Abstract
Introduction
We examined the effect of 5.0 g/day of collagen peptide (CP) or collagen peptide fermented with Aspergillus sojae (FCP) on skin pigmentation in healthy participants.
Methods
In this randomized, double-blind, placebo-controlled study, 44 men and women aged 25–63 years were placed into three groups by stratified random allocation and treated with CP, FCP, or placebo (PL) at 5.0 g/day for 3 months. Their skin condition was measured monthly from baseline to 3 months of intake.
Results
No adverse events were identified in any group. The CP group showed a significant reduction in pigmented patches and redness after 1 and 3 months of intake, respectively. In the FCP group, pigmented macules were significantly reduced after 1 month, and pigmented patches after 2 months. Both the all-ages analysis and the hierarchical analysis below 55 years old yielded similar results.
Conclusion
Intake of 5.0 g/day of FCP for 3 months is safe. CP and FCP intake is useful for suppressing pigmentation. In addition, CP intake may be useful for reducing redness. These results suggest a new beneficial effect on the skin of CP supplementation.
Trial Registration
UMIN clinical trials registry system, UMIN000040736.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Collagen peptide (CP) has been reported to be highly effective for skin beautification and has been confirmed to reduce invisible spots that appear during UV irradiation, but its effect on visible pigmentation is still unknown. |
In this clinical study, we evaluated the effect of CP or CP fermented with Aspergillus sojae (FCP) on skin pigmentation in healthy participants. |
What was learned from the study? |
CP and FCP intake are useful for suppressing visible pigmentation after 1 month. |
In addition, CP intake reduces the redness after 3 months. |
Introduction
Collagen is the most abundant protein in the body. It forms a triple-helix structure consisting of three polypeptide chains in which the glycine (Gly)–X–Y sequence (where X and Y represent arbitrary amino acids) is repeated. Almost all proline (Pro) residues at the Y position are posttranslated to collagen-specific hydroxyproline (Hyp). When collagen peptide (CP) is ingested orally, amino acids as well as dipeptides and tripeptides migrate to the blood [1, 2]. In a randomized, placebo, double-blind study, intake of fish- and porcine-derived CP improved skin moisture, elasticity, texture, and clarity and reduced wrinkles [3]. CP also reduces ultraviolet (UV) spots, which are basically defined as invisible spots induced by UV irradiation [4]. Choi et al. reported that ingesting 3 g of CP had no effect on skin pigmentation [5]. Therefore, the effect of reducing visible pigmentation by ingesting CP remains unconfirmed in human studies.
We have developed a fermented collagen peptide (FCP), which was fermented and enzymatically treated with Aspergillus sojae from gelatin. Aspergillus species produce various secondary metabolites, including useful proteins such as protease and amylase [6]. These products are used as detergents and for producing fermented foods [7]. In particular, A. sojae has an excellent ability to secrete protease; it is also safe because it does not produce toxins [8]. Owing to these excellent abilities, A. sojae has been used to produce traditional Japanese foods, such as koji and soy sauce. In addition, microorganisms produce different secondary metabolites that have beneficial effects on humans during the fermentation process [9]. Secondary metabolites, including kojic acid and 14-dehydroergosterol (14-DHE), from Aspergillus species have powerful effects on the skin. Kojic acid suppresses the activity of tyrosinase that synthesizes melanin, and it was registered as a quasi-drug in Japan in 1988 [10, 11]. Subsequently, clinical trials demonstrated that applying a cream containing kojic acid suppresses UV irradiation-induced pigmentation in healthy participants [10]. In a human study, intake of miso, which is a product fermented from rice koji, helps improve skin moisture [12]. Koji extract containing 14-DHE also increases skin moisture [13].
Furthermore, we reported that ingesting FCP reduces visceral fat mass and body weight in mice with a high-fat diet (unpublished). According to Armin et al., fermented fish gelatin-derived peptides exhibit antibacterial and inhibitory effects against angiotensin-converting enzyme and dipeptidyl peptidase IV [14]. However, the effect of FCP on skin condition remains unknown. Hence, the present randomized, double-blind, placebo-controlled trial aimed to evaluate the effects of CP and FCP intake on pigmentation in healthy individuals.
Methods
Study Design
This test conformed to the principles of the Declaration of Helsinki and the Ethical Guidelines for Epidemiological Research and complied with this test implementation plan. The study protocol was approved by the Nitta Gelatin Ethics Review Board (approval no. RCB2020-001–02) and was registered in the UMIN clinical trials registry system (UMIN000040736). All participants provided written consent for our randomized, double-blind, placebo-controlled study. This study was conducted under the guidance of Prof. Igase of Ehime University School of Medicine. After screening allocation, participants consumed their assigned test food at 5.0 g/day. Efficacy was assessed monthly from baseline up to 3 months of supplementation. These measurements were taken from baseline to 3 months later on 11 October 2020, 16 November 2020, 16 December 2020, and 12 January 2021, with a 1-week adjustment period between each measurement. Participants were instructed not to make any lifestyle changes (e.g., eating and exercise habits).
Participant Criteria
According to the protocol, the selection criteria were as follows: (1) age 20–70 years, (2) no abnormalities in the medical examination within 1 year after test initiation, (3) an initial visceral fat area of 80 cm2 or more, and (4) able to maintain an intake rate of 80% or more for 6 months.
Conversely, the exclusion criteria were (1) history of serious illness, (2) abnormal liver function and renal function test values in the medical examination, (3) impaired cardiopulmonary function, (4) having food and drug allergies, (5) playing intense sports or being on a diet, (6) being pregnant, or (7) being judged to be ineligible by the investigator or the examiner.
Participant Allocation
A total of 109 people applied to this study. The investigator created a stratified random allocation table using random numbers. The prepared allocation table was provided only to the test food management personnel, and the test food was distributed to the participants, who were blinded from such food. These participants were the staff of all testing institutions, including the organizers, investigators, test coordinators, test food management personnel, and members of the ethics review committee. After the test was completed, the person in charge of the statistical analysis opened the allocation table.
Test Food
Each participant received porcine CP (average molecular weight 500–1000 Da, content ≥ 3 g/kg concentration of Pro–Hyp and Hyp–Gly; Nitta Gelatin Inc., Osaka, Japan), porcine FCP (average molecular weight 700–2000 Da; Nitta Gelatin Inc., Osaka, Japan), or PL of maltodextrin (Paindex #2; Matsutani Chemical Industry Co., Ltd., Hyogo, Japan).
Intake method
CP, FCP, and PL were packed in an aluminum pouch to conceal the contents from the participants during the distribution, and each food was ingested at 5.0 g/day for 3 months at the participant’s preferred timing. If they forgot to take it, they were instructed to take it on the same day, up to one packet per day.
Evaluation
Examination Schedule
During screening, participants gave written confirmation regarding informed consent, medical history, and presence or absence of addiction/allergy as well as other appropriate confirmations. The participants’ blood pressure, body weight, and body mass index, and skin conditions were obtained at baseline, 1 month, 2 months, and 3 months. As mentioned above, efficacy was measured monthly at baseline and up to 3 months of intake. Study outcomes were the measured values at each time point and the amount of change from baseline.
Evaluation of Skin Condition
All skin parameters were measured in an air-conditioned room at 19.2 ± 3.5 °C and 37.3 ± 8.8% RH. The study was conducted from October to January, during Japan’s winter season. The monthly average amount of daily erythema induced by UV irradiation was 0.68–1.37 kJ/m2, and the monthly average of daily UV-B irradiation amount was 5.02–10.54 kJ/m2, which is the standard climate of autumn to winter in Japan. The average humidity was 44–94%, which indicates a relatively dry season compared with other seasons. Before the study, the participants washed their faces, followed by acclimation for 20 min. Subsequently, we captured an image of their faces using a Robo Skin Analyzer RSA50SII (MM & NIIC, Tokyo, Japan). Using WSK-P500U (MM & NIIC, Tokyo, Japan), we measured the oil and water contents of the skin. Subsequently, parameters such as pigmentation, redness, skin tone, wrinkles, oil content, and water content were evaluated.
Pigmentation was expressed as the number of spots and the area. The area of pigmentation was classified into pigmented macules (barely noticeable, measuring 0.6–1.2 mm2) and patches (noticeable, measuring ≥ 1.2 mm2). Moreover, the darkness level of pigmented patches evaluated by Robo Skin Analyzer was detected by levels (Lvs), which were classified into Lv. 1, Lv. 2, and Lv. 3. The number of pigmented patches increased as the concentration of pigmented patches increased. Lv. 1 corresponded to basic pigmented patches; as the concentration of pigmented patches increased, Lv. 2 and Lv. 3 could be reached. As Lv. 1 pigmented patches had the maximum detection sensitivity in this study, Lv. 2 and Lv. 3 constituted approximately 54% and 35% of Lv. 1, respectively (Fig. 1).
Pigmentation analysis using the Robo Skin Analyzer: Pigmented macules have less (size, 0.6–1.2 mm2) pigmentation, whereas pigmented patches (size, 1.2 mm2) have more pigmentation. The levels of pigmentation were defined based on successive tone gradations in monochrome images, which were created with a blue signal, which is one of the three elements of color (RGB) images. Detected tones were classified into three levels of darkness, and the areas were categorized into two categories according to size. Thereafter, the total area was calculated
Redness was also expressed as the number of spots and the area. Redness was measured according to the density of the one-dimensional (monochrome) image color information, which was the vector connecting the groups. Redness evaluated by Robo Skin Analyzer was classified into Lv. 1, Lv. 2, and Lv. 3. As the redness increased, the number also increased. Basic redness corresponded to Lv. 1, and Lv. 2 and Lv. 3 could be attained if the degree of redness concentration was increased further. In this study, given that redness Lv. 1 was the maximum detection sensitivity, Lv. 2 and Lv. 3 occupied approximately 38% and 23% of Lv. 1, respectively.
Statistical Analysis
Statistical analysis was performed using StatMate V for Windows (Atoms Co., Ltd., Tokyo, Japan.). All statistical data were analyzed using a two-sided test, and the significance level was set to 5%, with a confidence interval of 95%. Wilcoxon signed-rank test was used for intragroup comparisons of baseline and after 3 months of intake. For three-group comparison, we used Tukey’s test after one-way analysis of variance.
Results
Participant Background
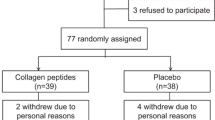
Out of 109 applicants, 44 were included and allocated to the CP, FCP, or PL group. Each group had 14–15 participants (Fig. 1). However, six of them dropped out because of the inability to visit the hospital, a state-of-emergency announcement, and coronavirus infection spread at the third month of measurement. Nevertheless, none of the participants had adverse events associated with the intake of the test food.
Additional analysis was conducted on participants aged below 55 years because of a significant difference in the mean age between the CP group and the PL group (Fig. 2).
Participant tracking: the participants were randomized after enrollment into CP, FCP, and placebo groups. Twelve participants in the CP group, 13 in the FCP group, and 13 in the placebo group completed the study. Additional analysis was conducted on participants aged below 55 years because of a significant difference in the mean age between the CP and PL groups. Therefore, additional analysis was performed by excluding two participants from the FCP group and three participants from the PL group. CP collagen peptide, FCP fermented collagen peptide, PL; placebo
The participants’ body composition changed; however, height, body weight, and body mass index were not significantly different between all groups and the baseline. In addition, the analysis of participants aged below 55 years showed no significant difference in body composition between all groups (Supplementary Material 1). The skin oil and water contents changed, but they were not significantly different between all groups (Supplementary Material 1). Compared with each baseline, the oil content showed no significant difference, but the water content was significantly lower in all groups. The same result was obtained in the analysis of participants aged below 55 years.
Pigmentation and Redness
Supplementary Material 2 lists the degree of change in the area and number of pigmentation and redness after intake of each sample. At baseline, pigmentation and redness in the CP and FCP groups were not significantly different from those in the PL group. After 1 month of treatment, pigmented macules, pigmented patches, and redness significantly decreased in both the CP and FCP groups compared with their baseline. In particular, the CP group had the significantly lowest number of Lv. 2 pigmented patches after 1 month. The number of Lv. 3 pigmented patches was significantly lower after 1 and 3 months (Fig. 3). Its area also decreased significantly from 1 to 3 months. However, the CP group had a significantly higher area of pigmented macules than the PL group after 2 months. In the FCP group, the area of pigmented macules was significantly lower after 1 and 3 months than the baseline, and the number was significantly lower after 2 months. Both the number and area of Lv. 1 pigmented patches were significantly lower after 2 and 3 months for the area, respectively (Fig. 4 and Supplementary Material 2). The area of Lv. 2 pigmented patches and the number of Lv. 3 pigmented patches were significantly lower after 2 months (Supplementary Material 2 and Fig. 3). After 3 months, the number of Lv. 3 redness decreased significantly compared with the baseline of the CP group (Fig. 6).
The number of Lv.3 pigmented patches improved with administration of PL, CP, and FCP supplements. Mean ± standard error. Intragroup comparison: Wilcoxon signed-rank test, #P < 0.05, ##P < 0.01 (versus baseline). CP collagen peptide, FCP fermented collagen peptide, PL placebo. Blue: baseline; orange: after 1 month; gray: after 2 months; yellow: after 3 months
The area of Lv.1 pigmented patches improved with administration of PL, CP, and FCP supplements. Mean ± standard error. Intragroup comparison: Wilcoxon signed-rank test, #P < 0.05, ##P < 0.01 (versus baseline). CP collagen peptide, FCP fermented collagen peptide, PL placebo. Blue: baseline; orange: after 1 month; gray: after 2 months; yellow: after 3 months
The number of Lv. 1 skin pigmented patches: Red dots indicate Lv.1 pigmented patches caused by PL, CP, and FCP supplementation. To protect privacy, the lower half of the face is displayed; however, measurements were taken on the entire face by using the Robo Skin Analyzer RSA50SII (MM & NIIC, Tokyo, Japan). CP collagen peptide, FCP fermented collagen peptide, PL placebo
The number of Lv.3 redness improved with administration of PL, CP, and FCP supplements. Mean ± standard error. Intragroup comparison: Wilcoxon signed-rank test, #P < 0.05, ##P < 0.01 (versus baseline). CP collagen peptide, FCP fermented collagen peptide, PL placebo, Blue: baseline; orange: after 1 month; gray: after 2 months; yellow: after 3 months
The PL group at baseline was significantly older than the CP group (Supplementary Material 1). To eliminate the possible effects of aging, we reanalyzed those participants aged below 55 years (Fig. 2); Supplementary Material 3 presents the results of this reanalysis. At baseline, pigmentation and redness in the CP and FCP groups were not significantly different from those in the PL group. In the CP group, no one was excluded from the reanalysis, so the results were the same as above.
Discussion
Many have reported that CP intake can improve skin quality. Reduction of UV spots was also confirmed, but the effect of CP on visible pigmentation was unverified. In this randomized, double-blind, placebo-controlled study, no adverse events were identified in any group.
The amino acid composition is not substantially different between CP and FCP, and FCP contains Hyp (unpublished). The composition of some di- and tripeptides alsso showed no considerable differences (Supplementary Material 4). After the orally ingested CP is absorbed into the blood, it is transported in the form of di- and tripeptides to the human skin via the peripheral blood vessels [15]. Pro–Hyp and Hyp–Gly are their typical constituents [15]. Pro–Hyp stimulates cell proliferation, cell growth, and hyaluronic acid synthesis in cultured dermal fibroblasts [16]. Hyp–Gly also promotes cell proliferation of mouse primary fibroblasts to a higher degree than Pro–Hyp [17]. These peptides have been suggested to regulate skin cells and extracellular matrix proteins in humans. Therefore, the CP and FCP used in the present study may have also reduced the area and number of pigmentations on the facial skin by controlling and regulating the dermis. One month of CP intake may have reduced the size of pigmented patches, resulting in an increase in the number of pigmented macules.
Moreover, we found no difference in the skin oil and water contents between the PL and the CP or FCP groups (Supplementary Material 1). In our previous clinical studies of CP intake, the water content was significantly improved compared with the baseline in the CP group [18]. This discrepancy may be explained by the difference in measuring instruments; previous studies used Corneometer to measure skin hydration, whereas the present study used the oil and water content device attached to the Robo Skin Analyzer. In other studies, skin hydration significantly recovered between groups and before and after 4 weeks or more of CP intake [19]. This finding is contrary to our present study results, possibly because of the use of a different type of CP. Previous studies used a high content of tripeptides or high-molecular-weight CP, whereas the present study used high contents of Pro–Hyp and Hyp–Gly. In addition, although this study was conducted during the winter in Japan, the results may differ because of differences in UV irradiation dose and humidity depending on the climate and season during implementation.
Regarding redness improvement, Koyama et al. reported that redness was improved 8 weeks after CP supplementation among humans [20]. In mice, Shin et al. observed that CP intake suppressed inflammation (erythema) caused by UV irradiation [21]. Wang et al. also showed that, in a mouse model of inflammation, oral supplementation of shark-derived peptides reduced inflammatory cytokines in the blood and reduced the risk for inflammation-related diseases [22]. Furthermore, Offengenden et al. and Kawaguchi et al. demonstrated that CP addition suppresses protein expression of adhesion molecules (Intercellular adhesion molecule 1 and vascular cell adhesion molecule 1) in different cell inflammation models [23, 24]. Therefore, the results of improving redness by CP supplementation may also reflect the inhibitory effect on inflammation, although further analysis is needed.
FCP is a new CP obtained by fermentation with koji, and components derived from koji may have been involved. Kojic acid extracted from koji can inhibit melanin production [25]. The Ministry of Health, Labour and Welfare of Japan approved quasi-drugs containing kojic acid for the purpose of preventing spots and freckles caused by sunburn, with an active ingredient and a compounding ratio of kojic acid of 0.1–1.0%. Considering that the concentration of kojic acid contained in the FCP used in the present study was considerably lower than the above concentration (data not shown), suppression of melanin production by kojic acid is unlikely. Therefore, the Pro–Hyp and Hyp–Gly of the dipeptides found in FCP contribute to pigmentation reduction. Furthermore, a previous study found that the skin water content was improved by ingesting 14-DHE derived from white Aspergillus [13]. Considering that 14-DHE may increase the activity of “arginase 1,” which is abundant on the surface of the stratum corneum and is involved in the control of the moisturizing function of each layer, the arginase activity was measured after providing Aspergillus extract solution for FCP. The result was below the detection limit (0 µg/mL). Additionally, our study showed no improvement in water content in the FCP group (Supplementary Material 1); thus, 14-DHE may not be involved.
As CP improved the pigmented patches faster than FCP, it is possible that other components, in addition to the Pro–Hyp and Hyp–Gly found in both CP and FCP, are involved. Moreover, although redness was only improved by CP, different factors may have an effect; hence, further research is needed. CP had a higher effect on the skin because it reduced pigmentation faster than FCP, and it also reduced redness. Nonetheless, given that FCP also reduced the pigmented patches after 2 months, long-term intake may have the same improvement effect as CP.
However, this study had a small sample size, with a very low ratio of women. There was no significant difference in the mean age of each group; however, it was lower in the ingestion group than in the PL group (difference: ~ 10.9 years). Moreover, it is possible that the effect on pigmentation and redness was confirmed in the ingestion group due to the low average age and good metabolism. Thus, we would like to conduct a study with a wide range of ages but a similar proportion between men and women in the future. Moreover, the patients’ lifestyle may affect both skin pigmentation and redness (UV exposure, cold exposure, outdoor sporting activities, sun avoidance behavior and use of sunblock, hot baths, sauna, and antihypertensive medications), and these factors are not easily modifiable throughout the study period. Furthermore, the mechanism of improvement in pigmentation and redness in CP and FCP are still unclear, but other factors aside from Pro–Hyp and Hyp–Gly are involved; thus, further research is needed.
Conclusions
This randomized, double-blind, placebo-controlled trial suggests that 3-month intake of 5.0 g/day of CP or FCP is safe. CP significantly reduced the pigmented patches and redness after 1 month of intake, and FCP significantly reduced the pigmented macules after 1 month of supplementation in all ages and in hierarchical analysis below 55 years of age. Therefore, CP and FCP intake are useful for suppressing pigmentation, and CP intake may be useful for reducing redness.
References
Iwai K, Hasegawa T, Taguchi Y, et al. Identification of food-derived collagen peptides in human blood after oral intake of gelatin hydrolysates. J Agric Food Chem. 2005;53:6531–6.
Ohara H, Matsumoto H, Ito K, Iwai K, Sato K. Comparison of quantity and structures of hydroxyproline-containing peptides in human blood after oral ingestion of gelatin hydrolysates from different sources. J Agric Food Chem. 2007;55:1532–5.
Inoue N, Sugihara F, Wang X. Ingestion of bioactive collagen hydrolysates enhance facial skin moisture and elasticity and reduce facial ageing signs in a randomised double-blind placebo-controlled clinical study. J Sci Food Agric. 2016;96:4077–81.
Sugihara F, Inoue N. Clinical effects of collagen hydrolysates ingestion on UV-induced pigmented spots of human skin: A preliminary study. Health Sci. 2012;28:153–6.
Choi SY, Kim WG, Ko EJ, et al. Effect of high advanced-collagen tripeptide on wound healing and skin recovery after fractional photothermolysis treatment. Clin Exp Dermatol. 2014;39:874–80.
Yamashita M, Tsujikami M, Murata S, Kobayashi T, Shimizu M, Kato M. Artificial AmyR::XlnR transcription factor induces α-amylase production in response to non-edible xylan-containing hemicellulosic biomass. Enzyme Microb Technol. 2021;145: 109762.
Mitidieri S, Souza Martinelli AH, Schrank A, Vainstein MH. Enzymatic detergent formulation containing amylase from Aspergillus niger: a comparative study with commercial detergent formulations. Bioresour Technol. 2006;97:1217–24.
Chang PK, Matsushima K, Takahashi T, et al. Understanding nonaflatoxigenicity of Aspergillus sojae a windfall of aflatoxin biosynthesis research. Appl Microbiol Biotechnol. 2007;76:977–84.
Katz L, Baltz RH. Natural product discovery past, present, and future. J Ind Microbiol Biotechnol. 2016;43:155–76.
Mishima Y, Hatta S, Ohyama Y, Inazu M. Induction of melanogenesis suppression: cellular pharmacology and mode of differential action. Pigment Cell Res. 1988;1:367–74.
Higa Y, Kawabe M, Nabae K, et al. Kojic acid-absence of tumor-initiating activity in rat liver, and of carcinogenic and photo-genotoxic potential in mouse skin. J Toxicol Sci. 2007;32:143–59.
Maeda K, Nakata K, Nakamura A, Kitagawa M, Ito S. Improvement in Skin Conditions by consumption of traditional Japanese miso soup and its mechanism. J Nutr Food Sci. 2018;08:660.
Sugihara Y, Ikushima S, Miyake M, Kirisako T, Yada Y, Fujiwara D. Improvement of skin conditions by ingestion of Aspergillus kawachii (Koji) extract containing 14-dehydroergosterol in a randomized, double-blind, controlled trial. Clin Cosmet Investig Dermatol. 2018;11:115–24.
Mirzapour-Kouhdasht A, Lee CW, Yun H, Eun JB. Structure-function relationship of fermented skate skin gelatin-derived bioactive peptides: a peptidomics approach. Food Sci Biotechnol. 2021;30:1685–93.
Ichikawa S, Morifuji M, Ohara H, Matsumoto H, Takeuchi Y, Sato K. Hydroxyproline-containing dipeptides and tripeptides quantified at high concentration in human blood after oral administration of gelatin hydrolysate. Int J Food Sci Nutr. 2010;61:52–60.
Shigemura Y, Iwai K, Morimatsu F, et al. Effect of prolyl-hydroxyproline (Pro–Hyp), a food-derived collagen peptide in human blood, on growth of fibroblasts from mouse skin. J Agric Food Chem. 2009;57:444–9.
Shigemura Y, Akaba S, Kawashima E, Park EY, Nakamura Y, Sato K. Identification of a novel food-derived collagen peptide, hydroxyprolyl-glycine, in human peripheral blood by pre-column derivatisation with phenyl isothiocyanate. Food Chem. 2011;129:1019–24.
Koizumi S, Inoue N, Sugihara F, Wang X. The effects of collagen hydrolysates derived from tilapia scales or skin on human faciai skin—a randormized double-blind pracebo-controlled clinical study. Jpn Pharmacol Ther. 2019;47:57–63.
Proksch E, Segger D, Degwert J, Schunck M, Zague V, Oesser S. Oral supplementation of specific collagen peptides has beneficial effects on human skin physiology: a double-blind, placebo-controlled study. Skin Pharmacol Physiol. 2014;27:47–55.
Koyama Y-I, Kuwaba K, Kondo S, Tsukada Y. Supplemental ingestion of collagen peptide suppresses ultraviolet-induced erythema. Jpn Pharmacol Ther. 2014;42:781–90.
Shin HJ, Park J, Kwon C, et al. The effect of collagen peptide intake on UVB-induced skin damage in hairless mice. J Korea Acad Ind Coop Soc. 2016;17:611–21.
Wang TX, Shen DY, Wang Q, et al. Protective effects of orally administered shark compound peptides from Chiloscyllium plagiosum against acute inflammation. J Food Biochem. 2021;45: e13618.
Offengenden M, Chakrabarti S, Wu J. Chicken collagen hydrolysates differentially mediate anti-inflammatory activity and type I collagen synthesis on human dermal fibroblasts. Food Sci Hum Wellness. 2018;7:138–47.
Kawaguchi T, Nanbu PN, Kurokawa M. Distribution of Prolylhydroxyproline and its metabolites after oral administration in rats. Biol Pharm Bull. 2012;35:422–7.
Mishima Y, Shibata T, Seto H, Ohyama Y, Hatae S. Inhibitory action of kojic acid on melanogensis and its therapeutic effect for various human hyper-pigmentation disorders. Skin. 1994;36:134–50.
Acknowledgements
We would like to express our sincere gratitude to all study participants.
Funding
The authors declare that this study received funding from Nitta Gelatin Inc. The sponsor is also funding the journal’s Rapid Service Fee.
Editorial Assistance
Editorial assistance in the preparation of this article was provided by Dr. Emelda of Enago (www.enago.jp). This assistance was funded by Nitta Gelatin Inc.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Seiko Koizumi funded acquisition. Michiya Igase and Seiko Koizumi conceptualized and created method of this study. Masaya Tsujikami and Ai Himeno calculated the data analyzed Masaya Tsujikamiand Ai Himeno wrote the initial draft of the manuscript. Toshiro Watanabe and Michiya Igase were supervisors of this study. All authors approved the final version of the manuscript, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Disclosures
Masaya Tsujikami, Ai Himeno, and Seiko Koizumi are employees of Nitta Gelatin Inc. Toshiro Watanabe and Michiya Igase have nothing to disclose. This study was conducted with study design, data collection and analysis, and decision to publish, and preparation of the manuscript, and this study funded by Nitta Gelatin Inc.
Compliance with Ethics Guidelines
This study was approved by the Nitta Gelatin Ethics Review Board (approval number: RCB2020-001-02) and was registered in UMIN clinical trials registry system (UMIN000040736). Moreover, it complied with the ethical standards of the 1964 Declaration of Helsinki. All participants provided written consent to this study. Participants who shared a photo of their face provided written consent to publish.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Himeno, A., Tsujikami, M., Koizumi, S. et al. Effect of Reducing Pigmentation by Collagen Peptide Intake: A Randomized, Double-Blind, Placebo-Controlled Study. Dermatol Ther (Heidelb) 12, 1577–1587 (2022). https://doi.org/10.1007/s13555-022-00748-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-022-00748-4