Abstract
Frontal fibrosing alopecia (FFA) represents a distinctive form of primary lymphocytic scarring alopecia characterized by fronto-temporal hair recession and eyebrow hair loss. While predominantly affecting postmenopausal women, FFA also occurs in women of reproductive age and men, with variations observed across different ethnic groups. Genetic predisposition, environmental factors and inflammatory pathways contribute to its pathogenesis, with evolving diagnostic criteria enhancing accuracy. FFA treatment lacks standardization, encompassing topical, systemic and physical therapies, while hair transplantation remains a temporary solution. This article reviews the current understanding of FFA, aiming to provide clinicians with updated insights for its management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Frontal fibrosing alopecia (FFA) is a scarring hair loss condition characterized by fronto-temporal hair recession and eyebrow hair loss. |
FFA predominantly manifests in postmenopausal women but can also occur in reproductive-aged women and men, with a higher prevalence observed in individuals of Caucasian ethnicity, although cases have been documented in black and Asian patients as well. |
The pathogenesis of FFA is multifactorial, involving genetic predisposition, environmental factors and inflammatory pathways, with notable involvement of PPAR-γ and mammalian target of rapamycin (mTOR). |
FFA diagnosis integrates clinical, trichoscopic and histopathologic criteria, with evolving guidelines enhancing accuracy. |
Treatment for FFA lacks standardized guidelines, with topical options like corticosteroids, calcineurin inhibitors, minoxidil and Janus kinase (JAK) inhibitors, systemic approaches including hydroxychloroquine, tetracyclines and 5α-reductase inhibitors, alongside adjunctive benefits from physical therapies such as excimer laser and LED photobiomodulation. |
Why carry out this study? |
FFA presents a growing challenge globally, characterized by its complexity and increasing prevalence. However, the lack of standardized diagnostic approaches and management strategies renders FFA a significant diagnostic challenge for clinicians. |
What was learned from the study? |
The study shed light on FFA, highlighting its distinct clinical features, including hairline recession and eyebrow loss, with a prevalence among specific demographic groups. Moreover, it highlighted the dynamic nature of diagnostic criteria, cutting-edge advancements in therapeutic options, absence of uniform treatment protocols and escalating demand for pioneering treatments and non-invasive diagnostic techniques to combat the increasing global prevalence of FFA. |
Introduction
Frontal fibrosing alopecia (FFA), initially described by Kossard in 1994 [1, 2], represents a variant of primary lymphocytic scarring alopecia characterized by a gradual band-like recession of the fronto-temporal hairline. Frontal area hairline recession presents in diverse presentations, including linear, diffuse zig-zag and pseudo-fringe patterns [3], often accompanied by involvement of sideburns and occipital hair. FFA can also involve the eyebrows in between 50 and 75% of cases [4], as well as the eyelashes, axillae, limbs and pubic region [5,6,7]. Beard cicatricial alopecia is common in males with FFA [5,6,7,8].
The true incidence of FFA is unclear, but the notable increase in reported cases has led some authors to label it as an epidemic condition [9]. Several cases of FFA have been documented globally, predominantly affecting postmenopausal women, but also in women with reproductive potential and in men [4, 9,10,11,12,13]. Although most documented cases involve individuals from Caucasian populations, FFA has also been observed in black and Asian patients from different countries [14]. Black patients typically experience FFA at a younger age and before menopause compared to white patients [15].
The debate persists regarding whether FFA simply falls under the category of lichen planopilaris (LPP) or if it constitutes a more intricate disorder [16,17,18]. Starace et al., in alignment with other smaller studies [19, 20], noted a correlation between FFA and LPP, suggesting that FFA might be a clinical variation of LPP, despite the manifestation of different symptoms [4].
While the clinical and trichoscopic presentation of FFA is often distinctive, in case of doubts it is advisable to perform a biopsy for histopathologic verification to ensure accurate treatment [21]. The need for non-invasive diagnostic techniques as well as new treatment approaches has surged alongside its rising incidence.
We conducted a narrative review to explore FFA's pathogenesis, diagnosis and treatment. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. The aim of this review is to provide a comprehensive overview of this condition, hopefully offering clinicians an updated guide to manage FFA properly.
Etiopathogenesis
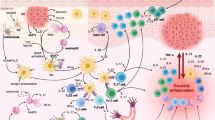
The exact etiopathogenesis of FFA is not fully understood. Inflammatory responses mediated by immune cells and cytokines seem to play a significant role. More specifically, increased infiltration of CD8 + cytotoxic T cells and dendritic cells around lesioned hair follicles (especially near the bulge area) exacerbates inflammation, leading to immune privilege (IP) collapse and damage to epithelial hair follicle stem cells (eHFSC) [41]. This results in gradual fibrosis of the entire follicle unit, leading to scarring alopecia [22]. However, the reason for this inflammatory process is unclear. Various factors such as immune dysregulation, abnormal fibrotic pathways, genetic predisposition, hormones and environmental influences have been suggested as potential contributors to the onset of FFA.
Genetic Factors
FFA has been the subject of genetic scrutiny for approximately a decade, initially explored through brief publications and case reports [23]. Familial patterns and cases in monozygotic twins have been reported, suggesting a genetic component [10, 24, 25]. Research has concentrated on the role of human leukocyte antigens (HLA) in FFA development. The HLA-B*07:02 mutation, uncovered through genome-wide association studies (GWAS), appears to promote auto-inflammatory responses against eHFSC, substantially elevating the risk of FFA [26].
Saceda-Corralo et al. conducted a cross-sectional study with 223 patients affected by FFA and determined that a total of 83.8% of patients with FFA carried the rs9258883 polymorphism in HLA- B*07:02, and the majority of them lacked the protective rs1800440 polymorphism in CYP1B1 (75.2%) [23]. Indeed, it seems that some patients possess protective alleles associated with the CYP1B1 gene (e.g., p.Asn453Ser), which could influence FFA risk [27].
Alterations in genetic pathways like PPAR-γ and mTOR have also been linked to FFA; they influence lipid metabolism, sebocyte differentiation and immune responses, potentially affecting the follicular health through inflammation and fibrosis induction [22]. Reduced PPAR-γ activity leads to fibrosis and inflammatory cell infiltration, while mTOR signaling interacts with PPAR-γ, modulating lipid homeostasis and inflammatory processes [22]. More specifically, a reduction in the production of peroxisomes and cholesterol likely contributes to the buildup of proinflammatory lipids in hair follicles, leading to the infiltration of inflammatory cells in the bulge region [42].
Environmental Factors
Factors such as formalin exposure, thyroid disease and alcohol use have been associated with FFA [31]. However, the exact mechanisms by which these factors contribute to FFA remain unclear and require further research. There is speculation that exposure to light, particularly ultraviolet (UV) radiation, may contribute to FFA [32]. Light exposure may affect the synthesis of certain compounds like 6-formylindolo[3,2-b]carbazole (FICZ), which can have pro- or anti-inflammatory effects depending on concentration. Additionally, studies conducted retrospectively using surveys and clinical cases have demonstrated a connection between FFA and use of sunscreens and other facial cosmetics among both male and female patients [33, 34]. Individuals affected by FFA reported a higher frequency of using sunscreen-based products, with 88% reporting daily usage throughout the year, in contrast to 29% in the control group [35]. Several hypotheses exist regarding the potential pathogenic role of sunscreen in FFA. One theory suggests that sunscreen penetrates the follicular infundibulum, triggering a lichenoid reaction against vellus and epidermal antigens [36, 37]. Another hypothesis posits that regular sunscreen application contributes to FFA by disrupting endocrine function. Some suggest that the systemic absorption of chemical (organic) UV filters might induce endocrine disruption due to estrogen-like activity observed in previous in vitro and animal experiments [38]. However, concrete evidence supporting a hormonal basis for FFA is lacking. Moreover, there is presently no substantiated evidence indicating that the systemic absorption of UV filters has any significant effect on the human endocrine system [39]. Lastly, it has been proposed that direct hair follicle damage resulting from oxidative stress may occur when titanium dioxide (TiO2), a common UV filter found in sunscreens, is exposed to UV radiation [40]. It is important to acknowledge, however, that TiO2 nanoparticles, present in virtually all sunscreen products and the majority of cosmeceuticals, are typically coated with a nonreactive substance to inhibit the release of reactive oxygen species into the tissue [39]. Additional research is necessary to arrive at definitive conclusions on this matter.
Hormones
Androgens, estrogen and other hormones play complex roles in hair growth and loss. While androgens are often associated with hair loss, the role of estrogen in FFA is still not fully understood. Changes in hormone levels, particularly during menopause, may influence the onset and progression of FFA [22]. Specifically, reduced levels of DHEA and androgens could induce a fibrotic condition in FFA. Research involving 30 female FFA patients and 34 healthy individuals demonstrated notably lower serum levels of DHEA and androstenedione in FFA patients compared to healthy subjects. This prompts inquiry into the potential effectiveness of 5α-reductase inhibitors for FFA treatment. However, the efficacy of these inhibitors might be overstated given the co-occurrence of female pattern hair loss (FPHL) or male androgenetic alopecia (MAGA) in many cases [28,29,30].
Understanding the multifactorial influences on FFA pathogenesis is crucial for developing effective treatments and prevention strategies. Further research is needed to elucidate the intricate interplay of these factors and their contributions to FFA.
Diagnostic Approach
Diagnosing FFA consists of a multi-step approach where clinical features, trichoscopy and sometimes histopathology are crucial to reach a proper diagnosis. The need for clarity concerning the diagnostic process of FFA is also linked to the cicatricial outcome of such condition, which can lead to irreversible hair loss in cases of late diagnosis [4]. Diagnostic clinical criteria for FFA were introduced in 2018 [43]. The primary criteria encompass scalp scarring alopecia in the frontotemporal region (without keratotic follicular papules elsewhere on the body) and bilateral diffuse alopecia of the eyebrows. Secondary criteria consist of trichoscopy showing typical features (peripilar erythema, peripilar desquamation or both), histopathologic features indicating scarring alopecia with FFA or LPP pattern, involvement of other areas indicative of FFA (such as the occipital region, face, sideburns, body hair) and the presence of non-inflammatory facial papules. The diagnosis necessitates the presence of two major criteria or one major and two minor criteria [43]. In 2021, the recommendations from the International Frontal Fibrosing Alopecia Consensus Group (IFFACG) enhanced diagnostic accuracy by establishing criteria for both "classic" and "probable" cases of FFA, aiming to prevent the exclusion of atypical variations [14]. Additionally, new techniques have been introduced into the diagnostic algorithm although further studies are still needed to define their utility during the daily clinical routine [4].
Clinical Features
FFA is a chronic condition characterized by a gradual band-like recession of the frontal hairline, with a progression rate of approximately 0.9 mm per month due to cicatricial alopecia [44]. The affected skin is smooth, light and lacks photoaging compared to the forehead skin exposed to chronic sunlight [45]. Raising the eyebrows can assist in identifying the original hairline [46]. Three clinical patterns of hairline loss have been outlined: linear, diffuse zig-zag and pseudo-fringe [47]. The linear pattern is the most common and describes a uniform frontal hairline recession without loss of hair density behind the hairline (Fig. 1a); the diffuse zig-zag pattern shows at least a 50% decrease in hair density behind the new hairline; the pseudo-fringe hairline recession shows some hair retained along the initial hairline [3]. Additional uncommon presentations include ophiasis-like, cockade-like and androgenetic alopecia-like patterns [14, 48]. Loss of sideburns, appearing as an absence of hair in front of the ears, is commonly associated (Fig. 1b). Occipital hairline involvement has been documented in 15–30% of cases [4,5,6, 19, 29, 43, 49]. A recent descriptive cross-sectional study by Perez-Mesonero et al. identified distinct patterns of occipital hair loss: ophiasic pattern, corresponding to a horizontal alopecic band affecting the entire occipital hairline, lateral pattern, indicating a recession on one or both sides with a preserved central tuft, patchy pattern, due to irregular areas of decreased hair density above the hairline and pseudo-fringe-sign pattern, which showed a band of decreased hair density located between the minimally affected occipital hairline and an upper zone of higher and homogeneous density (Fig. 1c) [49].
Partial or complete loss of the eyebrows, occasionally accompanied by peri- and interfollicular erythema, is a common feature, except in the 'pseudofringe' hair loss pattern that often spares the eyebrows. Eyebrow loss may precede or follow hairline involvement and, in some cases, may be the sole presentation, being misdiagnosed as alopecia areata or senile eyebrow loss [50, 51]. Loss of eyelashes and thinning of axillary, pubic, limb and body hair, sometimes accompanied by follicular keratosis and/or erythema, should also be noted and examined. It is important to be vigilant as patients may not report these symptoms, often attributing diffuse body hair thinning to menopause as a natural occurrence [4, 45].
Facial papules represent a hallmark clinical manifestation of frontal fibrosing alopecia (FFA) [4, 45, 52, 53]. These papular lesions are typically observed on the face as well as on the extremities and trunk [4]. Their presence signifies the involvement of facial vellus hair, which is obliterated because of the characteristic inflammatory infiltrate seen in FFA [54]. Histopathologically, established papules exhibit the absence of the hair follicle along with intact sebaceous glands and minimal to no inflammatory infiltration [52]. According to Pirmez et al. [53], the reduction and fragmentation of elastic fibers potentially contribute to the architectural changes in the papule, remodeling the sebaceous gland, enlarging it and causing its protrusion [45]. Supporting this theory, facial papules demonstrate improvement upon treatment with oral isotretinoin, which induces sebaceous gland atrophy and enhances the network of elastic fibers [45, 55].
In males, FFA may be detected early by observing involvement of the beard, eyebrows and/or sideburns (Fig. 1d) [8]. A multicenter study with 270 males revealed that one in five male patients with FFA reported beard hair loss at onset [13]. Exclusive sideburn involvement without frontotemporal hairline issues was observed in two individuals and patchy hair loss within the sideburns was common, with bilateral engagement in all cases [56].
FFA may coexist with classic lichen planus of the scalp or, occasionally, in other hairy areas, as well as with lichen planus pigmentosus [4, 11, 45]. Lichen planus pigmentosus, often misdiagnosed as melasma, is prevalent in dark phototype patients and may present alongside facial papules, erythema and hypo-/hyperpigmented macules [4, 11, 45]. Extra-facial red dots/erythema have been reported in the hip and upper chest, speculated to be an early manifestation of lichen planus pigmentosus [4, 11, 45]. Such clinical signs are histopathologically linked to FFA, characterized by a typical lichenoid inflammation, albeit interestingly not sparing the interfollicular epidermis as observed in the frontal hairline of the scalp [45].
Lastly, prominent frontotemporal veins, naturally appeared or induced by prolonged use of topical steroids, may be visible as well [4, 11, 45].
Trichoscopy
Trichoscopy can uncover the cicatricial nature of FFA, revealing diminished or absent follicular openings along the band-like recession [45] (see Table 1). The most distinctive features of FFA include the absence of vellus and intermediate hairs along the receded hairline, coupled with the presence of the lonely hair sign: these characteristics facilitate a swift differentiation from other hair disorders, such as alopecia areata (AA), traction alopecia (TA) and androgenetic alopecia (AGA) [57]. Follicular hyperkeratosis and perifollicular erythema, indicative of inflammation, are subtly observed around terminal hairs (Fig. 2a) and are often associated with dysaesthetic symptoms such as trichodynia and pruritus [4]. These indicators may manifest even in the prodromal phase, preceding the recession of the frontal hairline [44]. Other trichoscopic findings commonly include pili torti, broken hairs, black dots and yellow dots that are not pathognomonic of FFA [45]. A distinctive trichoscopic sign in FFA is the transparent proximal hair emergence, previously noted in sideburns (Fig. 2b) and found in 53% of occipital FFA cases (Fig. 2c) as well [49].
Trichoscopic findings in FFA. Trichoscopy (20×) of: front-temporal hairline (a) with perifollicular erythema, empty follicles and absence of follicular ostia, follicular hyperkeratosis; sideburns (b) and occipital regions (c) with single-hair follicular units, loss of follicular ostia, peripilar erythema and transparent proximal hair emergence; eyebrows (d) with absence of follicular openings, vellus hairs, diffuse erythema, upright regrowing hairs and hairs regrowth in distinct directions
Trichoscopy of eyebrows (Fig. 2d) in FFA reveals dystrophic hairs, whitish areas with no follicular openings and hair regrowth in distinct directions due to dermal fibrosis. Empty follicular openings, vellus hairs and upright regrowing hairs are considered favorable prognostic factors for eyebrow regrowth, whereas dystrophic hairs, regrowth in distinct directions, and whitish areas with no follicular openings are considered negative prognostic factors [58].
Ethnic variations in trichoscopic findings have been observed; for instance, black individuals with FFA may exhibit fewer perifollicular erythema and hyperkeratosis signs compared to the white population, while facial papules are more common in black individuals. Additionally, follicular hyperpigmentation and scalp pruritus are more prevalent in the black cohort [15]. Anzai et al. delineated more detailed trichoscopic features of eyebrows among FFA patients with Fitzpatrick Phototype skin types I through III as opposed to those with skin types IV through VI. The first group (Phototype skin types I through III) displayed a higher occurrence of telangiectasias, red dots, follicular plugs and perifollicular erythema, suggesting potential ethnic-related differences in the manifestations of FFA on the eyebrows [59].
It is crucial to note that trichoscopic signs of scarring, such as the absence of follicular ostia, may not be initially present in FFA. This suggests that FFA can develop without causing scarring, and early intervention may partially restore affected hair follicles [4].
Histopathology
Histologic findings typically reveal a varying density of lymphocytic infiltrate surrounding the infundibulum, isthmus and bulge areas of affected hair follicles, with early inflammatory changes at the follicular interface. In the advanced stage, a perifollicular ring of mucinous fibrosis separates the infiltrate from the outer root sheath. Hair follicle cycle changes may be associated, i.e., elevated presence of telogen and catagen hairs. The late fibrotic stage is characterized by loss of sebaceous glands, decrease in the number of hair follicles and their replacement with fibrotic scar tissue. These histologic alterations mirror those observed in LPP [60]. Doche et al. demonstrated that direct immunofluorescence (DIF) test can help in the diagnosis of FFA and LPP. They showed a DIF sensitivity of 41.6% in affected scalp and 28% in non-affected scalp, with higher sensitivity in non-affected scalp areas in FFA. Specificity was 66.6% in affected and 71% in non-affected scalp, displaying higher sensibility in FFA patients. The most prevalent immunoreacting agent was anti-immunoglobulin M antibody. This study suggests that both FFA and LPP may represent generalized inflammatory processes affecting the scalp, supported by positive findings in 'normal-appearing' scalp areas with notably higher specificity in FFA lesions [61].
New Instrumental Approaches
New technologies have been introduced in the diagnostic process of FFA. Two studies utilizing reflectance confocal microscopy (RCM) delineated a specific finding of FFA, the presence of widened follicular openings encircled by thickened, keratinized epithelium around the follicular orifice. Additionally, they noted multiple hair follicles merged into a single unit. Complete visualization of all hair shafts was not achievable in some cases [62, 63].
Ultrasonography (USG) has also emerged as a rapid and non-invasive diagnostic tool in dermatology. Recent descriptions of ultrasonographic signs associated with FFA include hypoechoic perifollicular thickening, heightened dermal vascular flow and the presence of one or more frontal veins at the dermo-hypodermic level [64].
Differential Diagnosis
FFA might be mistaken for non-scarring types of alopecia, including AA, TA and AGA, as scarring may not always be evident [4]. Clinical features and trichoscopic findings serve as a valuable tool in distinguishing FFA from such conditions [65].
TA may clinically resemble linear FFA but trichoscopy shows white dots, broken and miniaturized hair; AA is classically identified by the presence of patchy hair loss and typical trichoscopic findings such as black dots and exclamation mark hair [45, 66]. AGA is characterized by diffuse thinning in the crown area of the scalp and by hair diameter diversity and vellus-like hair. Several studies [4, 67, 68] suggest a link between AGA and FFA, indicating that both conditions might share common underlying pathogenic mechanisms, such as hormonal influences; AGA may also trigger fibrotic progression leading to FFA. However, the precise role of hormones remains uncertain and subject to debate, including the relationship among FFA, LPP and AGA. A retrospective analysis associated FFA with androgen deficiency, whereas LPP was more frequently linked with androgen excess [68].
Severity Assessment
Holmes et al. in 2016 introduced the FFA severity index (FFASI), intended for both clinical use and scientific literature assessment. FFASI includes two components: A and B. FFASI-A evaluates alopecia severity in four sections of the hairline, graded from 1 to 5 based on hairline recession. It also considers association with inflammatory band, non-scalp loss and associated features (e.g., nail lichen planus and mucosal LP). Scores combine for a maximum of 100. FFASIB employs a similar structure, but instead of assessing alopecia, it allows for personalized measurement of every hairline segment as defined by the user [69]. Criticisms of FFASI include its complexity for clinical use, inadequate representation of FFA prognosis and lack of validation [70]. Consequently, in 2018, a simpler FFA severity score (FFASS) was proposed, considering frontotemporal alopecia extent, eyebrow loss, peripilar erythema, peripilar desquamation, pruritus and pain. FFASS ranges from 0 to 25, with higher values indicating more severity. However, FFASS has its own limitations: absence of trichoscopy data and exclusive evaluation of female patients [71]. The main differences between the two scores are that FFASI gives points for facial papules, cutaneous lichen planus, oral or genital LP lesions, and nail involvement but no points for symptoms; FFASS gives no points for non-scalp/non-eyebrow involvement but points for pruritus and pain [14]. Recently, the IFFACG has developed a new structured staging system to assess the severity of FFA. This staging system, known as the FFA Global Staging Score, incorporates five key indicators commonly encountered in FFA cases. First, the staging system evaluates the extent of scalp hair loss attributed to frontal hairline recession. Severity levels range from absent to severe, with specific criteria delineated for minimal, mild, moderate and severe hair loss. Second, the degree of eyebrow loss is assessed, with scores indicating the presence of partial or total loss in one or both eyebrows. Additionally, the system considers the presence or absence of facial papules, with particular attention to the prominence of forehead veins and the occurrence of facial hyperpigmentation. The goal of this staging system is to standardize the evaluation process and provide clinicians with a comprehensive framework for assessing FFA severity [14].
Therapy
No currently standardized treatment for FFA has been approved. Both topical and systemic solutions, as well as physical therapies, have been proposed with varying results [7, 29, 45, 72,73,74,75,76] (see Table 2). It is important to make clear to patients that, being a cicatricial alopecia, the aim of the treatment is to stabilize hair loss, as hair growth is difficult to achieve. Furthermore, spontaneous stabilization has been reported in some cases with no treatment intervention [69].
Topical Treatment
Topical corticosteroids (tCS) have been a cornerstone in FFA treatment due to their relatively minimal adverse side effects. However, their efficacy as monotherapy is limited. A randomized controlled trial (RCT) evaluating the efficacy of monotherapy with topical clobetasol 0.05% or tacrolimus 0.1% compared to combination therapy with oral isotretinoin demonstrated that combination therapy was more effective than monotherapy [72]. Studies by Hepp et al. have shown that combining high-potency tCS with topical calcineurin inhibitors (tCI), like pimecrolimus 1% cream, quickly reduced hair loss and induced a decrease of subjective symptoms such as trichodynia and pruritus in a significant percentage of patients. Disease stabilization was obtained after a period of 9 months [77]. The authors prefer avoiding tCSs as they can worsen the skin atrophy and make the frontal veins more visible. Both topical calcineurin inhibitors (tCI), pimecrolimus and tacrolimus, are effective in FFA treatment, particularly when combined with other modalities [7, 74]. Strazzulla et al. in a retrospective cohort analysis of 92 FFA cases showed that patients treated with tacrolimus 0.3% were more likely to achieve stabilization of hair loss within 3 months compared with clobetasol/betamethasone [73]. A systematic review by Zhang et al. involving 29 patients reported that combination therapy with tCI, topical and intralesional corticosteroids, hydroxychloroquine and excimer laser therapy was among the most efficacious treatments at limiting hair loss [74]. Alternating tCS with tCI has been proposed to mitigate adverse side effects associated with prolonged tCS use, such as skin atrophy and telangiectasia [7].
Topical minoxidil is a well-established track treatment of AGA, as it induces increased hair density and quantity, also acting by both restraining excess fibroblasts and boosting vascular endothelial growth factors [29]. Combined with other therapies as a supplementary treatment, topical minoxidil should be prescribed to FFA patients needing to enhance hair volume. Tosti and colleagues documented favorable outcomes in FFA patient care by employing topical minoxidil 2% BID alongside oral finasteride 2.5 mg per day, successfully arresting disease progression in 50% of patients within 12–18 months of therapy [29].
Intralesional Treatment
Intralesional steroids (ILCSs) have been explored as a therapeutic alternative for FFA as well. Vañó-Galván et al. reported an overall positive clinical response rate of 83% in patients with FFA who received ILC injections every 3 to 6 months. They concluded that treatment with ILCs and 5α-reductase inhibitors should be combined in patients exhibiting follicular hyperkeratosis and perifollicular erythema [11]. In a retrospective cohort study involving 62 patients with FFA, Banka et al. reported that treatment with intralesional triamcinolone acetonide injected into the frontal scalp was beneficial in achieving disease stabilization in 97% of treated patients [6]. A review by Moreno-Ramirez et al. found that all 15 patients who received 20 mg/ml of intralesional triamcinolone acetonide injections every 3 months to the frontal hairline and eyebrows achieved disease stabilization [68]. The authors of this review reported efficacy and safety of ILCs (2.5 mg/ml) for eyebrow involvement in FFA, but regarding the frontal hairline they suggested to restrict injections only to the areas where inflammation is active at trichoscopy to avoid skin atrophy.
Considering the heightened activity of the JAK-STAT pathway in the pathogenesis of FFA, the therapeutic potential of JAK inhibitors (JAKis) has been explored lately in LPP/FFA treatments. While there are documented case reports and series illustrating the efficacy of oral JAKis in treating LPP/FFA, they are also linked with adverse effects, including serious infections in up to 4.8% of patients [78]. Topical formulations represent a potential alternative with reduced risks compared to systemic therapy. Currently, there has been only one case of a patient with FFA who exhibited improvement using 2% tofacitinib (a JAK-1/3 inhibitor) cream [79]. A retrospective chart review conducted on LPP/FFA patients treated with topical tofacitinib demonstrated reduced risks compared to systemic therapy [75]. In a case series by Dunn et al., two women with FFA showed significant improvement with topical ruxolitinib treatment. One patient experienced reduced itch, improved perifollicular erythema and scale, and hair regrowth after 12 weeks of 1.5% topical ruxolitinib use; another achieved complete itch resolution and significant improvement in erythema and scale after 15 weeks, discontinuing oral treatments while maintaining improvement with topical ruxolitinib alone at a 6-month follow-up [76].
Systemic Treatment
Numerous studies have investigated the efficacy of hydroxychloroquine in treating FFA, both as monotherapy and in combination with other agents [7, 11, 67, 80, 81]. This antimalarial medication is believed to exert its therapeutic effects through its anti-inflammatory properties, particularly its ability to decrease the upregulation of T-cells, which are implicated in the inflammatory process underlying FFA [7]. In a systematic review on 114 patients where 33 received treatment with either hydroxychloroquine or chloroquine, Rácz and colleagues showed a favorable clinical response in 30% of cases and a partial response (PR) in 39% after 6 months of therapy [80]. Vañó-Galván et al. reported disease stabilization in 59% of cases with oral hydroxychloroquine (at a daily dosage between 200 and 400 mg) and hair regrowth in 15% [11]. However, it is important to note that while hydroxychloroquine appears to be effective in managing FFA symptoms, its full therapeutic effects may take up to a year to manifest [82]. Additionally, clinicians must remain vigilant for rare adverse effects such as retinopathy associated with long-term use of hydroxychloroquine [7].
Oral tetracyclines, such as minocycline and doxycycline, have demonstrated some efficacy in treating FFA, primarily due to their anti-inflammatory properties [82]. Strazzulla et al. found that patients treated with doxycycline, tetracycline or minocycline did not show significant differences in outcomes. However, patients had a higher risk of experiencing adverse side effects, including nausea, esophagitis, lightheadedness, photosensitivity, skin eruption and candida infection [73].
5-α-Reductase inhibitors (5α-RI), such as finasteride and dutasteride, have emerged as another promising treatment option for FFA. These medications function by blocking the conversion of testosterone into the stronger androgen hormone dihydrotestosterone, effectively targeting a fundamental pathogenic mechanism in FFA [30]. Studies have consistently shown that a significant percentage of FFA patients treated with 5α-RI experience disease stabilization or symptomatic improvement [11, 29, 47, 77, 81]. Ho and Shapiro reported that treatment with finasteride at doses ranging from 1 to 5 mg per day or dutasteride at 0.5 mg per day resulted in the stabilization of hair loss in 88% (158 out of 180) of patients [81]. Seo et al. conducted a meta-analysis to evaluate the therapeutic efficacy of dutasteride in FFA that included seven studies involving 366 patients who received oral dutasteride. The estimated proportion of patients who experienced stabilization of FFA with oral dutasteride was 0.628 (95% CI: 0.398–0.859). Subgroup analyses on patients who experienced improvement revealed an estimated proportion of improvement of 0.356 (95% CI: 0.163–0.549). The findings of their study suggest that oral dutasteride is a promising treatment option for stabilizing or improving FFA in patients [83].
While other systemic agents, including peroxisome proliferator-activated receptor γ (PPAR-γ) agonists (e.g., pioglitazone), systemic immunosuppressants (e.g., methotrexate, mycophenolate mofetil), oral retinoids, naltrexone, oral minoxidil and platelet-rich plasma (PRP), have shown varying degrees of efficacy in treating FFA, further research is needed to establish their efficacy, safety profiles and optimal dosing regimens definitively [7, 45].
Lastly, as mentioned in the topical strategies, oral JAKis have been proposed to treat FFA refractory to systemic agents. Both oral baricitinib and tofacitinib (5 mg daily) have been employed with encouraging results but larger cohort studies are needed to claim the efficacy of such therapy [8, 76, 79].
Physical Therapies
The excimer laser, emitting 308-nm UV-B light, has been employed to treat various dermatologic conditions such as LPP, AA psoriasis and vitiligo. It is believed to alleviate inflammatory skin disorders by modulating T-cells and cytokines [7]. In a study of 29 FFA patients, Zhang et al. found that excimer laser therapy, along with tCS, tCI and oral hydroxychloroquine, was effective in reducing hair loss [74]. Similarly, Fertig and Tosti observed positive clinical responses to excimer laser treatment in FFA patients when combined with oral hydroxychloroquine, finasteride and tCI [84]. However, there is a lack of additional research on excimer therapy for FFA, highlighting the need for further evidence.
LEDs, particularly super-luminescent diodes, are emerging as a form of photobiomodulation therapy with several dermatologic applications. Although evidence is limited for treating FFA, a pilot study by Gerkowicz et al. showed promising results. Sixteen female patients, including those with FFA and LPP, underwent super-luminescent diode irradiation once a week for 10 weeks as part of combination therapy. The treatment was found to be safe and well tolerated, leading to a significant reduction in LPPAI and FFASS scores, along with an increase in thick hairs. While this study suggests the potential benefits of super luminescent diode therapy as adjunctive treatment for FFA, further studies with larger sample sizes and control groups are necessary to validate these findings [85].
Finally, while hair and eyebrow transplantation presents a potential reconstructive option for FFA patients, its outcomes are variable with an overall temporary result [86, 87]. Thus, careful patient selection and counseling are essential when considering hair transplantation as a treatment option for FFA.
Conclusion
In summary, FFA is a complex dermatologic hair disease with distinct features, including gradual hairline recession and eyebrow loss. Despite ongoing debates about its classification as a variant of LPP, the cicatricial process results from a lymphocyte inflammatory cicatricial infiltrate around the hair follicle. Diagnosis through trichoscopy and/or histopathology remains essential for prompt treatment due to the risk of scarring. As reported cases rise globally, there is a growing need for non-invasive diagnostic tools and innovative therapies. Through this review, we aimed to equip clinicians with updated insights into FFA, fostering improved management and patient outcomes amidst its evolving landscape. Continued research and collaborative efforts are essential to decipher FFA's complexities and advancing clinical care.
Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Kossard S. Postmenopausal frontal fibrosing alopecia. Scarring alopecia in a pattern distribution. Arch Dermatol. 1994;130(6):774.
Kossard S, Lee MS, Wilkinson B. Postmenopausal frontal fibrosing alopecia: a frontal variant of lichen planopilaris. J Am Acad Dermatol. 1997;36(1):8.
Pirmez R, Duque-Estrada B, Abraham LS. It’s not all traction: the pseudo ‘fringe sign’ in frontal fibrosing alopecia. Br J Dermatol. 2015;173(5):1336–8.
Starace M, Orlando G, Iorizzo M, Alessandrini A, Bruni F, Mandel VD, et al. Clinical and dermoscopic approaches to diagnosis of frontal fibrosing alopecia: results from a Multicenter Study of the International Dermoscopy Society. Dermatol Pract Concept. 2022;12(1):2022080.
Guan NN, Fan WX. Diagnostic and therapeutic assessment of frontal fibrosing alopecia. J Clin Dermatol. 2010;39(3):197.
Banka N, Mubki T, Bunagan MJ, McElwee K, Shapiro J. Frontal fibrosing alopecia: a retrospective clinical review of 62 patients with treatment outcome and long-term follow-up. Int J Dermatol. 2014;53(11):1324–30.
Imhof R, Tolkachjov SN. Optimal management of frontal fibrosing alopecia: a practical guide. Clin Cosmet Investig Dermatol. 2020;13:897–910.
Moussa A, Bennett M, Bhoyrul B, Kazmi A, Asfour L, Sinclair RD. Clinical features and treatment outcomes of frontal fibrosing alopecia in men. Int J Dermatol. 2022;61(10):372–4.
Mirmirani P, Tosti A, Goldberg L, Whiting D, Sotoodian B. Frontal fibrosing alopecia: an emerging epidemic. Skin Appendage Disord. 2019;5(2):90–3.
Tziotzios C, Fenton DA, Stefanato CM, McGrath JA. Familial frontal fibrosing alopecia. J Am Acad Dermatol. 2015;73(1):e37. https://doi.org/10.1016/j.jaad.2015.01.057.
Vañó-Galván S, Molina-Ruiz AM, Serrano-Falcón C. Frontal fibrosing alopecia: a multicenter review of 355 patients. J Am Acad Dermatol. 2014;70(4):670–8.
Valesky EM, Maier MD, Kippenberger S, Kaufmann R, Meissner M. Frontal fibrosing alopecia—review of recent case reports and case series in PubMed. Journal der Deutschen Dermatologischen Gesellschaft J Ger Soc Dermatol JDDG. 2018;16(8):992–9.
Pathoulas JT, Flanagan KE, Walker CJ, Collins MS, Ali S, Pupo Wiss IM, et al. A multicenter descriptive analysis of 270 men with frontal fibrosing alopecia and lichen planopilaris in the United States. J Am Acad Dermatol. 2023;88(4):937–9.
Olsen EA, Harries M, Tosti A, Bergfeld W, Blume-Peytavi U, Callender V, et al. Guidelines for clinical trials of frontal fibrosing alopecia: consensus recommendations from the International FFA Cooperative Group (IFFACG. Br J Dermatol. 2021;185(6):1221–31.
Hai J, Meyer SN, Agbai ON. Treatment of frontal fibrosing alopecia in black patients: a systematic review. Cutis. 2023;111(4):186–90.
Kossard S. Frontal fibrosing alopecia, just lichen planopilaris? J Am Acad Dermatol. 2019;81(2): e51.
Photiou L, Nixon RL, Tam M, Green J, Yip L. An update of the pathogenesis of frontal fibrosing alopecia: What does the current evidence tell us? Australas J Dermatol. 2019;60(2):99–104. https://doi.org/10.1111/ajd.12945
Tziotzios C, Stefanato CM, Fenton DA, Simpson MA, McGrath JA. Frontal fibrosing alopecia: reflections and hypotheses on aetiology and pathogenesis. Exp Dermatol. 2016;25(11):847–52.
Starace M, Brandi N, Alessandrini A, Bruni F, Piraccini BM. Frontal fibrosing alopecia: a case series of 65 patients seen in a single Italian centre. J Eur Acad Dermatol Venereol. 2019;33(2):438.
Ranasinghe GC, Piliang MP, Bergfeld WF. Prevalence of hormonal and endocrine dysfunction in patients with lichen planopilaris (LPP): A retrospective data analysis of 168 patients. J Am Acad Dermatol. 2017;76(2):314–20.
Gálvez-Canseco A, Sperling L. Lichen planopilaris and frontal fibrosing alopecia cannot be differentiated by histopathology. J Cutan Pathol. 2018;45(5):313–7.
Miao YJ, Jing J, Du XF, Mao MQ, Yang XS, Lv ZF. Frontal fibrosing alopecia: A review of disease pathogenesis. Front Med. 2022;9: 911944.
Saceda-Corralo D, Ortega-Quijano D, Muñoz-Martín G, Moreno-Arrones ÓM, Pindado-Ortega C, Rayinda T, et al. Genotyping of the rs1800440 polymorphism in CYP1B1 gene and the rs9258883 polymorphism in HLA-B gene in a spanish cohort of 223 patients with frontal fibrosing alopecia. Acta Derm Venereol. 2023;103:9604.
Carpanese MA, Guglielmo A, Alessandrini A, Bruni F, Piraccini B, Starace M. Familial frontal fibrosing alopecia: the first Italian case. Ital J Dermatol Venereol. 2022;157(5):457–8.
Navarro-Belmonte MR, Navarro-López V, Ramírez-Boscà A. Case series of familial frontal fibrosing alopecia and a review of the literature. J Cosmet Dermatol. 2015;14:64–9.
Tziotzios C, Petridis C, Dand N, Ainali C, Saklatvala PJRV. Genome-wide association study in frontal fibrosing alopecia identifies four susceptibility loci including HLA-B*07:02. Nat Commun. 2019;10:1–9.
Bandiera S, Weidlich S, Harth V, Broede P, Ko Y, Friedberg T. Proteasomal degradation of human CYP1B1: effect of the Asn453Ser polymorphism on the post-translational regulation of CYP1B1 expression. Mol Pharmacol. 2005;67:435–43.
Nasiri S, Dadkhahfar S, Mansouri P, Rahmani-Khah E, Mozafari N. Evaluation of serum level of sex hormones in women with frontal fibrosing alopecia in comparison to healthy controls. Dermatol Ther. 2020;33(e13842):13842.
Tosti A, Piraccini BM, Iorizzo M, Misciali C. Frontal fibrosing alopecia in postmenopausal women. J Am Acad Dermatol. 2005;52:55–60.
Murad A, Bergfeld W. 5-alpha-reductase inhibitor treatment for frontal fibrosing alopecia: an evidence-based treatment update. J Eur Acad Dermatol Venereol. 2018;32:1385–90.
Ramos PM, Anzai A, Duque-Estrada B, Farias DC, Melo DF, Mulinari-Brenner F. Risk factors for frontal fibrosing alopecia: a case-control study in a multiracial population. J Am Acad Dermatol. 2021;84:712–8.
Frontal NR, Alopecia F. An example of disrupted aryl hydrocarbon receptor-mediated immunological homeostasis in the skin? Clin Cosmet Investig Dermatol. 2020;13:479–84.
Aldoori N, Dobson K, Holden CR, McDonagh AJ, Harries M, Messenger AG. Frontal fibrosing alopecia: possible association with leave-on facial skin care products and sunscreens; a questionnaire study. Br J Dermatol. 2016;175(4):762–7.
Moreno-Arrones OM, Saceda-Corralo D, Rodrigues-Barata AR, Castellanos-González M, Fernández-Pugnaire MA, Grimalt R, et al. Risk factors associated with frontal fibrosing alopecia: a multicentre case–control study. Clin Exp Dermatol. 2019;44(4):404–10.
Cranwell WC, Sinclair R. Sunscreen and facial skincare products in frontal fibrosing alopecia: a case–control study. Br J Dermatol. 2019;180(4):943–4.
Donati A. Frontal fibrosing alopecia and sunscreens: cause or consequence? Br J Dermatol. 2016;175(4):675–6.
Debroy Kidambi A, Dobson K, Holmes S, Carauna D, Del Marmol V, Vujovic A, et al. Frontal fibrosing alopecia in men: an association with facial moisturizers and sunscreens. Br J Dermatol. 2017;177(1):260–1.
Wang J, Pan L, Wu S, Lu L, Xu Y, Zhu Y, et al. Recent advances on endocrine disrupting effects of UV filters. IJERPH. 2016;13(8):782.
Robinson G, McMichael A, Wang SQ, Lim HW. Sunscreen and frontal fibrosing alopecia: a review. J Am Acad Dermatol. 2020;82(3):723–8.
Thompson CT, Chen ZQ, Kolivras A, Tosti A. Identification of titanium dioxide on the hair shaft of patients with and without frontal fibrosing alopecia: a pilot study of 20 patients. Br J Dermatol. 2019;181(1):216–7.
Harries MJ, Meyer K, Chaudhry I, Kloepper JE, Poblet E, Griffiths CE. Lichen planopilaris is characterized by immune privilege collapse of the hair follicle’s epithelial stem cell niche. J Pathol. 2013;231(236–47):4233.
Karnik P, Tekeste Z, McCormick TS, Gilliam AC, Price VH, Cooper KD. Hair follicle stem cell-specific PPARgamma deletion causes scarring alopecia. J Invest Dermatol. 2009;129:1243–57.
Vañó-Galván S, Saceda-Corralo D, Moreno-Arrones ÓM, Camacho-Martinez FM. Updated diagnostic criteria for frontal fibrosing alopecia. J Am Acad Dermatol. 2018;78(1):21–2.
Rajan A, Rudnicka L, Szepietowski JC, Lallas A, Rokni GR, Grabbe S, et al. Differentiation of frontal fibrosing alopecia and Lichen planopilaris on trichoscopy: a comprehensive review. J Cosmet Dermatol. 2022;21(6):2324–30.
Iorizzo M, Tosti A. Frontal fibrosing alopecia: an update on pathogenesis, diagnosis, and treatment. Am J Clin Dermatol. 2019;20(3):379–90.
Mirmirani P, Zimmerman B. Cocking the eyebrows to find the missing hairline in frontal fibrosing alopecia: a useful clinical maneuver. J Am Acad Dermatol. 2016;75:e63–4.
Moreno-Arrones OM, Saceda-Corralo D, Fonda-Pascual P. Frontal fibrosing alopecia: clinical and prognostic classification. J Eur Acad Dermatol Venereol. 2017;31:1739–45.
Raihan A, Kumaran MS. Lichen planus pigmentosus and frontal fibrosing alopecia: the link explored. Pigment Int. 2018;5:73–7.
Pérez Mesonero R, Pinto Pulido EL, Gómez Zubiaur A, Vélez Velázquez D, Vega Díez D, Rodríguez-Villa Lario A, et al. Occipital involvement in classic frontal fibrosing alopecia: Clinical and trichoscopic cross-sectional study in 17 patients. J Am Acad Dermatol. 2023;89(4):815–7.
Anzai A, Donati A, Valente NY, Romiti R, Tosti A. Isolated eyebrow loss in frontal fibrosing alopecia: relevance of early diagnosis and treatment. Br J Dermatol. 2016;175(5):1099–101.
Starace M, Cedirian S, Alessandrini AM, Bruni F, Quadrelli F, Melo DF, et al. Impact and management of loss of eyebrows and eyelashes. Dermatol Ther. 2023;13(6):1243–53.
Pedrosa AF, Duarte AF, Haneke E, Correia O. Yellow facial papules associated with frontal fibrosing alopecia: A distinct histologic pattern and response to isotretinoin. J Am Acad Dermatol. 2017;77(4):764–6.
Pirmez R, Barreto T, Duque-Estrada B, Quintella DC, Cuzzi T. Facial papules in frontal fibrosing alopecia: beyond vellus hair follicle involvement. Skin Appendage Disord. 2018;4(3):145–9.
Donati A. Facial papules in frontal fibrosing alopecia: evidence of vellus follicle involvement. Arch Dermatol. 2011;147(12):1424.
Pirmez R, Duque-Estrada B, Barreto T, Quintella DC, Cuzzi T. Successful treatment of facial papules in frontal fibrosing alopecia with oral isotretinoin. Skin Appendage Disord. 2017;3(2):111–3.
Cervantes J, Miteva M. Distinct trichoscopic features of the sideburns in frontal fibrosing alopecia compared to the frontotemporal scalp. Skin Appendage Disord. 2018;4(1):50–4.
Tosti A, Miteva M, Torres F. Lonely hair: a clue to the diagnosis of frontal fibrosing alopecia. Arch Dermatol. 2011;147(10):1240.
Waśkiel-Burnat A, Rakowska A, Kurzeja M, Czuwara J, Sikora M, Olszewska M, et al. The value of dermoscopy in diagnosing eyebrow loss in patients with alopecia areata and frontal fibrosing alopecia. J Eur Acad Dermatol Venereol JEADV. 2019;33(1):213–9.
Anzai A, Pirmez R, Vincenzi C, Fabbrocini G, Romiti R, Tosti A. Trichoscopy findings of frontal fibrosing alopecia on the eyebrows: a study of 151 cases. J Am Acad Dermatol. 2021;85(5):1130–4.
Kępińska K, Jałowska M, Bowszyc-Dmochowska M. Frontal Fibrosing Alopecia—a review and a practical guide for clinicians. Ann Agric Environ Mede AAEM. 2022;29(2):169–84.
Doche I, Valente N, Sotto MN, Rivitti-Machado MC, Aoki V, Gerlero P, et al. Correlation of clinical, histopathologic, and direct immunofluorescence findings in lesional and nonlesional scalp of frontal fibrosing alopecia and lichen planopilaris—an observational study. JAAD international. 2023;13:7–9.
Thompson CT, Martinez Velasco MA, Tosti A. Three-dimensional imaging of a peripilar cast and compound follicle in frontal fibrosing alopecia. JAAD Case Rep. 2022;23:46–8.
Ardigò M, Agozzino M, Franceschini C, Donadio C, Abraham LS, Barbieri L, et al. Reflectance confocal microscopy for scarring and non-scarring alopecia real-time assessment. Arch Dermatol Res. 2016;308(5):309–18.
Moreno-Arrones OM, Alfageme F, Alegre A. Ultrasonographic characteristics of frontal fibrosing alopecia. Int J Trichol. 2019;11(4):183–4.
Pirmez R. The dermatoscope in the hair clinic: Trichoscopy of scarring and nonscarring alopecia. J Am Acad Dermatol. 2023;89(2S):9–15.
Miteva M, Tosti A. Hair and scalp dermatoscopy. J Am Acad Dermatol. 2012;67:1040–8.
Samrao A, Chew AL, Price V. Frontal fibrosing alopecia: a clinical review of 36 patients. Br J Dermatol. 2010;163(6):1296–300.
Moreno-Ramírez D, Camacho MF. Frontal fibrosing alopecia: a survey in 16 patients. J Eur Acad Dermatol Venereol. 2005;19(6):700–5.
Holmes S, Ryan T, Young D, Harries M, Hair B, Society N. Frontal Fibrosing Alopecia Severity Index (FFASI): a validated scoring system for assessing frontal fibrosing alopecia. Br J Dermatol. 2016;175:203–7.
Fechine COC, Valente NYS, Romiti R. Lichen planopilaris and frontal fibrosing alopecia: review and update of diagnostic and therapeutic features. An Bras Dermatol. 2022;97(3):348–57.
Saceda-Corralo D, Moreno-Arrones ÓM, Fonda-Pascual P, Pindado-Ortega C, Buendía-Castaño D, Alegre-Sánchez A, et al. Development and validation of the Frontal Fibrosing Alopecia Severity Score. J Am Acad Dermatol. 2018;78(3):522–9.
Mahmoudi H, Rostami A, Tavakolpour S. Oral isotretinoin combined with topical clobetasol 0.05% and tacrolimus 0.1% for the treatment of frontal fibrosing alopecia: a randomized controlled trial. J Dermatolog Treat. 2020;33(1):284–90. https://doi.org/10.1080/09546634.2020.1750553
Strazzulla LC, Avila L, Li X, Lo Sicco K, Shapiro J. Prognosis, treatment, and disease outcomes in frontal fibrosing alopecia: a retrospective review of 92 cases. J Am Acad Dermatol. 2018;78(1):203–5.
Zhang M, Zhang L, Rosman IS, Mann CM. Frontal fibrosing alopecia demographics: a survey of 29 patients. Cutis. 2019;103(2):E16–22.
Chen LC, Ogbutor C, Kelley KJ, Senna MM. Topical tofacitinib for patients with lichen planopilaris and/or frontal fibrosing alopecia [Internet]. J Am Acad Dermatol. 2024. https://doi.org/10.1016/j.jaad.2024.01.060.
Dunn C, Griffith V, Coican A, Dane A, Chow W, Aneja S, et al. Janus kinase inhibition for the treatment of refractory frontal fibrosing alopecia: a case series and review of the literature. JAAD case reports. 2023;40:47–52.
Heppt MV, Letulé V, Laniauskaite I. Frontal fibrosing alopecia: a retrospective analysis of 72 patients from a German Academic Center. Facial Plast Surg. 2018;34(1):88–94.
Samuel C, Cornman H, Kambala A, Kwatra SG. A review on the safety of using JAK inhibitors in dermatology: clinical and laboratory monitoring. Dermatol Therapy. 2023;13(3):729–49.
Plante J, Eason C, Snyder A, Elston D. Tofacitinib in the treatment of lichen planopilaris: a retrospective review. J Am Acad Dermatol. 2020;83(5):1487–9.
Rácz E, Gho C, Moorman PW, Noordhoek Hegt V, Neumann HAM. Treatment of frontal fibrosing alopecia and lichen planopilaris: a systematic review. Acad Dermatol Venereol. 2013;27(12):1461–70.
Ho A, Shapiro J. Medical therapy for frontal fibrosing alopecia: a review and clinical approach. J Am Acad Dermatol. 2019;81(2):568–80. https://doi.org/10.1016/j.jaad.2019.03.079
Gamret AC, Potluri VS, Krishnamurthy K, Fertig RM. Frontal fibrosing alopecia: efficacy of treatment modalities. IJWH. 2019;11:273–85.
Seo HM, Oh SU, Kim S, Park JH, Kim JS. Dutasteride in the treatment of frontal fibrosing alopecia: Systematic review and meta-analysis. J Eur Acad Dermatol Venereol [Internet]. 2024. https://doi.org/10.1111/jdv.19802.
Fertig R, Tosti A. Frontal fibrosing alopecia treatment options. Intractable Rare Dis Res. 2016;5(4):314–5.
Gerkowicz A, Bartosińska J, Wolska-Gawron K, Michalska-Jakubus M, Kwaśny M, Krasowska D. Application of superluminescent diodes (sLED) in the treatment of scarring alopecia—a pilot study. Photodiagnosis Photodyn Ther. 2019;28:195–200.
Audickaite A, Alam M, Jimenez F. Eyebrow hair transplantation in frontal fibrosing alopecia: pitfalls of short- and long-term results. Dermatol Surg. 2019;46(7):922–5. https://doi.org/10.1097/DSS.0000000000002207.
Vañó-Galván S, Villodres E, Pigem R. Hair transplant in frontal fibrosing alopecia: a multicenter review of 51 patients. J Am Acad Dermatol. 2019;81(3):865–6.
Gamret AC, Potluri VS, Krishnamurthy K, Fertig RM. Frontal fibrosing alopecia: efficacy of treatment modalities. Int J Women’s Health. 2019;11:273–85.
Funding
All authors declare that no funds, grants or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Stephano Cedirian and Michela Starace performed the research. Stephano Cedirian, Federico Quadrelli, Francesca Pampaloni, Bianca Maria Piraccini and Michela Starace designed the research study. Francesca Pampaloni, Francesca Bruni, Federico Quadrelli and Luca Rapparini analyzed the data. Stephano Cedirian, Bianca Maria Piraccini and Michela Starace wrote the paper. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of Interest
Michela Starace is an Editorial Board member of Dermatology and Therapy. Michela Starace was not involved in the selection of peer reviewers for the manuscript or in any of the subsequent editorial decisions. Michela Starace has nothing else to disclose. Stephano Cedirian, Luca Rapparini, Francesca Pampaloni, Francesca Bruni, Federico Quadrelli and Bianca Maria Piraccini have nothing to disclose.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Starace, M., Cedirian, S., Rapparini, L. et al. Enhanced Insights into Frontal Fibrosing Alopecia: Advancements in Pathogenesis Understanding and Management Strategies. Dermatol Ther (Heidelb) 14, 1457–1477 (2024). https://doi.org/10.1007/s13555-024-01186-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01186-0