Abstract
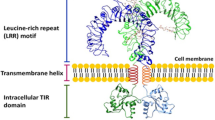
Toll-like receptors (TLRs) recognize specific patterns derived from invading microbial pathogens. TLR activation by microbial components triggers the activation of myeloid differential factor 88 (MyD88)- and tollinterleukin-1 receptor domain-containing adapter inducing interferon-β (TRIF)-dependent downstream signaling pathways, leading to the activation of nuclear factor-κB (NF-κB) and interferon regulatory factor 3 (IRF3). Triptolide (TP), an active ingredient of Tripterygium wilfordii Hook. F, has been reported to have positive effects on autoimmune and inflammatory diseases. To evaluate the therapeutic potential of TP, its effect on signal transduction via the TRIF-dependent pathway of TLRs induced by lipopolysaccharide (LPS) or polyinosinic-polycytidylic acid (poly[I:C]) was examined. TP inhibited NF-κB and IRF3 activation and IRF3 phosphorylation induced by LPS or poly[I:C]. TP also inhibited interferon-inducible genes such as, interferon inducible protein-10. These results suggest that TP can modulate TRIF-dependent signaling pathways of TLRs, and will become promising drug candidates in TLRs-based therapy in the future.
Similar content being viewed by others
References
Medzhitov, R., Preston-Hurlburt, P. & Janeway, C. A. Jr. A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature 388, 394–397 (1997).
Bjorkbacka, H. et al. The induction of macrophage gene expression by LPS predominantly utilizes Myd88-independent signaling cascades. Physiol. Genomics 19, 319–330 (2004).
O’Neill, L. A. & Bowie, A. G. The family of five: TIRdomain-containing adaptors in Toll-like receptor signalling. Nat. Rev. Immunol. 7, 353–364 (2007).
Kawai, T. & Akira, S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat. Immunol. 11, 373–384 (2010).
Takeda, K. & Akira, S. Toll-like receptors in innate immunity. Int. Immunol. 17, 1–14 (2005).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Fitzgerald, K. A. et al. IKKepsilon and TBK1 are essential components of the IRF3 signaling pathway. Nat. Immunol. 4, 491–496 (2003).
Kawai, T. et al. Lipopolysaccharide stimulates the MyD88-independent pathway and results in activation of IFN-regulatory factor 3 and the expression of a subset of lipopolysaccharide-inducible genes. J. Immunol. 167, 5887–5894 (2001).
Gao, J. J. et al. Autocrine/paracrine IFN-alphabeta mediates the lipopolysaccharide-induced activation of transcription factor Stat1alpha in mouse macrophages: pivotal role of Stat1alpha in induction of the inducible nitric oxide synthase gene. J. Immunol. 161, 4803–4810 (1998).
Jiang, X. Clinical observations on the use of the Chinese herb Tripterygium wilfordii Hook for the treatment of nephrotic syndrome. Pediatr. Nephrol. 8, 343–344 (1994).
Chen, B. J. Triptolide, a novel immunosuppressive and anti-inflammatory agent purified from a Chinese herb Tripterygium wilfordii Hook F. Leuk. Lymphoma 42, 253–265 (2001).
Tao, X. L. et al. A prospective, controlled, doubleblind, cross-over study of tripterygium wilfodii hook F in treatment of rheumatoid arthritis. Chin. Med. J. (Engl) 102, 327–332 (1989).
Qiu, D. & Kao, P. N. Immunosuppressive and antiinflammatory mechanisms of triptolide, the principal active diterpenoid from the Chinese medicinal herb Tripterygium wilfordii Hook. f. Drugs R&D 4, 1–18 (2003).
Jiao, J. et al. Triptolide inhibits amyloid-beta1-42-induced TNF-alpha and IL-1beta production in cultured rat microglia. J. Neuroimmunol. 205, 32–36 (2008).
Lu, Y. et al. Inhibitory effect of triptolide on chemokine expression induced by proinflammatory cytokines in human corneal fibroblasts. Invest Ophthalmol. Vis. Sci. 46, 2346–2352 (2005).
Kim, Y. H. et al. Triptolide inhibits murine-inducible nitric oxide synthase expression by down-regulating lipopolysaccharide-induced activity of nuclear factorkappa B and c-Jun NH2-terminal kinase. Eur. J. Pharmacol. 494, 1–9 (2004).
Geng, Y. et al. Triptolide down-regulates COX-2 expression and PGE2 release by suppressing the activity of NF-kappaB and MAP kinases in lipopolysaccharidetreated PC12 cells. Phytother. Res. 26, 337–343 (2012).
Yoneyama, M. et al. Direct triggering of the type I interferon system by virus infection: activation of a transcription factor complex containing IRF-3 and CBP/p300. Embo J. 17, 1087–1095 (1998).
Lin, R., Heylbroeck, C., Pitha, P. M. & Hiscott, J. Virus-dependent phosphorylation of the IRF-3 transcription factor regulates nuclear translocation, transactivation potential, and proteasome-mediated degradation. Mol. Cell. Biol. 18, 2986–2996 (1998).
Lin, R., Heylbroeck, C., Genin, P., Pitha, P. M. & Hiscott, J. Essential role of interferon regulatory factor 3 in direct activation of RANTES chemokine transcription. Mol. Cell. Biol. 19, 959–966 (1999).
Schafer, S. L., Lin, R., Moore, P. A., Hiscott, J. & Pitha, P. M. Regulation of type I interferon gene expression by interferon regulatory factor-3. J. Biol. Chem. 273, 2714–2720 (1998).
Youn, H. S. et al. Specific inhibition of MyD88-independent signaling pathways of TLR3 and TLR4 by resveratrol: molecular targets are TBK1 and RIP1 in TRIF complex. J. Immunol. 175, 3339–3346 (2005).
Park, S. J. & Youn, H. S. Isoliquiritigenin suppresses the Toll-interleukin-1 receptor domain-containing adapter inducing interferon-beta (TRIF)-dependent signaling pathway of Toll-like receptors by targeting TBK1. J. Agric. Food Chem. 58, 4701–4705 (2010).
Youn, H. S. et al. Suppression of MyD88- and TRIFdependent signaling pathways of Toll-like receptor by (-)-epigallocatechin-3-gallate, a polyphenol component of green tea. Biochem. Pharmacol. 72, 850–859 (2006).
Ahn, S. I., Lee, J. K. & Youn, H. S. Inhibition of homodimerization of toll-like receptor 4 by 6-shogaol. Mol. Cells 27, 211–215 (2009).
Park, S. J. et al. Costunolide inhibits cyclooxygenase-2 expression induced by Toll-like receptor 3 or 4 agonist. Toxicol. Environ. Health Sci. 1, 122–126 (2009).
Park, S. J., Lee, M. Y., Son, B. S. & Youn, H. S. TBK1-targeted suppression of TRIF-dependent signaling pathway of Toll-like receptors by 6-shogaol, an active component of ginger. Biosci. Biotechnol. Biochem. 73, 1474–1478 (2009).
Youn, H. S. et al. Sulforaphane suppresses oligomerization of TLR4 in a thiol-dependent manner. J. Immunol. 184, 411–419.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gu, GJ., Youn, HS. Suppression of the TRIF-dependent signaling pathway of toll-like receptor by triptolide. Toxicol. Environ. Health Sci. 5, 177–182 (2013). https://doi.org/10.1007/s13530-013-0173-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13530-013-0173-6