Abstract
Introduction
This study aimed to investigate the effects of cyclosporine on palmoplantar pustulosis (PPP) and serum expression of IL-17, IL-23, and TNF-α.
Methods
Patients with PPP (n = 48) were recruited and treated with cyclosporine alone. The Palmoplantar Pustulosis Area and Severity Index score was obtained, and ELISA was employed to detect the serum IL-17, IL-23, and TNF-α expression before treatment and after 8 weeks of treatment.
Results
Complete remission was achieved in 16.7% of the patients, remission in 45.8%, an improvement in 31.3%, and the treatment was ineffective in 6.25%, yielding an overall effectiveness of 62.5%. Adverse effects included hypertension (n = 6), frequent urination and enuresis nocturna (n = 6), gastrointestinal discomfort (n = 6), hypertrichosis (n = 3), and increased creatinine (n = 1). While serum IL-17, IL-23, and TNF-α concentrations were reduced after 8 weeks of treatment, the reductions were greatest for IL-23 and TNF-α, whereas the reduction in IL-17 was not significant.
Conclusion
Cyclosporine is a safe and effective treatment for PPP with few adverse effects, which might be related to the regulation of IL-23 and TNF-α.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Palmoplantar pustulosis (PPP) is a chronic, recurrent palmoplantar disease that occurs most frequently in patients aged 50–60 years and during the summer. The incidence of PPP is relatively low, and some clinicians speculate that PPP is the palmoplantar manifestation of pustular psoriasis [1]. Currently, PPP is treated with topical corticosteroids, PUVA, acitretin, methotrexate, cyclosporine, tetracyclines, or biological agents. However, the efficacy of each of these drugs in the treatment of PPP and the specific mechanisms that underlie their therapeutic effects are largely unknown. In the present study, patients with PPP who received treatment with cyclosporine alone were recruited, the adverse effects of this treatment were observed, and their serum expression of TNFa, IL-17, and IL-23 was measured in order to explore the mechanism underlying the therapeutic effects of cyclosporine on PPP.
Methods
Patients
All procedures performed in studies involving human participants were implemented in accordance with the ethical standards of the Second Hospital of Jilin University research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. A total of 48 patients with PPP were recruited from the Department of Dermatology of the Second Hospital of Jilin University between June 2017 and March 2018. There were 8 males and 40 females, with a mean age of 49.8 years (range 18–79 years). The mean course of the disease was 2 years (range 15–20 years). 24 patients were smokers. The inclusion criteria were as follows: (1) patients were 18 years or older; (2) patients were diagnosed with PPP; (3) informed consent was obtained before inclusion in the study. The exclusion criteria were as follows: (1) uncontrollable hypertension; (2) severe renal dysfunction; (3) severe infectious diseases; (4) a history of malignant tumors; (5) current malignant tumors (excluding basal cell carcinoma); (6) patients were receiving PUVA treatment; (7) patients received simultaneous active vaccine immunization.
Cyclosporine Treatment and Evaluation of Therapeutic Efficacy
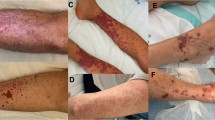
The patients were treated with oral cyclosporine at 3 mg/kg/day, twice daily. At the same time, moisturizer (WINONA’s Hyaluronic Acid Repair Biomask) was applied externally. Treatment lasted for 8 weeks, and the skin lesions were photographed before treatment and after 8 weeks of treatment. The symptoms and signs (pustules, erythema, scars, and lesion area) were scored using the Palmoplantar Pustulosis Area and Severity Index (PPPASI) score system (Table 1) [2, 3]. Before treatment, the PPPASI score was 0.6–47.4 (average 12.8). The reduction in the total score after 8 weeks of treatment was used to evaluate the therapeutic efficacy [4]. The therapeutic efficacy was classified as complete remission (reduction ≥ 90%), remission (60–89%), improvement (20–59%), or treatment ineffectiveness (< 20%). The complete remission rate was calculated as (number of patients in complete remission/total number of patients) × 100%, and the overall l effectiveness was evaluated as the complete remission rate plus the remission rate.
Adverse Effects
All the patients were tested for liver and kidney function, underwent full blood count and urine tests, and had their serum electrolytes, lipids, glucose, and blood pressure monitored before and during treatment (at 4, 8, 12, and 16 weeks) to assess treatment safety. During the treatment, the patients were asked to report adverse effects [blood pressure, kidney dysfunction, gastrointestinal reactions (anorexia, nausea, and vomiting), gingival hyperplasia with bleeding and pain].
Detection of Serum IL-17, IL-23, and TNF-α Concentrations
In the present study, ELISA was employed to detect the serum concentrations of IL-17, IL-23, and TNF-α before and after cyclosporine treatment. Data are expressed as the mean ± standard deviation (mean ± SD) and the t test was used for comparisons between groups. P < 0.05 was considered to indicate statistical significance. The blood was collected from 4 patients in complete remission, 12 in remission, and 8 with improvement.
Statistical Analysis
Statistical analysis was performed with SPSS version 21 (Oracle Co., USA). Data are expressed as the mean ± standard deviation (mean ± SD) and the t test was used for comparisons between groups. P < 0.05 was considered to indicate statistical significance.
Results
Therapeutic Efficacy
45 of the 48 patients who received cyclosporine treatment showed improvement in their skin lesions. The time taken to achieve this improvement was 15–30 days, and the duration of treatment ranged from 15 days to 6 months. Complete remission was noted in 8 of the 48 patients (16.7%), remission in 22 (45.8%), improvement in 15 (31.3%), and ineffectiveness in 3 (6.25%). Note that the duration of treatment was shorter than 1 month in 3 patients in whom the treatment was ineffective. In those patients, the initial dose of cyclosporine was lower than 2.5 mg/kg/day, and treatment was terminated due to adverse effects (nausea, gastric discomfort, and other adverse effects) and high cost. The overall rate of effectiveness was 62.5% (Figs. 1, 2).
Adverse Effects
The adverse effects observed included hypertension (n = 6), frequent urination and enuresis nocturna (n = 6), gastrointestinal reactions (nausea and gastric discomfort) (n = 6), hypertrichosis (n = 3), and increased creatinine (n = 1). They resolved after cyclosporine dose reduction or termination of cyclosporine treatment, although the hypertrichosis improved slowly.
Serum Concentrations of IL-17, IL-23, and TNF-α
Our results showed that the serum concentrations of IL-17, IL-23, and TNF-α reduced after 8 weeks of cyclosporine treatment. Significant reductions in IL-23 and TNF-α were noted, but there was no marked reduction in serum IL-17 (Table 2).
Discussion
PPP is a chronic disease with a high recurrence rate and a poor response to treatment. It occurs most often in patients aged 50–60 years [1]. In the present study, a total of 48 patients diagnosed with PPP were recruited, including 8 males and 40 females. The mean age was 49.8 years (range 18–79 years). Plantar involvement was more frequent than palmar involvement, which was consistent with previously reported cases [1, 5]. This suggests that PPP is frequently found in the palmoplantar region. In Olazagasti et al.’s retrospective review of 215 patients with PPP, systemic agents that elicited a complete response included acitretin (6 of 24 patients; 25%), methotrexate (2 of 20 patients; 10%), and cyclosporine (2 of 4 patients; 50%). Several treatments—dapsone (3 patients), etanercept (3 patients), and prednisone (5 patients)—elicited no response [5]. In the present study, 48 patients with PPP received monotherapy with cyclosporine. The PPPASI score was 0.6–47.4 before treatment (mean 12.8). After 8 weeks of treatment, the efficacy was evaluated. Results showed complete remission in 8.3% of patients, remission in 41.7%, improvement in 22.9%, and treatment ineffectiveness in 20.8%, with an overall effectiveness rate of 50%. These results are consistent with relevant results in the literature [5,6,7] and they confirm that cyclosporine is an effective treatment for PPP.
Cyclosporine A is safer than other commonly used systemic treatments. Adverse reactions disappeared after cyclosporine reduction or withdrawal, and increased body hair recovery was relatively slow. These findings indicate that cyclosporine is an effective treatment for PPP with few adverse effects, and that these adverse effects are mild and may resolve after treatment termination. However, routine monitoring is necessary and the dose of cyclosporine should be determined according to the guidelines in order to reduce or avoid the adverse effects of cyclosporine.
The pathogenesis of PPP is still poorly understood. Smoking, emotional stress, focal infection, certain drugs, genetics, immunity, and metal sensitization are reported to be involved in the occurrence or aggravation of pustulosis [5]. Smoking plays a particularly important role in the pathogenesis of PPP. Tobacco smoke is considered to be a possible trigger of PPP, and it may increase the expression of IL-17 and IL-22 [8]. Recent studies have shown that IL-17, IL-23, and TNF-α play crucial roles in the pathogenesis of psoriasis [9], and PPP is closely related to this pathogenesis [10, 11]. Several reports have reported an overexpression of IL-17 in periodontitis [12, 13]. IL-17 is expressed not only in Th17 cells but also in inflamed tissues such as gingiva [12, 13]. Elevated IL-17 expression has been noted in the skin lesions and sera of PPP patients, and TNF-α is significantly increased in the sera of PPP patients, but IL-23 is not [14]. As a result of this low IL-23 expression in the sera of PPP patients, it was assumed that the expression of IL-17 in inflamed tissues such as gingiva may relate to the pathogenesis of PPP [14]. Our results showed that the serum concentrations of IL-17, IL-23, and TNF-α decreased after 8 weeks of cyclosporine treatment in PPP patients. Significant reductions were noted in IL-23 and TNF-α, while the serum IL-17 concentration decreased slightly. This implies that the therapeutic effects of cyclosporine may be related to the downregulation of IL-17, IL-23, and TNF-α expression, especially that of IL-23 and TNF-α. Note that the sample size was small in the present study, and the specific mechanisms underlying the therapeutic effects of cyclosporine were not investigated further. Thus, more clinical studies with elegant designs and large sample sizes are needed to confirm the etiology and pathogenesis of PPP as well as the mechanisms underpinning the therapeutic effects of cyclosporine.
Conclusions
Cyclosporine is a safe and effective treatment for PPP with few adverse effects, which may be related to the regulation of IL-23 and TNF-α.
References
Zhao B. Chinese clinical dermatology. Nanjing: Jiangsu Science and Technology Press; 2010. p. 858–60.
Morales-Munera C, Vilarrasa E, Puig L. Efficacy of ustekinumab in refractory palmoplantar pustular psoriasis. Br J Dermatol. 2013;168:820–4.
Soleymani T, Vassantachart JM, Wu JJ. Comparison of guidelines for the use of cyclosporine for psoriasis: a critical appraisal and comprehensive review. J Drugs Dermatol. 2016;15:293–301.
Wang M, Wang R, Yu JB. Treatment of palmoplantar pustulosis with low-dose acitretin. Chin J Dermatovenereol. 2005;19:768.
Olazagasti JM, Ma JE, Wetter DA. Clinical features, etiologic factors, associated disorders, and treatment of palmoplantar pustulosis: the Mayo Clinic experience, 1996–2013. Mayo Clin Proc. 2017;92:1351–8.
Adisen E, Tekin O, Gulekon A, Gurer MA. A retrospective analysis of treatment responses of palmoplantar psoriasis in 114 patients. J Eur Acad Dermatol Venereol. 2009;23:814–9.
Reitamo S, Erkko P, Remitz A, Lauerma AI, Montonen O, Harjula K. Cyclosporine in the treatment of palmoplantar pustulosis. A randomized, double-blind, placebo-controlled study. Arch Dermatol. 1993;129:1273–9.
Torii K, Saito C, Furuhashi T, et al. Tobacco smoke is related to Th17 generation with clinical implications for psoriasis patients. Exp Dermatol. 2011;20:371–3.
Nestle FO, Kaplan DH, Barker J. Psoriasis. N Engl J Med. 2009;361:496–509.
Bissonnette R, Suarez-Farinas M, Li X, et al. Based on molecular profiling of gene expression, palmoplantar pustulosis and palmoplantar pustular psoriasis are highly related diseases that appear to be distinct from psoriasis vulgaris. PLoS One. 2016;11:e0155215.
Brunasso AMG, Massone C. Psoriasis and palmoplantar pustulosis: an endless debate? J Eur Acad Dermatol Venereol. 2017;31:e335–7.
Beklen A, Ainola M, Hukkanen M, Gurgan C, Sorsa T, Konttinen YT. MMPs, IL-1, and TNF are regulated by IL-17 in periodontitis. J Dent Res. 2007;86:347–51.
Michalowicz BS, Diehl SR, Gunsolley JC, et al. Evidence of a substantial genetic basis for risk of adult periodontitis. J Periodontol. 2000;71:1699–707.
Murakami M, Hagforsen E, Morhenn V, Ishida-Yamamoto A, Iizuka H. Patients with palmoplantar pustulosis have increased IL-17 and IL-22 levels both in the lesion and serum. Exp Dermatol. 2011;20:845–7.
Acknowledgements
We thank the participants of the study.
Funding
This study was supported by the Research Project of Science and Technology Department of Jilin Province (20160101090JC) and National Natural Science Foundation of China (81803160). Article processing charges were funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Xian-Hua Jin, Xi Chen, Yan Mou, and Jian-Xin Xia have nothing to disclose.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Second Hospital of Jilin University research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.8224022.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License ( http://creativecommons.org/licenses/by-nc/4.0/ ), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Jin, XH., Chen, X., Mou, Y. et al. Effects of Cyclosporine on Palmoplantar Pustulosis and Serum Expression of IL-17, IL-23, and TNF-α. Dermatol Ther (Heidelb) 9, 547–552 (2019). https://doi.org/10.1007/s13555-019-0308-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-019-0308-z