Abstract
Introduction
Inflammation is an established component of cardiovascular disease (CVD) and an underlying factor of several dermatologic conditions including rosacea, atopic dermatitis, and psoriasis. Identifying potential associations between these dermatologic and cardiovascular diseases can better inform holistic healthcare approaches. The objective of this study was to determine whether rosacea, psoriasis or atopic dermatitis are independent risk factors for CVD 1 year following diagnosis.
Methods
Using a large commercial claims database of 21,801,147 lives, we employed a propensity-matched logistic regression to evaluate the association between diagnoses of rosacea, psoriasis, or atopic dermatitis and a 1-year risk of being diagnosed with cardiovascular disease. Control patients were matched based on health-care utilization, age and overall health status as defined by a modified Deyo–Charlson comorbidity index.
Results
The analysis included 2105 rosacea, 622 atopic dermatitis, 1536 psoriasis, and 4263 control patients. Compared to propensity-matched controls, the adjusted odds of cardiovascular disease were not higher in patients with rosacea (odds ratio: 0.894, p = 0.2713), atopic dermatitis (OR 1.032, p = 0.8489), or psoriasis (OR 1.087, p = 0.4210). In univariate analysis, the unadjusted odds of cardiovascular disease was higher in patients with psoriasis (OR 1.223, p = 0.0347).
Conclusions
Limitations of this study include the short follow-up period and inclusion of only commercially insured patients limit the generalizability of these findings. In this large study of patients with rosacea, atopic dermatitis, and psoriasis, we did not detect an increased 1-year risk of cardiovascular disease after adjusting for confounders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most dermatologic conditions are readily apparent to the physician taking care of the patient and can be indicators of inflammatory processes (i.e. atherosclerosis) occurring internally. Some authors discuss an association between psoriasis and cardiovascular disease (CVD) [1–6]. Less clear associations exist between other dermatologic inflammatory conditions [atopic dermatitis (AD) and rosacea] and CVD. Similar to psoriasis, these conditions are (1) common and (2) may reflect underlying systemic inflammation. Following this same concept, these diseases are worth assessing for their independent contribution to CVD risk.
Adults diagnosed with or self-identifying to have AD are more likely to have risk factors associated with CVD including: being a current smoker (adjusted odds ratio (OR) 1.28, p < 0.001), being a current heavy drinker (OR 1.58, p < 0.001), having a lifetime prevalence of diabetes (OR 1.37, p < 0.001), having a lifetime prevalence of high cholesterol (OR 1.29, p < 0.001), or a lifetime prevalence of hypertension (OR 1.31, p < 0.001) [7]. Using a Taiwanese nationwide database, a retrospective population-based study identified 20,323 patients with AD and 20,323 comorbidity-matched subjects (age ≥20 years). After adjusting for age, sex, comorbidities, and medications used, the hazard ratio for experiencing ischemic stroke over a 4-year follow-up period was 1.33 (p = 0.001), suggesting AD may be an independent risk factor of ischemic stroke [8].
For rosacea, a case control study done by Duman et al. assessed the compared CVD risk factors for 60 rosacea patients and 50 age- and gender-matched controls. Rosacea patients were more likely than controls to have: high total cholesterol (199 mg/dL vs. 163, p < 0.001), LDL (121 vs 101, p = 0.002), CRP levels (0.43 vs. 0.24 mg/L, p = 0.007), and a family history of premature CVD (p = 0.002) [9]. This study only assessed risk factors and did not look at cardiovascular endpoints. Also, the control subjects were only age and gender matched, so invariably there was residual confounding. Nevertheless, patients with rosacea may be more likely to have CVD risk factors but this has not been shown to be an independent risk factor for CVD.
In this study, we evaluate whether three dermatologic diseases that have inflammatory components contribute an independent risk for development of CVD outcomes at 1-year follow-up using a large commercial claims database.
Methods
The data were analyzed as a case–control study with matching on covariates using propensity scores to quantify the association between the dermatologic diseases and CVD outcomes. The matching done with propensity scores used a Mahalanobis 1:1 algorithm using all the main effects from the regression except the dermatologic disease indicator variable [9].
Setting and Participants
All variables come from the MarketScan™ Commercial Claims and Encounters database for 2005–2007 [10]. No new studies with human or animal subjects were performed by any of the authors. The people included in the analysis were in the range of 30–64 years old and had at least 1 year of follow-up data and 6 months of baseline data. We chose the lower age limit because CVD is less common in individuals younger than this age. We chose the upper age limit to minimize loss to follow-up that could occur from 65 year olds switching from commercial insurance to Medicare.
The dermatologic diseases group was composed of three arms: rosacea (ICD-9 695.3), psoriasis (ICD-9 696.1), or AD (ICD-9 691.XX, except for 691.0X). None of the arms have people with any of the other two skin conditions. They were all defined with diagnosis codes, having at least 2 entries each. The dermatologic disease group was selected further using a washout period. We defined our washout period as at least a 180-day period after a person’s first diagnosis on record since the beginning of 2005 during which they received no treatment for their skin condition. Treatments identified during this washout period included biologics, systemic treatments (e.g., methotrexate and cyclosporine), vitamin D analogs, calcineurin inhibitors, topical corticosteroids, anthralin, coal tar, retinoids, and ammonium lactate for psoriasis; systemic treatments, antihistamines (oral and topical), calcineurin inhibitors, topical corticosteroids ± anti-itching or anti-fungal/bacterial agents, and anti-itch treatments (such as calamine lotion) for AD); and oral antibiotics, metronidazole, isotretinoin, retinoids, azelic acid, benzoyl peroxide, calcineurin inhibitors, sulfacetamide products, permethrin and oxymetazoline. This method allowed for creating a homogeneous set of recently diagnosed people with medication prescriptions for their illness.
The control group was comprised of people selected with a simple random sample from the database. None of the people in the control group had any of the three skin diseases. In order to match to the 1-year follow-up period for the dermatologic disease group, the control group was required to have complete data for the whole of 2006.
The cases of cardiovascular disease were defined as having at least 2 diagnosis codes (ICD 9-CM) or at least one diagnosis code plus a procedure code (CPT) for the disease.
Variables and Quantitative variables
We examined several covariates for explanation of variance in the outcome and for their intrinsic interest. Potential confounders included age and gender, number of inpatient days, number of days for which people had outpatient visits, and total health-care utilization costs. Since diabetes is a large risk factor for CVD, we included it as a predictor of CVD, defined by having at least 2 diagnosis codes between 2005 and 2007. Overall comorbid disease severity was examined with the Deyo–Charlson Comorbidity Index with the components related to the outcome removed. Those components were CVD, peripheral vascular disease (PVD), and old myocardial infarction (MI). We also examined the number of comorbid conditions, which could be as high as 14 (as opposed to the 17 conditions usually examined with Deyo–Charlson). In addition to the number of days spent in the hospital, we examined the total number of hospitalizations for the year but did not include this in the regression in order to reduce multi-collinearity. We also looked at the medication costs directly related to the skin conditions as defined in the washout period, for a total of 365 days post-treatment. In addition to summing the costs per person, we looked at the number of prescription drug refills related to these costs for that year. We further examined the total medical costs divided into yearly inpatient, outpatient, and medication costs.
Study Size
The sample size was determined by the exclusion criteria and the eventual propensity score selection. The control group originally was comprised of 20,000 people and many were not included because they did not match well to the dermatologic disease group. Given the large sample size, we did not perform a power analysis prior to the study to estimate the effect size we could detect.
Statistical Methods
The study was designed to examine the independent risk of CVD among the dermatologic conditions compared with a general population of people in a commercial insurance database. Cardiovascular disease outcomes included ischemic heart disease (ICD-9: 410–414), transient cerebral ischemia (ICD-9: 435), heart failure (ICD-9: 428), occlusion and stenosis of pre-cerebral arteries (ICD-9: 433), and occlusion of cerebral arteries (ICD-9: 434). Cases were required to have two diagnosis codes or a diagnosis code plus a cardiovascular-related procedure code (Supplementary Table 1). We used a 365-day follow-up period for the exposure and control in order to standardize the results. Thus, variables such as cost and healthcare utilization will also be generalizable to other studies.
The propensity scores were generated for the three dermatologic disease arms versus the control group, and certain people were selected from the randomly created control group using Mahalanobis matching [11]. All variables, except for the exposure variable, were included in the propensity score calculation. In order to look for a balanced propensity score set, we compared the covariates individually with effect sizes and with a global test designed by Hansen and Bowers [11, 12]. Continuous data were reported as mean (standard deviation) and categorical data were reported as n (%). Analyses were performed in SAS 9.4 (SAS Institute, Cary, NC, USA) and R [12].
Compliance with Ethics Guidelines
This article does not contain any new studies with human or animal subjects performed by any of the authors.
Results
Participants
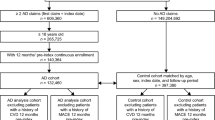
The sample sizes for the disease and control groups were arrived at separately. For the dermatologic disease group, we began with people having either outpatient or inpatient visits with ages inclusive between 30 and 64 years (n = 21,801,147). After excluding patients that lacked diagnosis criteria, sufficient wash-out period, at least 1 year of follow-up, and at least one outpatient record, there were 4263 remaining patients (Fig. 1). For the control group, we began with a simple random sample of 20,000 people from the 2006 database. After excluding those of age less than 30 years or greater than 64 years, with less than 12 months of continuous enrollment, or with a diagnosis of the dermatologic diseases investigated, there were 14,162 patients. Following the propensity score matching with the combined dermatologic disease group, the control sample size was 4263 patients. Our sample consisted of n = 8526 people, matched 1:1 between the dermatologic diseases and control groups.
Descriptive Data and Outcome Data
Our sample was comprised of people between the ages of 30–64 years old with an average age of 49.1 years [standard deviation (SD) 9.0]. Women comprised 67% of our total sample, and 13% of the population had either type I or type II diabetes. The sample had an average of 15.7 days of outpatient visits (SD 15.1), 0.3 days of inpatient visits (SD 1.5), and 0.08 hospitalizations (SD 0.34) per year. The average number of comorbidities was 0.28 (SD 0.57) and the average score of the modified Charlson Comorbidity index was 0.34 (SD 0.8). For health-related expenses, there were an average US$8508 of total costs (SD 16,069), $1049 of inpatient costs (SD 6756), $3925 of outpatient costs (SD 8735), and $1987 of medication costs (SD 3927). For patients diagnosed with AD, psoriasis, or rosacea, an average of 3 dermatologic medications (SD 3) were filled, resulting in an average annual cost of $840 (SD 3261). All covariates, except the number of inpatient and number of outpatient days, had statistically significant differences between the groups (Table 1).
The propensity score matching reduced the Hansen and Bowers global test for balance between treatment and control populations from a Chi-square value of 1155 (p < 0.0001) to 12.4 (p = 0.0528).
Logistic Regression Model
We considered first an effects modification model, and found that the overall test for the variable was insignificant (F = 3.5118, p = 0.3912), and we reported only the main effects model. The model was highly predictive of the outcome with an AUC value of 0.7935. None of the three dermatologic disease groups had statistically significant increased odds of experiencing a CVD event at one year compared to the control group, after controlling for all other covariates (Table 2). In univariate analysis, the unadjusted odds of cardiovascular disease was higher in patients with psoriasis.
Discussion
Systemic inflammation is a risk factor for cardiovascular disease as inflammation accelerates atherosclerosis [13–15]. Rheumatoid arthritis, psoriasis and other diseases associated with chronic systemic inflammation have been associated with increased risk of CVD [16–23]. Our study assessed whether three inflammatory dermatologic conditions: psoriasis, AD, and rosacea contribute to an independent risk of cardiovascular at 1 year following diagnosis.
While psoriasis is associated with an increased risk for cardiovascular outcomes, in this study we did not detect independent increased risk at 1-year follow-up after adjusting for confounders. Prior studies have shown patients with psoriasis and CVD share many risk factors and the heterogeneity of existing studies makes it difficult to determine if psoriasis is truly an independent risk factor for CVD [1]. Association of a disease with confounders does not establish any association with the corresponding disease itself. In our study, patients with psoriasis had an increased risk of developing CVD 1 year after diagnosis before accounting for comorbidities, but this difference vanished after accounting for comorbidities. In our study, limited follow-up of 1 year may also play a role in not identifying psoriasis as an independent risk factor for CVD. The risk between other inflammatory skin conditions (AD and rosacea) has received only limited evaluation.
We found that patients with AD did not have statistically significant independent increased odds of being diagnosed with CVD compared to our propensity-matched control population at 1 year following diagnosis. Silverberg et al. found that patients with atopic dermatitis are more likely to have comorbid (high cholesterol, hypertension, and diabetes) and environmental habits (increased smoking and drinking with decreased activity) that would pre-dispose AD patients to developing CVD [7]; however, AD does not appear to be a significant independent risk factor. Our findings do contrast with the findings from a Taiwanese retrospective population-based study, which found patients with AD having an elevated risk for ischemic stroke (hazards ratio of 1.33) [8], while patients identified with severe AD had a more pronounced risk of developing ischemic stroke (hazards ratio of 1.7) over a follow-up period of 1–4 years compared to their comorbidity-matched counterparts. We suspect that the longer follow-up period in their study may explain the potential discrepancy with our findings.
In our study, patients with rosacea were not statistically more likely to have comorbidities or an independent risk for cardiovascular disease. This corroborates recent findings of Thyssen et al. which also found patients with rosacea were not more likely to have an independent risk of CVD or an increase in CVD risk factors (high cholesterol, LDL, and CRP) and environmental habits (increased smoking and drinking) [24]. A Taiwanese study did find that rosacea was significantly associated with CV comorbidities including dyslipidemia, hypertension, and coronary artery disease, although it lacked adjustment for important CV risk factors such as smoking and alcohol abuse [25].
Limitations
The large sample size gave our study power to find even small clinical effect size. There were several prominent potential confounders for which the database does not collect data, including obesity and smoking and drinking history. Consequently, we expect a degree of residual confounding that may bias our estimate upward from the null. The washout period can potentially select a population with lower severity because it looks for new treatment patients, but this may be balanced by the fact that they are at least being treated by prescription medications. The study had a short follow-up period of 1 year, but there was a cross-section of many patient ages. With younger patients, 1 year may be insufficent to detect an increased risk of CVD. We used a large commercial claims database that is applicable to a large proportion of the population. Because this claims database did not include Medicaid patients, the magnitude of effect may not be generalizable for indigent populations. Insurance claims data do not provide information on systemic inflammatory burden beyond the diagnosis. Consequently, we have no way of measuring the systemic inflammation burden of skin disease.
Conclusions
Among patients diagnosed with one of three common inflammatory dermatologic conditions— psoriasis, AD, and rosacea—we did not detect an independent increased risk of CVD 1 year following diagnosis when compared with a random sample of propensity-matched patients with no evidence of these three diseases. Before accounting for covariates, patients with psoriasis appeared to be at an increased risk for CVD 1 year following diagnosis, but this was not significant after adjusting for confounders. Physicians should be aware of the higher rates of covariates in patients with psoriasis which can predispose to an increased risk of CVD.
References
Miller IM, Ellervik C, Yazdanyar S, Jemec GB. Meta-analysis of psoriasis, cardiovascular disease, and associated risk factors. J Am Acad Dermatol. 2013;69(6):1014–24.
Mehta NN, Li K, Szapary P, Krueger J, Brodmerkel C. Modulation of cardiometabolic pathways in skin and serum from patients with psoriasis. J Transl Med. 2013;11:194.
Gisondi P, Girolomoni G. Cardiometabolic comorbidities and the approach to patients with psoriasis. Actas Dermosifiliogr. 2009;100(Suppl 2):14–21.
Friedewald VE Jr, Cather JC, Gordon KB, Kavanaugh A, Ridker PM, Roberts WC. The editor’s roundtable: psoriasis, inflammation, and coronary artery disease. Am J Cardiol. 2008;101(8):1119–26.
Wakkee M, Thio HB, Prens EP, Sijbrands EJ, Neumann HA. Unfavorable cardiovascular risk profiles in untreated and treated psoriasis patients. Atherosclerosis. 2007;190(1):1–9.
Alexandroff AB, Pauriah M, Camp RD, Lang CC, Struthers AD, Armstrong DJ. More than skin deep: atherosclerosis as a systemic manifestation of psoriasis. Br J Dermatol. 2009;161(1):1–7.
Silverberg JI, Greenland P. Eczema and cardiovascular risk factors in 2 US adult population studies. J Allergy Clin Immunol. 2015;135(3):721–8.
Su VY, Chen TJ, Yeh CM, Chou KT, Hung MH, Chu SY, et al. Atopic dermatitis and risk of ischemic stroke: a nationwide population-based study. Ann Med. 2014;46(2):84–9.
Duman N, Ersoy ES, Atakan N. Rosacea and cardiovascular risk factors: a case control study. J Eur Acad Dermatol Venereol. 2014;28(9):1165–9.
Danielson E. White paper: Health research data for the real world: The MarketScan® databases. Truven Health Analytics. Accessed July 7, 2014.
Feng W, Jun Y, Xu R. A method/macro based on propensity score and Mahalanobis distance to reduce bias in treatment comparison in observational study. 2005. http://www.lexjansen.com/pharmasug/2006/publichealthresearch/pr05.pdf. Accessed Apr 01, 2015.
R Core Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing. http://www.R-project.org/. Accessed Apr 01, 2015.
Willerson JT, Ridker PM. Inflammation as a cardiovascular risk factor. Circulation. 2004;109(21 Suppl 1):II2–10.
Libby P. Inflammation in atherosclerosis. Nature. 2002;420(6917):868–74.
Kaplan RC, Frishman WH. Systemic inflammation as a cardiovascular disease risk factor and as a potential target for drug therapy. Heart Dis. 2001;3(5):326–32.
Dhawan SS, Quyyumi AA. Rheumatoid arthritis and cardiovascular disease. Curr Atheroscler Rep. 2008;10(2):128–33.
Wolfe F, Mitchell DM, Sibley JT, Fries JF, Bloch DA, Williams CA, et al. The mortality of rheumatoid arthritis. Arthritis Rheum. 1994;37(4):481–94.
Solomon DH, Goodson NJ, Katz JN, Weinblatt ME, Avorn J, Setoguchi S, et al. Patterns of cardiovascular risk in rheumatoid arthritis. Ann Rheum Dis. 2006;65(12):1608–12.
Fischer LM, Schlienger RG, Matter C, Jick H, Meier CR. Effect of rheumatoid arthritis or systemic lupus erythematosus on the risk of first-time acute myocardial infarction. Am J Cardiol. 2004;93(2):198–200.
Neimann AL, Shin DB, Wang X, Margolis DJ, Troxel AB, Gelfand JM. Prevalence of cardiovascular risk factors in patients with psoriasis. J Am Acad Dermatol. 2006;55(5):829–35.
Sin DD, Man SF. Why are patients with chronic obstructive pulmonary disease at increased risk of cardiovascular diseases? The potential role of systemic inflammation in chronic obstructive pulmonary disease. Circulation. 2003;107(11):1514–9.
Mosca S, Gargiulo P, Balato N, Di CL, Parente A, Paolillo S, et al. Ischemic cardiovascular involvement in psoriasis: a systematic review. Int J Cardiol. 2015;15(178):191–9.
Evensen K, Slevolden E, Skagen K, Ronning OM, Brunborg C, Krogstad AL, et al. Increased subclinical atherosclerosis in patients with chronic plaque psoriasis. Atherosclerosis. 2014;237(2):499–503.
Egeberg A, Hansen P, Gislason G, Thyssen J. Assessment of the risk of cardiovascular disease in patients with rosacea. J Am Acad Dermatol. 2016;75(2):336–9.
Hua TC, Chung PI, Chen YJ, et al. Cardiovascular comorbidities in patients with rosacea: a nationwide case-control study from Taiwan. J Am Acad Dermatol. 2015;73:249–54.
Acknowledgments
No funding or sponsorship was received for this study or publication of this article. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Disclosures
The Center for Dermatology Research is supported by an unrestricted educational grant from Galderma Laboratories, L.P. S. R. Feldman is a consultant and speaker for Galderma, Stiefel/GlaxoSmithKline, Abbott Labs, Warner Chilcott, Janssen, Amgen, Photomedex, Genentech, BiogenIdec, and Bristol Myers Squibb. S. R. Feldman has received grants from Galderma, Astellas, Abbott Labs, Warner Chilcott, Janssen, Amgen, Photomedex, Genentech, BiogenIdec, Coria/Valeant, Pharmaderm, Ortho Pharmaceuticals, Aventis Pharmaceuticals, Roche Dermatology, 3M, Bristol Myers Squibb, Stiefel/GlaxoSmithKline, Novartis, Medicis, Leo, HanAll Pharmaceuticals, Celgene, Basilea, and Anacor and has received stock options from Photomedex. S. R. Feldman is the founder and holds stock in Causa Research. V. D. Marshall, F. Moustafa, S. Hawkins and R. Balkrishnan have nothing to disclose.
Compliance with Ethics Guidelines
This article does not contain any new studies with human or animal subjects performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/AFD6F06058FA9064.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Marshall, V.D., Moustafa, F., Hawkins, S.D. et al. Cardiovascular Disease Outcomes Associated with Three Major Inflammatory Dermatologic Diseases: A Propensity-Matched Case Control Study. Dermatol Ther (Heidelb) 6, 649–658 (2016). https://doi.org/10.1007/s13555-016-0144-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-016-0144-3