4-methylpiperazin-1-y l)methyl]-3-(trifluoromethyl)phenyl}benzamide (AP24534), a potent, orally active pan-inhibitor of breakpoint cluster region-abelson (BCR-ABL) kinase including the T315I gatekeeper mutant. J Med Chem. 2010;53:4701–19.
Ueda H, Nakajima H, Hori Y, et al. FR901228, a novel antitumor bicyclic depsipeptide produced by Chromobacterium violaceum No. 968. I. Taxonomy, fermentation, isolation, physico-chemical and biological properties, and antitumor activity. J Antibiot. 1994;47:301–10.
Burstein HJ, Mangu PB, Somerfield MR, et al. American Society of Clinical Oncology clinical practice guideline update on the use of chemotherapy sensitivity and resistance assays. J Clin Oncol. 2011;29:3328–30.
Samson DJ, Seidenfeld J, Ziegler K, Aronson N. Chemotherapy sensitivity and resistance assays: a systematic review. J Clin Oncol. 2004;22:3618–30.
Acknowledgements
We thank Drs. M. Endo, Y. Minami, K. Shimizu, T. Mori, T. Uehara, M. Sugawara, Y. Araki, and Ms. R. Nakano of the Division of Musculoskeletal Oncology, National Cancer Center Hospital for sampling tumor tissue specimens from surgically resected materials. We also thank Dr. Ishigamori and Mr. Uchiya of National Cancer Center Research Core facility for technical assistance in animal experiments. This research was supported by the National Cancer Center Research and Development Fund (26-A-9 and 29-A-2). The experiments of SNP array and oncopanel were supported by Fundamental Innovative Oncology Core in the National Cancer Center, and we appreciate the technical supports by Drs. Ichikawa. We would like to thank Editage (http://www.editage.jp) for English language editing and constructive comments regarding the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oyama, R., Kito, F., Takahashi, M. et al. Establishment and characterization of a novel dedifferentiated chondrosarcoma cell line, NCC-dCS1-C1. Human Cell 32, 202–213 (2019). https://doi.org/10.1007/s13577-018-00232-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-018-00232-2