Abstract
Introduction
Drugs and vaccines have been less studied as inducing or aggravating factors for psoriatic arthritis (PsA) compared with psoriasis. Thus, the present study collected and summarized the publications to date about this issue.
Methods
We conducted a systematic literature search through the PubMed, Embase, and Cochrane databases to identify all reports on potential drug- and vaccine-related PsA events until 28 February 2023.
Results
In total, 179 cases from 79 studies were eligible for study. Drugs commonly reported include coronavirus disease 2019 (COVID-19) mRNA vaccines (6 cases), bacillus Calmette–Guerin (BCG) vaccine (3 cases), interferon (18 cases), immune-checkpoint inhibitors (ICI) (19 cases), and biologic disease-modifying antirheumatic drugs (bDMARDs) (127 cases). Drugs causing psoriasis may also induce or aggravate PsA (6 cases). BDMARD-related PsA mostly occurred in a “paradoxical” setting, in which the bDMARDs approved for the treatment of psoriasis induce or aggravate PsA. The reported latency may be delayed up to 2 years. Peripheral arthritis (82.3%) was the most common manifestation of drug- and vaccine-related PsA, followed by dactylitis (29.1%), enthesitis (23.4%), and spondyloarthritis (17.7%).
Conclusions
Drugs and vaccines may be implicated in the aggravation of PsA. Possible mechanisms include cytokine imbalance, immune dysregulation, or inadequate PsA treatment response compared with psoriasis. Most reports are case based without controls, so more studies are needed to further prove the causality. However, early recognition of factors causing or aggravating PsA is important to prevent the irreversible joint damage.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
We conducted a systematic literature search and summarized all potential drug- or vaccine-related psoriatic arthritis (PsA) events, especially coronavirus disease 2019 (COVID-19) and bacillus Calmette–Guerin (BCG) vaccines and interferons, immune-checkpoint inhibitors (ICI), and biologic disease-modifying antirheumatic drugs (DMARDs). |
Peripheral arthritis is the most common manifestation, and the latency may be up to 2 years. |
Drug-induced cytokine imbalance, immune dysregulation, or inadequate PsA treatment response compared with psoriasis may be underlying pathomechanisms. |
Further studies are needed to prove the causality, but physicians should be aware of potential adverse events of these drugs and vaccines. |
Introduction
Psoriasis is a complex immune-mediated inflammatory skin disease that has been associated with multiple comorbidities, especially psoriatic arthritis (PsA). In different studies, the incidence of PsA among patients with psoriasis ranged from 0.27 to 2.7 per 100 person-years [1]. Multiple inducing or aggravating factors for psoriasis have been reported, including drugs, infection, stress, hormonal changes, and more recently, COVID-19 infection and its vaccination [2]. PsA has been associated with the severity of psoriasis, physical traumas, obesity, and smoking [3]. Many reviews on drug-induced or drug-provoked psoriasis exist [4,5,6], but the relationship between drugs and PsA induction or aggravation has been less studied. A systematic review was performed on cases of drug- or vaccine-induced/aggravated PsA and their potential pathomechanisms are discussed.
Methods
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Ethics board approval was waived due to the nature of the study. This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Study Identification
This review used a two-step screening process to identify reports of drug- or vaccine-induced/aggravated PsA published to date for:
Step 1: Two authors screened the Embase, PubMed, and Cochrane databases by using the keywords “psoriatic arthritis” AND “de novo OR onset OR flare OR exacerbation OR aggravation OR induced OR development OR paradoxical” to identify potential drug- and vaccine-related PsA.
Step 2: Articles published from inception to 28 February 2023, using the following search keywords: “psoriatic arthritis” AND “vaccines” OR “specific drugs” reported to aggravate psoriasis in reviews [4,5,6] OR “biologic disease-modifying anti-rheumatic drugs (DMARDs)” were identified.
(Vaccine: SARS-CoV-2 vaccine, bacillus Calmette–Guerin (BCG) vaccine).
(Specific drugs: type 1 interferon, immune checkpoint inhibitors (ICI), beta-blocker, granulocyte colony-stimulating factor (G-CSF), imiquimod, lithium, natalizumab).
(Biologics: adalimumab, brodalumab, dupilumab, efalizumab, etanercept, guselkumab, infliximab, ixekizumab, rituximab, ustekinumab, secukinumab, and risankizumab).
On the basis of the predetermined inclusion criteria, titles/abstracts and full texts of retrieved articles were independently screened by two reviewers. We supplemented our research by manually searching the reference lists to identify additional articles that the initial database search may have missed.
Inclusion Criteria
Cases were included if they were:
-
1.
Reports of new development or increased disease activity of PsA after specific drug treatment or vaccine inoculation
-
2.
Studies that followed an observational study design (i.e., case reports, case series, cross-sectional or cohort studies).
Exclusion Criteria
Studies were excluded if they were:
-
1.
conference abstracts and studies that did not provide individualized patient information
-
2.
reports of patients infected with COVID-19 before vaccination
-
3.
studies conducted using animal models
-
4.
irretrievable full texts both online and through the international article delivery system
The full search strategy is illustrated in Fig. 1, and number of studies extracted through systematic search of each agent is summarized in the Supplementary Material-Table 1.
Data Extraction
Two reviewers independently extracted data on study characteristics (study author, study country, year of publication, sample size), patient demographics information (age, sex, comorbidities, relevant family or personal history of psoriatic disease, previous relevant treatments), information on drugs or vaccines (types, dose, frequency), and characteristics of PsA diagnosis and progression (preexisting or new-onset PsA, peripheral or axial involvement, biologic discontinuation, subsequent treatment, and recurrence or residual side effects).
Data Synthesis and Analysis
Due to considerable heterogeneity among the included studies, meta-analysis was not performed. A descriptive analysis was conducted, and frequencies were represented as percentages (%).
Results
COVID-19 and BCG Vaccines
Vaccine-related PsA was reported in nine patients [7,8,9,10,11,12,13], with six cases occurring after COVID-19 vaccination [7,8,9,10] and three after intravesical BCG vaccination [11,12,13].
Of the six patients experiencing PsA after COVID-19 vaccination, two patients (33.3%) were male, and four patients (66.7%) were female. These patients had a mean age of 43.0 ± 10.5 (range 30–58) years. All patients received two doses of mRNA-based vaccine, and a vaccination history was specified for four patients [7, 8, 10], of whom three (75%) received the BNT162b2 (Pfizer-BioNTech, Germany) vaccine [7, 10] and one (25%) received the mRNA-1273 (Moderna) vaccine [8]. There were five patients (83.3%) experiencing exacerbation of preexisting PsA [7,8,9,10], while one patient (16.7%) experienced new onset of psoriasis and PsA [10]. Among these patients, 66.7% (n = 4/6) had peripheral arthritis [7, 9, 10], 33.3% (n = 2/6) had spondylitis [8, 9], and 16.7% (n = 1/6) had enthesitis [8].
Mean latency period from vaccination to PsA was 6.5 days (range 2–18) days. As for the treatment after the adverse event, only one patient (16.7%) resolved spontaneously in 10 days [9], three patients (50%) received non-steroid antiinflammatory drugs (NSAIDs) or methotrexate [9, 10] (one recovered in 3 days [9], another had persistent symptoms [10], whereas the other patient’s response was not recorded [10]), and two patients (33.3%) improved under ixekizumab [7, 8]).
New onset of PsA after adjuvant intravesical BCG immunotherapy for bladder urothelial carcinoma was reported in three male patients (mean age 70.3 ± 16.3 years, range 52–83 years). The duration between the last immunotherapy and PsA onset ranged from 1 day to 1 year, and two patients were reported to have peripheral arthritis and dactylitis [11, 12]. The symptoms of three patients improved after treatment with different modalities, including NSAIDs and steroid infiltration [11], apremilast [13], and etanercept twice a week [12]. Human leukocyte antigen (HLA)-B27 was negative in two patients receiving HLA typing [11, 12].
Interferon
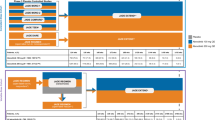
There were 18 cases of PsA reported after interferon immunotherapy [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31], with 14 male (77.8%) and 4 female (22.2%) patients with a mean age of 44.9 ± 11.8 years. Interferon alpha was used for hairy cell leukemia (n = 1) [26], essential thrombocytopenia (n = 1) [14], metastatic renal cell carcinoma (n = 1) [21], or hepatitis C (n = 13) [15,16,17,18,19,20, 22, 23, 27,28,29,30], and interferon beta for relapsing–remitting multiple sclerosis (n = 2) [24, 25]. After interferon therapy, six patients with preexisting psoriasis had new onset of PsA [15, 16, 20, 24, 25, 28], eight patients developed new onset of both psoriasis and PsA [14, 17,18,19, 21,22,23, 30] (five with concurrent onset [14, 17, 21, 23, 30], two with psoriasis first [18, 19], and one with arthritis first [22]), and four patients experienced exacerbation of preexisting PsA [26, 27, 29, 31]. Peripheral arthritis with documented joint involvement was present in all 17 patients, dactylitis in 6 (35.3%) [15, 20,21,22, 25, 30], and axial involvement in 4 (13.8%) [14, 18, 19, 22]. There were two patients (11.1%) who reported arthritis “after” the treatment ended, at 2 weeks and 3 months, respectively [20, 30]. The mean latency was 4.18 ± 5.67 months. It should be noted that two patients with relapsing–remitting multiple sclerosis (RRMS) had tolerated interferon-beta treatment in the past, but re-institution of interferon-beta either after childbearing [24] or a switch to a different brand [25] had likely caused the arthritis. Rechallenge led to aggravation of arthritis in two patients [16, 26], but not in another patient who had been pretreated with etanercept [31]. All patients discontinued interferon therapy. Most patients (15/18, 83.3%) received NSAIDs, conventional synthetic DMARDs (csDMARDs), steroid, or combination therapy, and 10 patients’ PsA improved [14,15,16,17,18,19,20,21,22,23,24,25, 27, 29, 30] (including partial improvement in three patients [16, 24, 30]), while five patients did not improve despite treatment [15, 20, 21, 25, 27].
ICI
There were a total of 19 reported cases of PsA-like adverse events after ICI therapy [32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48]. The mean age was 63.9 ± 10.4 years for the 13 men (68.4%) and 6 women (31.6%). All patients received immunotherapy after progressive disease or intolerance to previous chemotherapy or immunotherapy for different advanced malignancy. The majority of patients received anti-programmed cell death-1(PD-1) monoclonal antibody (nivolumab or pembrolizumab, 17/19, 89.5%), and the other two cases received either darvalumab (an anti-programmed cell death-ligand 1 (PD-L1) monoclonal antibody) [42] or anti-T-cell immunoglobulin and mucin domain-3 (TIM3) antibody [43]. Of these, nine patients (47.4%) had no psoriatic history and developed concurrent psoriasis skin and joint disease [32,33,34, 36, 37, 42, 45, 46], seven patients (36.8%) developed new-onset PsA [35, 38, 39, 41, 43, 47, 48], and three patients (15.8%) experienced exacerbated arthritis events [40, 44, 46]. The mean latency was 21 ± 23.6 (range 3–93) weeks after initiation of immunotherapy. Of the 14 cases with documented sites of arthritis involvement [32, 33, 35, 36, 38,39,40,41,42,43, 46,47,48], all reported peripheral involvement, five patients (35.7%) had dactylitis [35, 42, 46,47,48], and only one patient described spondylitis [47]. Most of the patients (13/19, 68.4%) improved with systemic steroid with or without methotrexate [32,33,34,35,36,37,38, 40, 41, 44, 46]. After failure of steroid therapy, four patients improved with apremilast, guselkumab, secukinumab, or etanercept [39, 42, 47, 48].
Efalizumab
Efalizumab was the first biologic approved in Europe, and then worldwide, to treat psoriasis but it was subsequently withdrawn due to reports of fatal progressive multifocal leukoencephalopathy. A phase II, randomized, placebo-controlled study of PsA [49] showed a numerically higher but statistically insignificant improvement in PsA compared with placebo. However, there were four subsequent reports of new-onset PsA in 30 patients with cutaneous psoriasis who received efalizumab treatment between 2006 and 2011 [50,51,52,53]. The mean age was 45.0 ± 13.2 years, and 18 patients (18/30, 60%) were male and 12 patients (12/30, 40%) were female. When HLA-B27 typing was performed in 86.7% (26/30) of the patients, eight patients (8/26, 30.8%) tested positive [51, 52]. All but 1 patient (29/30, 96.7%) reported peripheral arthritis [50,51,52,53], 8 patients (8/30, 26.7%) reported spinal involvement [52], 16 patients (16/30, 53.3%) had concurrent enthesitis [52, 53], and 12 patients (12/30, 40.0%) developed dactylitis [50, 52, 53].
A review of three studies, including 19 patients, recorded a mean latency of 17.1 ± 14.2 (range 1–48) weeks between treatment initiation and onset of arthritis [50,51,52]. After discontinuation of efalizumab, arthritis resolved spontaneously in one patient (1/19, 5.3%), whereas five other patients (5/19, 26.3%) achieved at least partial improvement of arthritis under NSAIDs [52]. Still another eight patients (8/19, 42.1%) experienced symptom relief after efalizumab suspension and subsequent treatment with csDMARDs alone or in combination with NSAIDs [52]. The remaining (5/19, 26.3%) required biologic treatment to alleviate arthritis [50,51,52].
Colina et al. reported another 11 cases in which arthritis developed after efalizumab treatment (on average 29 weeks) was discontinued [53]. The mean time to arthritis onset was 13.6 ± 2.98 weeks after efalizumab withdrawal, but treatment and outcomes were not recorded in the study.
Key information of vaccine- and specific drug-related PsA is summarized in Table 1, and the latency of each group is illustrated in Fig. 2.
Other Biologic DMARDs
In our review, 23 studies, including 94 patients, had reported either de novo PsA or acute exacerbation thereof after FDA-approved biologic treatments for psoriatic disease [54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80]. These events were classified as “paradoxical psoriatic arthritis (PPsA).” The age of these patients was 49.7 ± 13.5 years, ranging from 18 years to 85 years, and the male to female ratio was 1.21:1 (51M:42F). More than half (48/94, 51.1%) of these events involved ustekinumab treatment [57, 60, 62, 63, 65,66,67, 72, 74, 75, 77, 78], and tumor necrosis factor (TNF)-inhibitors accounted for another 39.4% (37/94) [56, 58, 61, 65, 66, 68, 70, 72, 79, 80]. The remaining 10% belong to five novel biologics that target the Th17/IL-23 pathway [64, 71,72,73, 76]. Most patients received biologic treatment for long-standing psoriasis, and only three patients with no personal history of psoriasis developed psoriasis and PsA after TNF-inhibitors treatment [56, 58, 79]. There were 41 patients (41/94, 43.6%) who were bio-naïve [60, 62, 65, 67, 75], and others had received at least one TNF-inhibitor previously and switched biologics due to unsatisfactory psoriasis control.
Ustekinumab
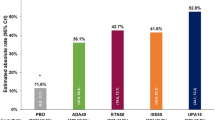
Of the 48 patients who were reported PPsA after ustekinumab, 30 patients (62.5%) had new-onset arthralgia [57, 60, 62, 65,66,67, 72, 75, 78], while the other 18 patients (37.5%) had exacerbation of preexisting arthritis [57, 62, 63, 67, 74, 77]. Most of the patients had peripheral arthritis (34/39, 87.2%) [57, 60, 62, 63, 66, 67, 74, 75, 77], followed by dactylitis (8/39, 20.5%) [60, 62, 63, 75], enthesitis (8/39, 20.5%) [62, 63, 75], and spondylitis (5/39, 12.8%) [63, 66, 75, 78]. The mean latency for the 44 cases that reported the exact time to arthritis onset was 11.42 ± 11.45 months (range 3 days to 4 years). We divided the patients into three groups: 18 patients experiencing PsA exacerbation [57, 62, 63, 67, 74, 77], 13 bio-naïve patients with psoriasis [60, 62, 65, 67, 75], and 17 patients pretreated with TNF-inhibitors [57, 62, 65, 66, 72, 75, 78]. After grouping, mean latency was 7.42 ± 8.93 months in the PsA exacerbation group, 18.2 ± 14.4 months in the bio-naïve group, and 10.2 ± 8.59 months in the TNF-inhibitors pretreated group. Latencies of the latter two groups did not differ significantly (p = 0.0977).
About 50% of the patients required a biologic switch for arthritis treatment, and TNF-inhibitors were the most common alternative [57, 62, 63, 67, 74, 77, 78]. Some patients did not discontinue ustekinumab because of satisfactory improvement in psoriasis and relatively mild activity of arthritic disease [57, 62, 63, 65, 72, 74].
TNF-Inhibitors
There were 37 patients who reported new-onset PsA after TNF-inhibitors, with 20 cases after adalimumab [61, 65, 66, 68, 70, 72, 79], 10 cases after etanercept [58, 65, 66, 72], and 7 cases after infliximab [56, 65, 66, 70, 80]. Notably, three patients who received TNF-inhibitors had no history of psoriasis, but developed both psoriasis and PsA. Instead, they had a history of Crohn’s disease [56], rheumatoid arthritis [58], or hidradenitis suppurativa [79]. Mean latency for 19 patients was 15.34 ± 14.75 months (range from after first infusion to 43 months) [56, 58, 61, 65, 68, 70, 72, 79, 80]. Peripheral arthritis occurred in more than 50% of the patients (14/27) [56, 58, 61, 66, 68, 70, 80], spondylitis in 13.8% (4/27) [66, 68, 79], dactylitis in 29.6% (8/27) [61, 66, 70], and enthesitis in 25.9% (7/27) [66]. Most patients (23/37, 62.2%) required a biologic switch for arthritis control [58, 61, 65, 66, 68, 70, 72, 79, 80], and some patients (6/37, 16.2%) chose to continue biologics with add-on methotrexate [66].
Th17/IL-23 Pathway Inhibitors
PsA transpired after Th17/IL-23 pathway inhibitors, including brodalumab, ixekizumab, secukinumab, risankizumab, and guselkumab, in nine patients (Table 2). The mean age of the six male and three female patients was 50 ± 10.1 years. The latency period after introduction ranged from immediately after the first dose of biologics to 48 months afterward. Half of these patients continued biologic treatment with methotrexate or steroid for arthritis control [72], and three patients switched biologics [64, 73, 76].
Key information of paradoxical PsA (PPsA) (except efalizumab) is summarized in Table 3, the latency to PPsA event is illustrated in Fig. 3, and the composition of each biologic agent is illustrated in Fig. 4.
Rituximab
De novo psoriasis and PsA developed in three female patients after two doses of rituximab for rheumatoid arthritis [54], lymphoma [55], and granulomatosis with polyangiitis [59], respectively. One patient was 17 years old, and the other two patients were both 66 years old. The latency period ranged between 6 weeks to 6 months.
Miscellaneous
Skerritt et al. reported a case of de novo psoriasis with arthropathy after 24 months of lithium treatment [81]; Macfarlane et al. and Tatu et al. reported two cases of new onset psoriasis and PsA after receiving oxyprenolol and metoprolol for blood pressure control [82, 83]. Kavanaugh et al. reported a patient with psoriatic disease who experienced periodic exacerbation after G-CSF institution with relief when neutropenic [84]. Nakamura et al. reported a case of PsA aggravation after topical imiquimod for 4 weeks; symptoms improved after discontinuation and worsened again after rechallenge [85]. Vacchiano et al. described a patient with multiple sclerosis and a family history of psoriasis who developed psoriasis and PsA after 19 infusions of natalizumab, a monoclonal antibody that binds to alpha-4 integrin receptors on endothelial cells lining blood vessels [86].
All PsA events for which the manifestation was specified are summarized in Table 3.
In our review, 37 patients (20.7%) had exacerbation of preexisting PsA [7,8,9,10, 26, 27, 29, 31, 40, 44, 46, 52, 57, 62,63,64, 67, 71, 74, 77, 84, 85], 113 patients (63.1%) had new-onset PsA [11, 13, 15, 16, 20, 24, 25, 28, 35, 38, 39, 41, 43, 47, 48, 50,51,52,53, 57, 60,61,62, 65,66,67,68, 70, 72, 73, 75, 76, 78, 80], 28 patients (15.6%) had de novo psoriatic skin and joint disease [10, 12, 14, 17,18,19, 21,22,23, 30, 32,33,34, 36, 37, 42, 45, 46, 54,55,56, 58, 79, 81,82,83, 86], and 1 patient had PsA without psoriasis [59]. Of the 28 patients developing concurrent psoriasis and PsA, PsA symptoms preceded psoriasis in 1 patient (3.6%) [22], while 7 patients (25%) reported psoriasis before PsA onset [18, 19, 45, 79, 81, 82, 86]. The remaining 20 patients (71.4%) reported simultaneous onset of both psoriasis and PsA [10, 12, 14, 17, 21, 23, 30, 32,33,34, 36, 37, 42, 46, 54,55,56, 58, 83].
Discussion
In total, 8–40% of patients with psoriasis have comorbid PsA, depending on the classification criteria and study methods [87]. Moreover, up to 80% of these patients develop psoriasis before joint manifestations [88] and 5–10% of patients develop concurrent skin and joint symptoms, while only 5–20% of patients manifest arthropathy before psoriasis [89,90,91]. The evolution from psoriasis to PsA usually takes 10 years [92], and the risk of PsA increased linearly over time in the Utah Psoriasis Initiative study. However, another study in European dermatology clinics found a constant risk of PsA at 74 per 1000 person-years, following the initial diagnosis of psoriasis [93]. Nevertheless, under-diagnosis of PsA and dactylitis [94] is common in clinical practice [95]. Several factors have been identified for the transition from preclinical to subclinical and then clinical PsA, including trauma, comorbidities, microbiome-related factors, biomechanical stress [96], and infections that required antibiotics [97]. Drugs and vaccines were generally not listed as factors for the transition except NSAIDs and acetaminophen. In a large long-term prospective study among US women, regular use of NSAIDs and acetaminophen was correlated with a higher risk of developing PsA, even after multivariate adjustment [98]; this may be explained by their use in subclinical PsA symptoms, and not as true inducers of PsA. Rubella vaccination was found to be associated with an increased risk [odds ratio (OR) 12.4] of developing inflammatory arthritis in psoriasis patients [99], but the confidence interval was too large (1.2–122), and another study showed conflicting results [97]. The paucity of reports of drug-induced or aggravated PsA is interesting, since drug-induced or aggravated psoriasis is common, and psoriasis and PsA are two closely related diseases often grouped under the umbrella category of psoriatic disease. In fact, several reviews of drug-induced psoriasis have been published [4,5,6], but there is currently no review of drug-induced PsA.
Although only case series and case reports exist, it is worthwhile to consider the possibility of drugs and vaccines as inducers and aggravators of PsA and to investigate their potential underlying pathogenesis. The exact mechanism of PsA remains incompletely understood, but there are essentially two major hypotheses. One theory considers PsA as an entheseal-based, autoinflammatory disease that may be aggravated by local trauma [100]. The other theory considers PsA as an immune-mediated disease driven by HLA-class 1 antigens presenting unidentified peptides to CD8+ T cells [101, 102]. However, these two hypotheses are not mutually exclusive, since different immune cells are known to be present in the entheses. In drug- or vaccine-induced/aggravated PsA, an immunological mechanism is more likely, and most cases in this review involved drugs, mostly biologics, which act primarily through immune-modulation or stimulation. In addition, the current COVID-19 pandemic and the use of COVID-19 vaccination offers a unique opportunity to see the aggravation or de novo development of many immune-mediated diseases, including psoriasis and PsA [103,104,105,106].
Recently, a national retrospective cohort study of consecutive rheumatic disease in Singapore found that patients with inflammatory arthritis and with active disease had a higher flare rate after at least 1 dose of COVID-19 mRNA vaccination, while age greater than 53 years serves as a protective factor [107].
In the present review, all COVID-19-vaccine-induced new onset of PsA were caused by mRNA vaccines. No protein subunit vaccine has been reported, suggesting that viral mRNA might be central for triggering PsA. Alternatively, it may reflect higher usage of mRNA vaccines in the reporting countries. Interestingly, LL-37 complexed with endogenous or exogenous DNA and RNA is thought to initiate disease exacerbation via plasmacytoid dendritic cells (pDCs) in psoriasis [108, 109]. In PsA, the roles of DNA and RNA were less well studied. To unveil the pathogenesis of COVID-19-induced PsA, Qiao Zhou et al. investigated spinal entheseal pDCs in vitro, simulating the environment after viral infection [110]. Toll-like receptor (TLR)-7 and TLR-9 of pDCs were activated through CpG oligodeoxynucleotides (ODN) and significant production of TNF and interferon-alpha was observed. The study emphasizes the role of human entheseal pDCs in the pathogenesis of PsA.
Additionally, we also discovered that BCG vaccination was reported to cause PsA. Intravesical instillation of the BCG vaccine could reduce recurrence more effectively than surgery in patients with superficial bladder cancer [111]. The exact mechanism for the tumoricidal effect remains unknown, but two immunological effects have been discovered [112]. “Trained immunity” refers to the short-term effects mediated by epigenetic reprogramming and metabolic remodeling of innate immune cells [113], while “heterologous Th1/Th17 immunity” is the prominent long-term effect of BCG vaccination on the immune system that persists at least 1 year after vaccination. The key players of trained immunity are monocytes, macrophages, and natural killer (NK) cells, but the process occurs independently of B and T cells [114]. In fact, immunization with heat-inactivated M. tuberculosis promotes the onset of spondylitis and arthritis in a HLA-B27/Huβ2m transgenic rat model [115]. Collectively, “trained immunity” likely initiated PsA in these patients receiving BCG vaccination.
In addition to vaccination, exogenous type 1 interferon may trigger PsA through its immune-modulatory effect. In addition, imiquimod is a potent TLR 7 agonist, which induces type 1 interferon production and causes polarization toward a Th1 immune response [116].
The incidence of overt autoimmune disease after interferon treatment was 3% [117]. Previous studies reported that type 1 interferon treatment may either induce de novo psoriasis or exacerbate preexisting psoriasis [118, 119]. Interferon-α is an important mediator of cutaneous pDCs in psoriasis development [120]. It links innate and adaptive immunity by promoting the differentiation of circulating monocytes into dendritic cells [121]. Dermal dendritic cells has potent autoinflammatory capacity to support the immune response to resting peripheral blood T cells and activate Th1 type cytokines [122], leading to T cell activation and proliferation. In patients with psoriasis, the response to interferon-α was intensified in T cells [123], which probably induced PsA formation.
ICI block the inhibitory co-receptor and augment Th1 and Th17 responses in patients with advanced cancers, which could be correlated with anti-tumor effect [124] and PsA induction or aggravation. Inflammatory arthritis in this setting often presents symmetrically in association with synovitis, but serological markers for rheumatoid arthritis were usually negative [125]. Additionally, both CD8 and CD4 T cells in peripheral blood express significantly reduced PD-1 but increased proinflammatory cytokines compared with the control group [126].
Patients with psoriasis, both with and without arthritis, were found to have significantly lower expression of PD-1+ on peripheral CD4 and CD8 T cells, a finding consistent with the study of inflammatory arthritis after ICI treatment [127]. Differential PD-1 expression in T cells suggests that impaired inhibitory co-receptor, PD-1, expression on effector T lymphocytes is related to PsA. Although PsA after ICI is rare, these studies can provide an immunological basis for ICI-induced PsA.
Arthritis occurring after dupilumab treatment constitutes another scenario of drug-induced PsA. Dupilumab has been approved for the treatment of several Th2-mediated inflammatory diseases, including atopic dermatitis, asthma, prurigo nodularis, etc. It acts through binding the alpha subunit of IL-4 receptor, which blocks the signaling of both IL-4 and IL-13. Dupilumab-induced psoriasis [128] and seronegative inflammatory arthritis/enthesitis [129] have been increasingly reported, presumably due to skewed cytokine imbalance toward Th17/IL-23 axis by removing the inhibitory function of IL-4, which serves as a brake on Th17 pathway [130,131,132]. Nevertheless, the characteristics of arthritis were not detailed in these reports [133].
Biologic DMARDs (bDMARDs) are the most common drugs causing PsA in this review (127/179, 70.9%). Most patients experienced PsA exacerbation despite improvement of psoriasis during efalizumab treatment. Although efalizumab is already withdrawn from the market, the understanding of its role in triggering PsA is important for our understanding of drug-induced PsA. The discrepant response between skin and joints suggests insufficient efficacy of efalizumab on PsA control. Alternatively, it may be related to the occurrence of transient papular psoriasis noted during efalizumab [134] use, suggesting uncontrolled efflux of inflammatory cells into the skin or joints. HLA-B27 typing yielded a 30.8% positive rate in our review, but it is unknown whether other genetic predisposition or prior (or concurrent) local trauma may be present in such cases.
In general, current biologics for psoriasis are also approved for PsA. Although the use of these biologics in patients with psoriasis has been reported to delay the occurrence of new-onset PsA [135], with IL-12/IL-23 or IL-23 inhibitors showing the best effects, many biologics, especially ustekinumab (an IL-12/IL-23 inhibitor), were reported to cause “paradoxical” PsA.
We discovered 94 reported cases of paradoxical PsA caused by biologics with different modes of actions, including all classes of biologics currently in use for psoriasis, i.e., anti-TNF (adalimumab, etanercept, infliximab), anti IL-12p40 (ustekinumab), anti IL-17 (brodalumab, ixekizumab, secukinumab), and anti IL-23p19 (guselkumab, risankizumab) agents.
Two mechanisms have been proposed:
1. “Cytokine imbalance” between TNF and interferon:
Skin biopsies from paradoxical psoriasis were characterized by overproduction of type 1 interferons and dermal accumulation of pDCs but reduced number of T cells, distinct from classical psoriasis [136]. TNF-inhibitor treatment inhibited pDC maturation, which resulted in prolonged and excessive type 1 interferon production from pDCs. The resulting aberrant innate autoinflammatory reaction was proposed to cause paradoxical psoriasis [137], and in turn, PsA. Invigorated production of interferon might account for the three patients who experienced de novo psoriasis and PsA induction after TNF-inhibitors, as the patients developed PsA after exogenous interferon therapy. However, this theory is not currently favored, as PsA induction occurs in biologic treatment blocking different axes (anti-IL-12/23, anti-Th17/IL-23) and experimental evidence remains lacking [72].
2. Less satisfactory and incomplete response to biologic therapy for PsA compared with psoriasis:
2.1 “Unmasking” effect revealing subclinical arthritis controlled by previous treatments [74]:
In one recent study, both IL-12/23 blockers and IL-23 blockers are associated with longer time to incident inflammatory arthritis compared with TNF-inhibitors and IL-17 blockers [135]. However, in the present review, more than 50% of paradoxical PsA events occurred after treatment with ustekinumab. We suspect it is because ustekinumab is the least effective biologic approved for PsA [138], and thus ustekinumab was listed as the second-line treatment of PsA in both GRAPPA and EULAR [139]. To evaluate the unmasking effect, we classified the patients who developed new-onset arthritis after receiving ustekinumab treatment into a bio-naïve group (n = 13) and a group pretreated with TNF-inhibitors (n = 14). Indeed, mean latency to the onset of arthritis was shorter in the pretreated group, but the difference did not reach statistical significance (p = 0.0977).
2.2 Residual local inflammation after biologic treatment:
Treating psoriasis with biologics does reduce but does not completely eliminate the risk of PsA development. In fact, the risk of new-onset PsA during biologic treatment was 6.7–9.4% in different studies [66, 72, 140].
TNF-blockade rapidly and effectively extinguishes inflammation in PsA joints by down-regulating effector macrophages, expression of CD3+ T lymphocytes, and metalloproteinase (MMP)-3,9 [141,142,143]. Nevertheless, IL-17 expression persists in synovial tissue after adalimumab treatment despite clinical resolution of symptoms [144]. Similarly, when Fiechter et al. sampled psoriatic synovium in 11 patients receiving ustekinumab, significant decreases in sublining CD68+ macrophages and MMP-3 were observed at week 12. However, only IL-23p19 was significantly decreased, and other proinflammatory cytokines, including IL-17A, IL-17F, TNF, IL-6, and IL-8, were not significantly down-regulated. Instead, the results revealed ustekinumab could have modulated the MAPK-ERK pathway at the transcriptional level, modifying the Wnt pathway and potentially also the PI3K-AKT-mTOR pathway. In addition, secukinumab also significantly suppressed synovial CD68+ macrophages and MMP-3 expression but not TNF mRNA expression in 20 patients with active peripheral spondyloarthritis [145].
Taken together, the inflammatory milieu in psoriatic joints is complex, and the therapeutic pathways of different biologics ultimately share a final common pathway involving effector macrophage and MMP-3 down-regulation. Indeed, MMP-3 was shown to correlate with PsA disease activity [146].
Residual tissue-resident effector cells, especially mast cells, may be implicated in the pathogenesis of “paradoxical PsA.” Noordenbos et al. reported that abundant IL-17-expressing mast cells constituted a major reservoir (up to 70%) of IL-17-expressing cells infiltrating the synovial lining of PsA, and these cells were not modulated by TNF blockade [147]. As mentioned earlier, ustekinumab does not affect IL-17 expression. IL-17A-positive mast cells in synovial tissue were also increased after successful inhibition of IL-17A with secukinumab and correlated inversely with inflammation. These tissue mast cells may act as sentinel cells, storing IL-17 under homeostatic conditions and later releasing these cytokines in response to inflammatory stimuli [148].
In conclusion, targeting inflammatory molecules may be sufficient to resolve clinical symptoms in patients with psoriasis, but fails to completely reverse the complex inflammatory milieu on the cellular and molecular levels.
Beta-blockers and lithium are among the best-known drugs to initiate or exacerbate psoriasis [149]. Beta-blockers inactivate the adenyl cyclase, thereby decreasing intracellular cyclic adenosine monophosphate (cAMP) and calcium levels [150], which impairs differentiation and promotes proliferation of keratinocytes [151]. Lithium was proposed to reduce intracellular calcium and cause psoriasis through similar mechanisms [152] in earlier studies. However, lithium-provoked psoriasis is reversed by inositol supplementation, supporting the “inositol depletion hypothesis” that lithium actually blocked the phosphatidyl inositol pathway and depleted intracellular calcium therein [153]. Moreover, both drugs increase the protein tyrosine kinase activity in psoriatic T cells [154], implying a potential immunomodulatory effect. In our study, all three patients receiving either oxprenolol, metoprolol, or lithium developed de novo psoriasis and PsA; this suggests that an overall activation of psoriatic T cell may be pathogenic.
The potential pathomechanisms of drug- or vaccine-induced/aggravated PsA are summarized in Fig. 5.
Mean latency was about 1 week in COVID-19-vaccine-related PsA, and 4–5 months in drug (other than bDMARDs)-related PsA. BDMARDs (except efalizumab) took the longest time to provoke PsA, 1 year on average. Brown et al.’s systematic analysis of TNF-inhibitor-induced psoriasis found a mean latency (14.0 months) [155] slightly shorter than that of our TNF-inhibitor-induced PsA group (15.3 months). All except two patients with drug- or vaccine-induced PsA events occurred in the first year after treatment, while 82% (59/72) of new onset paradoxical PsA events occurred within the 2 years following a new biologic drug introduction. The long latency from exposure to PsA is not unexpected, since there is also a long latency between some drug exposure to psoriasis onset [156], and the long latency from psoriasis to PsA is also typical. For example, the latency from drug ingestion to psoriasis induction was 10 months for lithium [157], 3 days to 12 months for beta-blockers [158], and 4–12 weeks for antimalarials [159]. Peripheral arthritis was present in 82.3% of patients with drug-related arthritis, followed by dactylitis (29.1%) as the second most common manifestation. The rate of dactylitis was lower than traditional dactylitis prevalence in psoriatic patients (39%) [160]. Enthesitis was seen in 23.4% of the patients, and the least common manifestation was psoriatic spondyloarthritis (17.7%). Only the ICI and efalizumab subgroup reported a higher frequency (35.7–36.7%) of dactylitis, and more than half of the patients (53.3%) in the former group had enthesitis.
PsA is an inflammatory arthritis often associated with psoriasis. In our review, cardinal symptoms were extracted from the included articles, but a specific diagnosis for PsA is often difficult due to the lack of pathognomic features, except for the presence dactylitis and arthritis mutilans. Thus the diagnosis/classification of PsA relies heavily on the presence of psoriasis, and the diagnosis of PsA in patients with recent onset of inflammatory arthritis following drugs or vaccines may be overlooked in the absence of psoriasis. The most appreciated criteria, which was also utilized in the present review, for the “diagnosis” of PsA is the CASPAR classification due to its relative high sensitivity and specificity. However, the sensitivity and specificity of CASPAR criteria in drug-induced PsA are unknown and deserve further investigation.
Another limitation comes from the possibility of “hidden” PsA cases among reports of drug-induced/aggravated psoriasis, although we have tried to search for such reports and reviewed the full texts to find reports of PsA. Early inflammatory cells or cytokines in PsA were believed to originate from the skin or sometimes the gut [161]. Because most PsA occurs after the occurrence of psoriasis, it is difficult to differentiate between drugs induced versus naturally occurring PsA in patients who already have psoriasis.
We are also limited by evidence quality and case quantity. Reports of drug-induced PsA are rare, and most were published only as case reports or case series. Heterogeneous data and small sample size constrain the generalizability of findings. Infrequent rechallenges and long mean latency time in the bDMARD group make it difficult to draw convincing conclusions about causality between drugs and PsA events.
Conclusions
PsA is closely related to psoriasis, but compared with psoriasis, drugs or vaccines as triggers and aggravators for PsA are less studied. It is in part because most patients with PsA are preceded by psoriasis with a delay of 8–10 years on average, and the causes of PsA are often unknown. Thus, the diagnosis of drug- or vaccine-induced/aggravated PsA is often difficult. In addition, the diagnosis of PsA in the absence of psoriasis is difficult, with the exception of dactylitis, which is somewhat pathognomic after exclusion of an infectious process. In this review, biologics and COVID-19 mRNA vaccines are the most common culprits that support the immune dysregulation as one cause of PsA. Drugs causing psoriasis, such as lithium and beta-blockers, topical imiquimod, G-CSF, and natalizumab, may also induce or aggravate PsA. The reported latency may be delayed up to 2 years. The long latency between drug exposure and onset of psoriasis and/or PsA makes a definitive causal relationship conclusion difficult. For the possible mechanism, drug-induced cytokine imbalance, immune dysregulation, or inadequate PsA treatment response of preclinical/subclinical PsA might be possible underlying pathomechanisms, but the exact mechanism is unknown due to the unavailability of tissue biopsies from the affected site in drug-related PsA. More studies are definitely needed for our understanding of the condition. However, early recognition of factors causing or aggravating PsA is important to prevent the irreversible joint damage.
Data Availability
This article is based on previously conducted studies. Searching strategies are specified in the Methods section. All data generated or analyzed during this study are included in this published article/as supplementary information files. Figure 5 was created with Biorender.com.
References
Alinaghi F, Calov M, Kristensen LE, et al. Prevalence of psoriatic arthritis in patients with psoriasis: a systematic review and meta-analysis of observational and clinical studies. J Am Acad Dermatol. 2019;80(1):251–65 (e19).
Chao J-P, Tsai T-F. Psoriasis flare following ChAdOx1-S/nCoV-19 vaccination in patients with psoriasis under biologic treatment. Dermatol Sin. 2021;39(4):208–9.
Zabotti A, De Lucia O, Sakellariou G, et al. Predictors, risk factors, and incidence rates of psoriatic arthritis development in psoriasis patients: a systematic literature review and meta-analysis. Rheumatol Ther. 2021;8(4):1519–34.
Tsankov N, Angelova I, Kazandjieva J. Drug-induced psoriasis. Recognition and management. Am J Clin Dermatol. 2000;1(3):159–65.
Grzelj J, Sollner DM. The role of xenobiotics in triggering psoriasis. Arch Toxicol. 2020;94(12):3959–82.
Balak DM, Hajdarbegovic E. Drug-induced psoriasis: clinical perspectives. Psoriasis (Auckl). 2017;7:87–94.
Watabe D, Tsunoda K, Amano H. Psoriatic arthritis flare-up after a second dose of BNT162b2 COVID-19 mRNA vaccine. Eur J Dermatol. 2022;32(3):420–1.
Ohmura S, Hanai S, Ishihara R, et al. A case of psoriatic spondyloarthritis exacerbation triggered by COVID-19 messenger RNA vaccine. J Eur Acad Dermatol Venereol. 2022;36(6):e427–9.
Spinelli FR, Favalli EG, Garufi C, et al. Low frequency of disease flare in patients with rheumatic musculoskeletal diseases who received SARS-CoV-2 mRNA vaccine. Arthritis Res Ther. 2022;24(1):21.
Stander S, Zirpel H, Bujoreanu F, et al. Case report: Clinical features of COVID-19 vaccine-induced exacerbation of psoriasis—a case series and mini review. Front Med (Lausanne). 2022;9: 995150.
Queiro R, Ballina J, Weruaga A, et al. Psoriatic arthropathy after BCG immunotherapy for bladder carcinoma. Br J Rheumatol. 1995;34(11):1097.
Dudelzak J, Curtis AR, Sheehan DJ, Lesher JL Jr. New-onset psoriasis and psoriatic arthritis in a patient treated with Bacillus Calmette-Guerin (BCG) immunotherapy. J Drugs Dermatol. 2008;7(7):684.
Tampouratzi E, Katsantonis J. Psoriatic arthritis induced by intravesical bacillus calmette-guerin infusion for bladder cancer : successfull treatment with apremilast 2019; 24th world congress of dermatology]. https://www.wcd2019milan-dl.org/abstract-book/documents/abstracts/35-psoriasis/psoriatic-arthritis-induced-by-intravesical-626.pdf. Assessed 15 Mar 2023.
Jucgla A, Marcoval J, Curco N, Servitje O. Psoriasis with articular involvement induced by interferon alfa. Arch Dermatol. 1991;127(6):910–1.
Berge E, Laurent-Puig P, Clerc D, et al. Psoriatic arthritis in a patient treated with interferon alpha. Rev Rhum Ed Fr. 1993;60(1):77.
Makino Y, Tanaka H, Nakamura K, et al. Arthritis in a patient with psoriasis after interferon-alpha therapy for chronic hepatitis C. J Rheumatol. 1994;21(9):1771–2.
Piqueras Alcol B, de Diego LA, Perez Roldan F, et al. Interferon-alpha and development of psoriatic arthropathy. An Med Interna. 1996;13(10):512–3.
Lombardini F, Taglione E, Riente L, Pasero G. Psoriatic arthritis with spinal involvement in a patient receiving alpha-interferon for chronic hepatitis C. Scand J Rheumatol. 1997;26(1):58–60.
Germain P, Delbrel X, Traissac T, et al. Psoriatic rheumatism in a patient taking interferon alpha, reversible with treatment cessation. Presse Med. 1999;28(40):2226–7.
Olivieri I, Padula A, Ciancio G, La Civita L. Psoriatic arthritis and alpha-interferon. Scand J Rheumatol. 2001;30(3):175.
Gaal J, Kalmanchey J, Szegedi A, et al. Interferon induced psoriatic arthritis in a patient with renal cancer. Orv Hetil. 2005;146(49):2503–6.
Oliveira TL, Caetano AZ, Belem JM, et al. Interferon-alpha induced psoriatic arthritis and autoimmune hemolytic anemia during chronic hepatitis C treatment. Acta Reumatol Port. 2014;39(4):327–30.
Rodrigues-Pinto E, Cardoso H, Pimenta S, Macedo G. The clinical significance of autoantibodies in hepatitis C patients submitted to interferon treatment. GE Port J Gastroenterol. 2015;22(1):24–7.
La Mantia L, Capsoni F. Psoriasis during interferon beta treatment for multiple sclerosis. Neurol Sci. 2010;31(3):337–9.
Toussirot E, Bereau M, Bossert M, et al. Occurrence of psoriatic arthritis during interferon beta 1a treatment for multiple sclerosis. Case Rep Rheumatol. 2014;2014: 949317.
Kusec R, Ostojic S, Planinc-Peraica A, et al. Exacerbation of psoriasis after treatment with alpha-interferon. Dermatologica. 1990;181(2):170.
Spadaro A, Riccieri V, Nisi A, Taccari E. IGM rheumatoid factor occurrence with recurrence of polyarthritis and psoriasis after treatment with interferon alpha. Rev Rhum Ed Fr. 1993;60(5):376–7.
Georgetson MJ, Yarze JC, Lalos AT, et al. Exacerbation of psoriasis due to interferon-alpha treatment of chronic active hepatitis. Am J Gastroenterol. 1993;88(10):1756–8.
Malipeddi AS, Kallarackal G. Exacerbation of psoriasis and psoriatic arthritis induced by interferon therapy. Rheumatology. 2008;47:Ii29–30.
Singh DK, Badwal JS, Demello D. Drug-induced psoriatic arthritis—Rare presentation after hepatitis C treatment. SGIM National Meeting; 2015.
Mederacke I, Witte T, Wedemeyer H, Meyer-Olson D. Successful clearance of hepatitis C virus with pegylated interferon alpha-2a and ribavirin in an etanercept-treated patient with psoriatic arthritis, hepatitis B virus coinfection and latent tuberculosis. Ann Rheum Dis. 2011;70(7):1343–4.
Law-Ping-Man S, Martin A, Briens E, et al. Psoriasis and psoriatic arthritis induced by nivolumab in a patient with advanced lung cancer. Rheumatology (Oxford). 2016;55(11):2087–9.
Schmutz JL. Psoriasis and psoriatic arthritis induced by nivolumab (Opdivo((R))). Ann Dermatol Venereol. 2016;143(12):881–2.
Sugiura Y, Fujimoto H, Yamamoto M, et al. Psoriasis and psoriatic arthritis induced by nivolumab in a patient with advanced non-small-cell lung cancer. Gan To Kagaku Ryoho. 2017;44(9):787–9.
Ruiz-Banobre J, Perez-Pampin E, Garcia-Gonzalez J, et al. Development of psoriatic arthritis during nivolumab therapy for metastatic non-small cell lung cancer, clinical outcome analysis and review of the literature. Lung Cancer. 2017;108:217–21.
Elosua-Gonzalez M, Pampin-Franco A, Mazzucchelli-Esteban R, et al. A case of de novo palmoplantar psoriasis with psoriatic arthritis and autoimmune hypothyroidism after receiving nivolumab therapy. Dermatol Online J. 2017. https://doi.org/10.5070/D3238036018.
Lidar M, Giat E, Garelick D, et al. Rheumatic manifestations among cancer patients treated with immune checkpoint inhibitors. Autoimmun Rev. 2018;17(3):284–9.
Hara T, Mikita N, Ikeda T, et al. Psoriatic arthritis induced by anti-programmed death 1 antibody pembrolizumab. J Dermatol. 2019;46(12):e466–7.
Nigro O, Pinotti G, Gueli R, et al. Psoriatic arthritis induced by anti-PD1 and treated with apremilast: a case report and review of the literature. Immunotherapy-Uk. 2020;12(8):549–54.
Tirpack A, Chan KK. Pembrolizumab-induced phenotypic switch in psoriatic arthritis. JCR-J Clin Rheumatol. 2021;27(8s):S683–4.
Jatwani K, Kaur H, Chugh K, Jatwani S. Nivolumab-induced psoriatic arthritis in a patient with advanced small cell lung cancer. JCR-J Clin Rheumatol. 2021;27(5):E162–3.
Takeda K, Yanagitani N. Guselkumab for treating immune checkpoint inhibitor-induced psoriatic arthritis. Ann Rheum Dis. 2022;81(10):1479–80.
Gomez-Puerta JA, Lobo-Prat D, Perez-Garcia C, et al. Clinical patterns and follow-up of inflammatory arthritis and other immune-related adverse events induced by checkpoint inhibitors. A multicenter study. Front Med Lausanne. 2022. https://doi.org/10.3389/fmed.2022.888377.
Zimmer L, Goldinger SM, Hofmann L, et al. Neurological, respiratory, musculoskeletal, cardiac and ocular side-effects of anti-PD-1 therapy. Eur J Cancer. 2016;60:210–25.
Sapalidis K, Kosmidis C, Michalopoulos N, et al. Psoriatic arthritis due to nivolumab administration a case report and review of the literature. Respir Med Case Rep. 2018;23:182–7.
Le Burel S, Champiat S, Mateus C, et al. Prevalence of immune-related systemic adverse events in patients treated with anti-programmed cell death 1/anti-programmed cell death-ligand 1 agents: a single-centre pharmacovigilance database analysis. Eur J Cancer. 2017;82:34–44.
Ma VT, Lao CD, Fecher LA, Schiopu E. Successful use of secukinumab in two melanoma patients with immune checkpoint inhibitor-induced inflammatory arthropathy. Immunotherapy-Uk. 2022;14(8):593–8.
Kim S, Sun JH, Kim H, et al. Pembrolizumab-induced psoriatic arthritis treated with disease-modifying anti-rheumatic drugs in a patient with gastric cancer: a case report. World J Clin Cases. 2023;11(1):218–24.
Papp KA, Caro I, Leung HM, et al. Efalizumab for the treatment of psoriatic arthritis. J Cutan Med Surg. 2007;11(2):57–66.
Myers WA, Najarian D, Gottlieb AB. New onset, debilitating arthritis in psoriasis patients receiving efalizumab. J Investig Dermatol. 2006;126:108–108.
Bang B, Gniadecki R. Severe exacerbation of psoriatic arthritis during treatment with efalizumab. A case report. Acta Dermvenereol. 2006;86(5):456–7.
Viguier M, Richette P, Aubin F, et al. Onset of psoriatic arthritis in patients treated with efalizumab for moderate to severe psoriasis. Arthritis Rheum-Us. 2008;58(6):1796–802.
Colina M, Ciancio G, Khodeir M, et al. De novo onset of arthritis in patients previously treated with efalizumab: an observational case series. Clin Exp Rheumatol. 2011;29(1):141–141.
Dass S, Vital EM, Emery P. Development of psoriasis after B cell depletion with rituximab. Arthritis Rheum-Us. 2007;56(8):2715–8.
Mielke F, Schneider-Obermeyer J, Doerner T. Onset of psoriasis with psoriatic arthropathy during rituximab treatment of non-Hodgkin lymphoma. Ann Rheum Dis. 2008;67(7):1056–7.
Tichy M, Tichy M, Kopova R, et al. Psoriasis and psoriatic arthritis induced in a patient treated with infliximab for Crohn’s disease. J Dermatol Treat. 2012;23(3):208–11.
Stamell EF, Kutner A, Viola K, Cohen SR. Ustekinumab associated with flares of psoriatic arthritis. Jama Dermatol. 2013;149(12):1410–3.
Villani AP, Weiler L, Jullien D, et al. Paradoxical psoriatic arthritis in a patient with rheumatoid arthritis treated by TNFalpha blocker. Jt Bone Spine. 2014;81(5):455–6.
Aussy A, Girszyn N, Vandhuick T, et al. Psoriatic arthritis during rituximab treatment of granulomatosis with polyangiitis: a new paradoxical side effect? Rev Med Interne. 2015;36(7):491–4.
Carija A, Ivic I, Marasovic-Krstulovic D, Puizina-Ivic N. Paradoxical psoriatic arthritis in a patient with psoriasis treated with ustekinumab. Rheumatology (Oxford). 2015;54(11):2114–6.
Takahashi T, Asano Y, Shibata S, et al. Arthritis possibly induced and exacerbated by a tumour necrosis factor antagonist in a patient with psoriasis vulgaris. Br J Dermatol. 2015;172(5):1458–60.
Jones BB, Millsop JW, Walsh JA, et al. Onset of psoriatic arthritis during ustekinumab treatment for psoriasis: a case series of seven patients. Br J Dermatol. 2015;173(1):272–4.
Bonifati C, Graceffa D. How effective is ustekinumab in controlling psoriatic arthritis? Dermatol Ther. 2016;29(3):155–9.
Su PQ, Pan JY. Paradoxical flare of psoriasis, psoriatic spondyloarthritis, and psoriatic uveitis after switching from infliximab to secukinumab. Dermatol Sin. 2017;35(2):112–3.
Ruiz-Genao D, Perez-Zafrilla B, Lopez-Estebaranz JL, et al. Possible paradoxical occurrence of inflammatory arthritis in patients with psoriasis treated with biologics: findings in the Biobadaderm cohort. Br J Dermatol. 2017;176(3):797–9.
Napolitano M, Balato N, Caso F, et al. Paradoxical onset of psoriatic arthritis during treatment with biologic agents for plaque psoriasis: a combined dermatology and rheumatology clinical study. Clin Exp Rheumatol. 2017;35(1):137–40.
Asahina A, Umezawa Y, Momose M, et al. New onset or transition of disease state of psoriatic arthritis during treatment with ustekinumab: a single-center retrospective study. J Dermatol. 2017;44(12):1380–4.
Yasuda M, Morimoto N, Yonemoto Y, et al. Concurrent development of psoriatic arthritis, drug-induced interstitial pneumonia, and increased anti-nuclear antibodies in a patient with psoriasis vulgaris undergoing adalimumab treatment. Eur J Dermatol. 2019;29(1):81–2.
Watabe D, Amano H. Successful treatment of adalimumab-induced paradoxical reaction with risankizumab in a patient with psoriatic arthritis. Eur J Dermatol. 2020;30(5):611–2.
Di Costanzo L, Ferrucci MG, Cusano F. “Paradoxical joint inflammation” possibly induced by a tumor necrosis factor antagonist in three patients with psoriasis. Giorn Ital Dermat. 2020;155(3):370–1.
Kashlan R, Slater K, Garber ME, Kartono F. Paradoxical psoriatic arthritis flare with the initiation of brodalumab and guselkumab. JAAD Case Rep. 2021;10:47–9.
Megna M, Ocampo-Garza SS, Potestio L, et al. New-onset psoriatic arthritis under biologics in psoriasis patients: an increasing challenge? Biomedicines. 2021;9(10):1482.
Carriero A, Lubrano E, Picerno V, et al. Paradoxical psoriatic arthritis in a patient with psoriasis treated with guselkumab. Clin Exp Dermatol. 2022;47(4):783–5.
Stevenson ML, Markowitz O. Selection of therapies in patients with psoriasis and psoriatic arthritis. Psoriasis Forum. 2011;17a(2):144–7.
de Souza A, Ali-Shaw T, Reddy SM, et al. Inflammatory arthritis following ustekinumab treatment for psoriasis: a report of two cases. Br J Dermatol. 2013;168(1):210–2.
Vidal D, Ros S, Reina D. Paradoxical arthritis due to ixekizumab in a patient with plaque psoriasis. Actas Dermo-Sifilogr. 2019;110(3):255–6.
Onsun N, Yalcin B, Sallahoglu K, Rezvani A. Worsening of psoriatic arthritis after ustekinumab treatment. Am J Ther. 2018;25(3):E381–2.
Vilaca S, Torres T, Selores M. Sacroiliitis in a psoriasis patient after switching from etanercept to ustekinumab. Eur J Dermatol. 2013;23(6):897–8.
Garcia-Melendo C, Vilarrasa E, Cubiro X, et al. Sequential paradoxical psoriasiform reaction and sacroiliitis following adalimumab treatment of hidradenitis suppurativa, successfully treated with guselkumab. Dermatol Ther. 2020. https://doi.org/10.1111/dth.14180.
Moustou AE, Stratigos AJ, Vergou T, et al. Arthralgia as an adverse event to infliximab: a reactive arthritis or triggering of psoriatic arthritis? Successful management by switching to etanercept. J Eur Acad Dermatol. 2009;23(8):968–9.
Skerritt PW. Psoriatic-arthritis during lithium-therapy. Aust Nz J Psychiat. 1987;21(4):601–4.
Macfarlane DG, Settas L. Acute psoriatric arthropathy precipitated by oxprenolol. Ann Rheum Dis. 1984;43(1):102–4.
Tatu AL, Nwabudike LC. Metoprolol-associated onset of psoriatic arthropathy. Am J Ther. 2017;24(3):e370–1.
Kavanaugh A. Flare of psoriasis and psoriatic arthritis following treatment with granulocyte colony-stimulating factor. Am J Med. 1996;101(5):567–8.
Nakamura K, Arakawa Y, Masuda K, et al. Exacerbation of psoriatic arthritis due to topical imiquimod therapy. Int J Dermatol. 2023;62(3):E156–8.
Vacchiano V, Foschi M, Sabattini L, et al. Arthritic psoriasis during natalizumab treatment: a case report and review of the literature. Neurol Sci. 2018;39(1):181–3.
Helliwell P, Coates L, Chandran V, et al. Qualifying unmet needs and improving standards of care in psoriatic arthritis. Arthritis Care Res (Hoboken). 2014;66(12):1759–66.
Gottlieb AB, Mease PJ, Mark Jackson J, et al. Clinical characteristics of psoriatic arthritis and psoriasis in dermatologists’ offices. J Dermatol Treat. 2006;17(5):279–87.
Yamamoto T. Psoriatic arthritis: from a dermatological perspective. Eur J Dermatol. 2011;21(5):660–6.
Catanoso M, Pipitone N, Salvarani C. Epidemiology of psoriatic arthritis. Reumatismo. 2012;64(2):66–70.
Taylor W, Gladman D, Helliwell P, et al. Classification criteria for psoriatic arthritis—Development of new criteria from a large international study. Arthritis Rheum-Us. 2006;54(8):2665–73.
Soltani-Arabshahi R, Wong B, Feng BJ, et al. Obesity in early adulthood as a risk factor for psoriatic arthritis. Arch Dermatol. 2010;146(7):721–6.
Christophers E, Barker JNWN, Griffiths CEM, et al. The risk of psoriatic arthritis remains constant following initial diagnosis of psoriasis among patients seen in European dermatology clinics. J Eur Acad Dermatol. 2010;24(5):548–54.
Lo Y, Wang TS, Li KJ, Tsai TF. Correlation of clinical diagnosis of dactylitis by the dermatologist and ultrasonographic diagnosis by the rheumatologist in patients with psoriasis arthritis: experience of a single clinic. Dermatol Sin. 2021;39(1):27–32.
Villani AP, Rouzaud M, Sevrain M, et al. Prevalence of undiagnosed psoriatic arthritis among psoriasis patients: systematic review and meta-analysis. J Am Acad Dermatol. 2015;73(2):242–8.
Scher JU, Ogdie A, Merola JF, Ritchlin C. Preventing psoriatic arthritis: focusing on patients with psoriasis at increased risk of transition. Nat Rev Rheumatol. 2019;15(3):153–66.
Eder L, Law T, Chandran V, et al. Association between environmental factors and onset of psoriatic arthritis in patients with psoriasis. Arthrit Care Res. 2011;63(8):1091–7.
Wu S, Han J, Qureshi AA. Use of aspirin, non-steroidal anti-inflammatory drugs, and acetaminophen (paracetamol), and risk of psoriasis and psoriatic arthritis: a cohort study. Acta Derm Venereol. 2015;95(2):217–23.
Pattison E, Harrison BJ, Griffiths CE, et al. Environmental risk factors for the development of psoriatic arthritis: results from a case-control study. Ann Rheum Dis. 2008;67(5):672–6.
McGonagle D, Gibbon W, Emery P. Classification of inflammatory arthritis by enthesitis. Lancet. 1998;352(9134):1137–40.
Costello P, Bresnihan B, O’Farrelly C, FitzGerald O. Predominance of CD8+ T lymphocytes in psoriatic arthritis. J Rheumatol. 1999;26(5):1117–24.
Fitzgerald O, Winchester R. Psoriatic arthritis: from pathogenesis to therapy. Arthritis Res Ther. 2009;11(1):214.
Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8: 812010.
Wu PC, Huang IH, Wang CW, et al. New onset and exacerbations of psoriasis following COVID-19 vaccines: a systematic review. Am J Clin Dermatol. 2022;23(6):775–99.
Novelli L, Motta F, Ceribelli A, et al. A case of psoriatic arthritis triggered by SARS-CoV-2 infection. Rheumatology (Oxford). 2021;60(1):e21–3.
Ohmura SI, Homma Y, Hanai S, Miyamoto T. Successful use of certolizumab pegol for refractory psoriatic arthritis triggered by COVID-19 infection. Intern Med. 2022;61(3):433–8.
Ma M, Santosa A, Fong W, et al. Post-mRNA vaccine flares in autoimmune inflammatory rheumatic diseases: results from the COronavirus National Vaccine registry for ImmuNe diseases SINGapore (CONVIN-SING). J Autoimmun. 2023;134: 102959.
Lande R, Gregorio J, Facchinetti V, et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature. 2007;449(7162):564–9.
Ganguly D, Chamilos G, Lande R, et al. Self-RNA-antimicrobial peptide complexes activate human dendritic cells through TLR7 and TLR8. J Exp Med. 2009;206(9):1983–94.
Zhou Q, Vadakekolathu J, Watad A, et al. SARS-CoV-2 infection induces psoriatic arthritis flares and enthesis resident plasmacytoid dendritic cell type-1 Interferon inhibition by JAK antagonism offer novel spondyloarthritis pathogenesis insights. Front Immunol. 2021. https://doi.org/10.3389/fimmu.2021.635018.
Doroud D, Hozouri H. Role of intravesical BCG as a therapeutic vaccine for treatment of bladder carcinoma. Iran Biomed J. 2022;26(5):340–9.
Kleinnijenhuis J, Quintin J, Preijers F, et al. Long-lasting effects of BCG vaccination on both heterologous Th1/Th17 responses and innate trained immunity. J Innate Immun. 2014;6(2):152–8.
Netea MG, Crevel R. BCG-induced protection: effects on innate immune memory. Semin Immunol. 2014;26(6):512–7.
Funes SC, Rios M, Fernandez-Fierro A, et al. Trained immunity contribution to autoimmune and inflammatory disorders. Front Immunol. 2022. https://doi.org/10.3389/fimmu.2022.868343.
van Tok MN, Satumtira N, Dorris M, et al. Innate immune activation can trigger experimental spondyloarthritis in HLA-B27/Huβ2m transgenic rats. Front Immunol. 2017;8:920.
Wenzel J, Uerlich M, Haller O, et al. Enhanced type I interferon signaling and recruitment of chemokine receptor CXCR3-expressing lymphocytes into the skin following treatment with the TLR7-agonist imiquimod. J Cutan Pathol. 2005;32(4):257–62.
Fritzsch J, Krug J, Heberling HJ. Interferon therapy and autoimmunity. Med Klin. 1997;92(5):265–72.
Erkek E, Karaduman A, Akcan Y, et al. Psoriasis associated with HCV and exacerbated by interferon alpha: complete clearance with acitretin during interferon alpha treatment for chronic active hepatitis. Dermatology. 2000;201(2):179–80.
Funk J, Langeland T, Schrumpf E, Hanssen LE. Psoriasis induced by interferon-alpha. Br J Dermatol. 1991;125(5):463–5.
Nestle FO, Conrad C, Tun-Kyi A, et al. Plasmacytoid predendritic cells initiate psoriasis through interferon-alpha production. J Exp Med. 2005;202(1):135–43.
Santini SM, Lapenta C, Donati S, et al. Interferon-alpha-conditioned human monocytes combine a Th1-orienting attitude with the induction of autologous Th17 responses: role of IL-23 and IL-12. PLoS One. 2011;6(2): e17364.
Nestle FO, Turka LA, Nickoloff BJ. Characterization of dermal dendritic cells in psoriasis. Autostimulation of T lymphocytes and induction of Th1 type cytokines. J Clin Investig. 1994;94(1):202–9.
Eriksen KW, Lovato P, Skov L, et al. Increased sensitivity to interferon-alpha in psoriatic T cells. J Investig Dermatol. 2005;125(5):936–44.
Dulos J, Carven GJ, van Boxtel SJ, et al. PD-1 blockade augments Th1 and Th17 and suppresses Th2 responses in peripheral blood from patients with prostate and advanced melanoma cancer. J Immunother. 2012;35(2):169–78.
Thapa B, Ali A, Nair R, et al. A case of severe seronegative inflammatory arthritis due to nivolumab and review of the literature. Case Rep Rheumatol. 2019;2019:1326734.
Thanarajasingam U, Zhu XX, Zhou X, et al. Inflammatory arthritis induced by immune checkpoint inhibitor therapy: a distinct clinical entity and immunologic phenotype. Arthritis Rheumatol. 2019;71(suppl 10).
Bartosinska J, Zakrzewska E, Purkot J, et al. Decreased blood CD4+PD-1+and CD8+PD-1+T cells in psoriatic patients with and without arthritis. Postep Dermatol Alergol. 2018;35(4):344–50.
Brumfiel CM, Patel MH, Zirwas MJ. Development of psoriasis during treatment with dupilumab: a systematic review. J Am Acad Dermatol. 2022;86(3):708–9.
Bridgewood C, Wittmann M, Macleod T, et al. T Helper 2 IL-4/IL-13 dual blockade with dupilumab is linked to some emergent T helper 17-type diseases, including seronegative arthritis and enthesitis/enthesopathy, but not to humoral autoimmune diseases. J Investig Dermatol. 2022;142(10):2660–7.
Su Z, Zeng YP. Dupilumab-associated psoriasis and psoriasiform manifestations: a scoping review. Dermatology. 2023;239(4):646–57.
Bridgewood C, Sharif K, Freeston J, et al. Regulation of entheseal IL-23 expression by IL-4 and IL-13 as an explanation for arthropathy development under dupilumab therapy. Rheumatology. 2021;60(5):2461–6.
Bridgewood C, Newton D, Bragazzi N, et al. Unexpected connections of the IL-23/IL-17 and IL-4/IL-13 cytokine axes in inflammatory arthritis and enthesitis. Semin Immunol. 2021;58: 101520.
Nathan J, Hughes C, Patel S, et al. AB0573 dupilumab-induced enthesitis/arthritis in patients with atopic dermatitis: a retrospective observational study. Ann Rheum Dis. 2021;80(Suppl 1):1323–4.
Hassan AS, Simon D, Simon HU, et al. Efalizumab-associated papular psoriasis. Arch Dermatol. 2007;143(7):900–6.
Singla S, Putman M, Liew J, Gordon K. Association between biological immunotherapy for psoriasis and time to incident inflammatory arthritis: a retrospective cohort study. Lancet Rheumatol. 2023;5(4):200–7.
Conrad C, Di Domizio J, Mylonas A, et al. TNF blockade induces a dysregulated type I interferon response without autoimmunity in paradoxical psoriasis. Nat Commun. 2018. https://doi.org/10.1038/s41467-017-02466-4.
Mylonas A, Conrad C. Psoriasis: classical vs. paradoxical. The Yin-Yang of TNF and type I interferon. Front Immunol. 2018;9:2746.
Lu CY, Wallace BI, Waljee AK, et al. Comparative efficacy and safety of targeted DMARDs for active psoriatic arthritis during induction therapy: a systematic review and network meta-analysis. Semin Arthritis Rheu. 2019;49(3):381–8.
Coates L, Gossec L. The updated GRAPPA and EULAR recommendations for the management of psoriatic arthritis: similarities and differences. Jt Bone Spine. 2023;90(1):105469.
van Muijen ME, van Hal TW, Groenewoud HMM, et al. The skin may clear but the arthritis won’t disappear: focusing on concomitant and new-onset psoriatic arthritis in a daily practice cohort of psoriasis patients on biologic therapy. Psoriasis (Auckl). 2020;10:29–37.
Kruithof E, De Rycke L, Roth J, et al. Immunomodulatory effects of etanercept on peripheral joint synovitis in the spondylarthropathies. Arthritis Rheum. 2005;52(12):3898–909.
Canete JD, Pablos JL, Sanmarti R, et al. Antiangiogenic effects of anti-tumor necrosis factor alpha therapy with infliximab in psoriatic arthritis. Arthritis Rheum. 2004;50(5):1636–41.
Pontifex EK, Gerlag DM, Gogarty M, et al. Change in CD3 positive T-cell expression in psoriatic arthritis synovium correlates with change in DAS28 and magnetic resonance imaging synovitis scores following initiation of biologic therapy—a single centre, open-label study. Arthritis Res Ther. 2011;13(1):R7.
Bolt JW, van Kuijk AW, Teunissen MBM, et al. Impact of adalimumab treatment on interleukin-17 and interleukin-17 receptor expression in skin and synovium of psoriatic arthritis patients with mild psoriasis. Biomedicines. 2022;10(2):324.
van Mens LJJ, van de Sande MGH, Menegatti S, et al. Brief report: interleukin-17 blockade with secukinumab in peripheral spondyloarthritis impacts synovial immunopathology without compromising systemic immune responses. Arthritis Rheumatol. 2018;70(12):1994–2002.
Waszczykowski M, Bednarski I, Narbutt J, et al. Interleukin-18, interleukin-20, and matrix metalloproteinases (MMP-1, MMP-3) as markers of psoriatic arthritis disease severity and their correlations with biomarkers of inflammation and turnover of joint cartilage. Postep Derm Alergol. 2020;37(6):1001–8.
Noordenbos T, Yeremenko N, Gofita I, et al. Interleukin-17-positive mast cells contribute to synovial inflammation in spondylarthritis. Arthritis Rheum-Us. 2012;64(1):99–109.
Chen S, Noordenbos T, Blijdorp I, et al. Histologic evidence that mast cells contribute to local tissue inflammation in peripheral spondyloarthritis by regulating interleukin-17A content. Rheumatology (Oxford). 2019;58(4):617–27.
Pierard-Franchimont C, Pierard GE. Drug-related psoriasis. Rev Med Liege. 2012;67(3):139–42.
Fry L, Baker BS. Triggering psoriasis: the role of infections and medications. Clin Dermatol. 2007;25(6):606–15.
Boyce ST, Ham RG. Calcium-regulated differentiation of normal human epidermal keratinocytes in chemically defined clonal culture and serum-free serial culture. J Investig Dermatol. 1983;81(1 Suppl):33s–40s.
Kim GK, Del Rosso JQ. Drug-provoked psoriasis: is it drug induced or drug aggravated? Understanding pathophysiology and clinical relevance. J Clin Aesthet Dermatol. 2010;3(1):32–8.
O’Brien M, Koo J. The mechanism of lithium and beta-blocking agents in inducing and exacerbating psoriasis. J Drugs Dermatol. 2006;5(5):426–32.
Ockenfels HM, Nussbaum G, Schultewolter T, et al. Tyrosine phosphorylation in psoriatic T-cells is modulated by drugs that induce or improve psoriasis. Dermatology. 1995;191(3):217–25.
Brown G, Wang E, Leon A, et al. Tumor necrosis factor-alpha inhibitor-induced psoriasis: Systematic review of clinical features, histopathological findings, and management experience. J Am Acad Dermatol. 2017;76(2):334–41.
Wu S, Han J, Li WQ, Qureshi AA. Hypertension, antihypertensive medication use, and risk of psoriasis. Jama Dermatol. 2014;150(9):957–63.
Bakris GL, Smith DW, Tiwari S. Dermatologic manifestations of lithium: a review. Int J Psychiatry Med. 1980;10(4):327–31.
Abel EA, DiCicco LM, Orenberg EK, et al. Drugs in exacerbation of psoriasis. J Am Acad Dermatol. 1986;15(5 Pt 1):1007–22.
Armstrong AW. Psoriasis provoked or exacerbated by medications: identifying culprit drugs. Jama Dermatol. 2014;150(9):963.
Gladman DD, Ziouzina O, Thavaneswaran A, Chandran V. Dactylitis in psoriatic arthritis: prevalence and response to therapy in the biologic era. J Rheumatol. 2013;40(8):1357–9.
Guggino G, Rizzo A, Mauro D, et al. Gut-derived CD8(+) tissue-resident memory T cells are expanded in the peripheral blood and synovia of SpA patients. Ann Rheum Dis. 2021;80(11): e174.
Funding
No funding or sponsorship was received for this study.
Author information
Authors and Affiliations
Contributions
Dr. Y-TY conducted the literature search, summarized the identified papers, and drafted the manuscript. Dr. T-FT is responsible for the ideation of this topic and final refinement of this article.
Corresponding author
Ethics declarations
Conflict of interest
Yao-Tsung Yeh has no conflicts of interest to disclose. Tsen-Fang Tsai has conducted clinical trials or received honoraria for serving as a consultant for Abbvie, AnaptysBio, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Eli-Lilly, Galderma, GSK, Janssen-Cilag, Leo Pharma, Merck Sharp & Dohme, Novartis International, Pfizer, PharmaEssentia, Sanofi, Sun Pharma and UCB Pharma.
Ethics approval
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Yeh, YT., Tsai, TF. Drug- or Vaccine-Induced/Aggravated Psoriatic Arthritis: A Systematic Review. Dermatol Ther (Heidelb) 14, 59–81 (2024). https://doi.org/10.1007/s13555-023-01082-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-023-01082-z