Abstract
Objective
Previous studies have indicated that neurotransmitters play important roles in the occurrence and development of gastric cancer. MAOA is an important catecholamine neurotransmitter-degrading enzyme involved in the degradation of norepinephrine, epinephrine and serotonin. To find a potential therapeutic target for the treatment of gastric cancer, the biological functions of MAOA and the underlying mechanism in gastric cancer need to be explored.
Methods
The Cancer Genome Atlas (TCGA), Gene Expression Omnibus (GEO) datasets, Kaplan‒Meier (KM) plotter were used to identify the differentially expressed genes, which mainly involved the degradation and synthesis enzymes of neurotransmitters in gastric cancer. We also investigated the expression pattern of MAOA in human and mouse tissues and cell lines by immunohistochemistry and Western blotting analysis. Western blotting, quantitative real-time PCR, enzyme-linked immunosorbent assay (ELISA) and a Seahorse experiment were used to identify the molecular mechanism of cancer cell glycolysis. MAOA expression and patient survival were analysed in the Ren Ji cohort, and univariate and multivariate analyses were performed based on the clinicopathological characteristics of the above samples.
Results
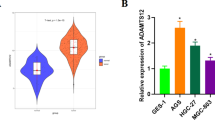
MAOA expression was significantly downregulated in gastric cancer tissue and associated with poor patient prognosis. Moreover, the expression level of MAOA in gastric cancer tissue had a close negative correlation with the SUXmax value of PET-CT in patients. MAOA suppressed tumour growth and glycolysis and promoted cancer cell apoptosis. We also reported that MAOA can interact with NDRG1 and regulate glycolysis through suppression of the PI3K/Akt/mTOR pathway. MAOA expression may serve as an independent prognostic factor in gastric cancer patients.
Conclusions
MAOA attenuated glycolysis and inhibited the progression of gastric cancer through the PI3K/Akt/mTOR pathway. Loss of function or downregulation of MAOA can facilitate gastric cancer progression. Overexpression of MAOA and inhibition of the PI3K/Akt/mTOR pathway may provide a potential method for gastric cancer treatment in clinical therapeutic regimens.






Similar content being viewed by others
Data availability
The data and materials supporting the conclusions of this study are included within the article and its additional files. The public database for this study can be accessed on the following websites: TCGA database (https://cancergenome.nih.gov/), Gene Expression Omnibus (https://www.ncbi.nlm.nih.gov/pubmed) and Kaplan Meier‑Plotter website (http://kmplot.com/analysis/index.php?p=service&cancer=gastric).
Abbreviations
- TCGA :
-
The Cancer Genome Atlas
- GEO :
-
Gene Expression Omnibus
- KM :
-
Kaplan‒Meier
- ELISA :
-
Enzyme-linked immunosorbent assay
- NE :
-
Norepinephrine
- E :
-
Epinephrine
- MAOA :
-
Monoamine oxidase A
- DBH :
-
Dopamine β-hydroxylase
- MAOB :
-
Monoamine oxidase B
- COMT :
-
Catechol-O-methyltransferase
- IHC :
-
Immunohistochemistry
- RT‒qPCR :
-
Reverse transcription-quantitative polymerase chain reaction
- TMA :
-
Tissue microarray
- TNM :
-
Tumor‑Node‑Metastasis
- FFPE :
-
Formalin‑fixed paraffin‑embedded
- OS :
-
Overall survival
- PCNA :
-
Proliferating cell nuclear antigen
- ECAR :
-
Extracellular acidification rate
- OCR :
-
Oxygen consumption rate
References
H. Sung, J. Ferlay, R.L. Siegel, M. Laversanne, I. Soerjomataram, A. Jemal, F. Bray, Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 71(3), 209–249 (2021)
W. Cao, H.D. Chen, Y.W. Yu, N. Li, W.Q. Chen, Changing profiles of cancer burden worldwide and in China: a secondary analysis of the global cancer statistics 2020. Chin. Med. J. (Engl) 134(7), 783–791 (2021)
S.S. Joshi, B.D. Badgwell, Current treatment and recent progress in gastric cancer. CA: Cancer J. Clin. 71(3), 264–279 (2021)
S.M. Gysler, R. Drapkin, Tumor innervation: peripheral nerves take control of the tumor microenvironment. J. Clin. Investig. 131(11), e147276 (2021)
B.W. Renz, R. Takahashi, T. Tanaka, M. Macchini, Y. Hayakawa, Z. Dantes, H.C. Maurer, X. Chen, Z. Jiang, C.B. Westphalen, M. Ilmer, G. Valenti, S.K. Mohanta, A.J.R. Habenicht, M. Middelhoff, T. Chu, K. Nagar, Y. Tailor, R. Casadei, M. Di Marco, A. Kleespies, R.A. Friedman, H. Remotti, M. Reichert, D.L. Worthley, J. Neumann, J. Werner, A.C. Iuga, K.P. Olive, T.C. Wang, beta2 Adrenergic-Neurotrophin Feedforward Loop Promotes Pancreatic Cancer. Cancer Cell 33(1), 75-90.e7 (2018)
J. Pundavela, Y. Demont, P. Jobling, L.F. Lincz, S. Roselli, R.F. Thorne, D. Bond, R.A. Bradshaw, M.M. Walker, H. Hondermarck, ProNGF correlates with Gleason score and is a potential driver of nerve infiltration in prostate cancer. Am. J. Pathol. 184(12), 3156–3162 (2014)
J. Tao, J.N. Campbell, L.T. Tsai, C. Wu, S.D. Liberles, B.B. Lowell, Highly selective brain-to-gut communication via genetically defined vagus neurons. Neuron. 109(13), 2106-2115.e4 (2021)
R.A. Travagli, L. Anselmi, Vagal neurocircuitry and its influence on gastric motility. Nat. Rev. Gastroenterol. Hepatol. 13(7), 389–401 (2016)
J. Calam, J.H. Baron, ABC of the upper gastrointestinal tract: Pathophysiology of duodenal and gastric ulcer and gastric cancer. BMJ 323(7319), 980–982 (2001)
L.E. Hansson, O. Nyren, A.W. Hsing, R. Bergstrom, S. Josefsson, W.H. Chow, J.F. Fraumeni Jr., H.O. Adami, The risk of stomach cancer in patients with gastric or duodenal ulcer disease. N. Engl. J. Med. 335(4), 242–9 (1996)
N.J. Kolla, M. Bortolato, The role of monoamine oxidase A in the neurobiology of aggressive, antisocial, and violent behavior: A tale of mice and men. Prog. Neurobiol. 194, 101875 (2020)
R. McDermott, D. Tingley, J. Cowden, G. Frazzetto, D.D. Johnson, Monoamine oxidase A gene (MAOA) predicts behavioral aggression following provocation. Proc. Natl. Acad. Sci. U.S.A. 106(7), 2118–2123 (2009)
R.L. Sjoberg, F. Ducci, C.S. Barr, T.K. Newman, L. Dell’osso, M. Virkkunen, D. Goldman, A non-additive interaction of a functional MAO-A VNTR and testosterone predicts antisocial behavior. Neuropsychopharmacology 33(2), 425–430 (2008)
L. Passamonti, F. Fera, A. Magariello, A. Cerasa, M.C. Gioia, M. Muglia, G. Nicoletti, O. Gallo, L. Provinciali, A. Quattrone, Monoamine oxidase-a genetic variations influence brain activity associated with inhibitory control: new insight into the neural correlates of impulsivity. Biol. Psychiatry 59(4), 334–340 (2006)
J. Li, X.M. Yang, Y.H. Wang, M.X. Feng, X.J. Liu, Y.L. Zhang, S. Huang, Z. Wu, F. Xue, W.X. Qin, J.R. Gu, Q. Xia, Z.G. Zhang, Monoamine oxidase A suppresses hepatocellular carcinoma metastasis by inhibiting the adrenergic system and its transactivation of EGFR signaling. J. Hepatol. 60(6), 1225–1234 (2014)
Y. Wang, S. Wang, Q. Yang, J. Li, F. Yu, E. Zhao, Norepinephrine Enhances Aerobic Glycolysis and May Act as a Predictive Factor for Immunotherapy in Gastric Cancer. J. Immunol. Res. 2021, 5580672 (2021)
Y. Wang, S. Wang, C. Zhu, H. Cao, Z. Zhang, E. Zhao, The Association Between Immune Characteristic and Clinical Pathology in Chinese Patients with Adenocarcinoma of Esophagogastric Junction. Cancer Manag. Res. 12, 3259–3269 (2020)
Y. Wang, E. Zhao, Z. Zhang, G. Zhao, H. Cao, Association between Tim3 and Gal9 expression and gastric cancer prognosis. Oncol. Rep. 40(4), 2115–2126 (2018)
S.H. Jiang, J. Li, F.Y. Dong, J.Y. Yang, D.J. Liu, X.M. Yang, Y.H. Wang, M.W. Yang, X.L. Fu, X.X. Zhang, Q. Li, X.F. Pang, Y.M. Huo, J. Li, J.F. Zhang, H.Y. Lee, S.J. Lee, W.X. Qin, J.R. Gu, Y.W. Sun, Z.G. Zhang, Increased Serotonin Signaling Contributes to the Warburg Effect in Pancreatic Tumor Cells Under Metabolic Stress and Promotes Growth of Pancreatic Tumors in Mice. Gastroenterology 153(1), 277–291 (2017)
J. Wei, L. Yin, J. Li, J. Wang, T. Pu, P. Duan, T.P. Lin, A.C. Gao, B.J. Wu, Bidirectional Cross-talk between MAOA and AR Promotes Hormone-Dependent and Castration-Resistant Prostate Cancer. Can. Res. 81(16), 4275–4289 (2021)
J. Li, T. Pu, L. Yin, Q. Li, C.P. Liao, B.J. Wu, MAOA-mediated reprogramming of stromal fibroblasts promotes prostate tumorigenesis and cancer stemness. Oncogene 39(16), 3305–3321 (2020)
L. Chen, L. Guo, Z. Sun, G. Yang, J. Guo, K. Chen, R. Xiao, X. Yang, L. Sheng, Monoamine Oxidase A is a Major Mediator of Mitochondrial Homeostasis and Glycolysis in Gastric Cancer Progression. Cancer Manag. Res. 12, 8023–8035 (2020)
M.V. Liberti, J.W. Locasale, The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem. Sci. 41(3), 211–218 (2016)
V. Fresquet, M.J. Garcia-Barchino, M. Larrayoz, J. Celay, C. Vicente, M. Fernandez-Galilea, M.J. Larrayoz, M.J. Calasanz, C. Panizo, A. Junza, J. Han, C. Prior, P. Fortes, R. Pio, J. Oyarzabal, A. Martinez-Baztan, B. Paiva, M.J. Moreno-Aliaga, M.D. Odero, X. Agirre, O. Yanes, F. Prosper, J.A. Martinez-Climent, Endogenous Retroelement Activation by Epigenetic Therapy Reverses the Warburg Effect and Elicits Mitochondrial-Mediated Cancer Cell Death. Cancer Discov. 11(5), 1268–1285 (2021)
A.S. Alzahrani, PI3K/Akt/mTOR inhibitors in cancer: At the bench and bedside. Semin. Cancer Biol. 59, 125–132 (2019)
J. Polivka Jr., F. Janku, Molecular targets for cancer therapy in the PI3K/AKT/mTOR pathway. Pharmacol. Ther. 142(2), 164–175 (2014)
Y. Zhang, P. Kwok-Shing Ng, M. Kucherlapati, F. Chen, Y. Liu, Y.H. Tsang, G. de Velasco, K.J. Jeong, R. Akbani, A. Hadjipanayis, A. Pantazi, C.A. Bristow, E. Lee, H.S. Mahadeshwar, J. Tang, J. Zhang, L. Yang, S. Seth, S. Lee, X. Ren, X. Song, H. Sun, J. Seidman, L.J. Luquette, R. Xi, L. Chin, A. Protopopov, T.F. Westbrook, C.S. Shelley, T.K. Choueiri, M. Ittmann, C. Van Waes, J.N. Weinstein, H. Liang, E.P. Henske, A.K. Godwin, P.J. Park, R. Kucherlapati, K.L. Scott, G.B. Mills, D.J. Kwiatkowski, C.J. Creighton, A Pan-Cancer Proteogenomic Atlas of PI3K/AKT/mTOR Pathway Alterations. Cancer Cell 31(6), 820-832.e3 (2017)
J. Liu, C. Pan, L. Guo, M. Wu, J. Guo, S. Peng, Q. Wu, Q. Zuo, A new mechanism of trastuzumab resistance in gastric cancer: MACC1 promotes the Warburg effect via activation of the PI3K/AKT signaling pathway. J. Hematol. Oncol. 9(1), 76 (2016)
E.S. Villodre, X. Hu, B.L. Eckhardt, R. Larson, L. Huo, E.C. Yoon, Y. Gong, J. Song, S. Liu, N.T. Ueno, S. Krishnamurthy, S. Pusch, D. Tripathy, W.A. Woodward, B.G. Debeb, NDRG1 in Aggressive Breast Cancer Progression and Brain Metastasis. J. Natl. Cancer Inst. 114(4), 579–591 (2022)
K.C. Park, J. Paluncic, Z. Kovacevic, D.R. Richardson, Pharmacological targeting and the diverse functions of the metastasis suppressor, NDRG1, in cancer. Free Radic. Biol. Med. 157, 154–175 (2020)
L. Mi, F. Zhu, X. Yang, J. Lu, Y. Zheng, Q. Zhao, X. Wen, A. Lu, M. Wang, M. Zheng, J. Ji, J. Sun, The metastatic suppressor NDRG1 inhibits EMT, migration and invasion through interaction and promotion of caveolin-1 ubiquitylation in human colorectal cancer cells. Oncogene 36(30), 4323–4335 (2017)
M. Wei, Y. Zhang, X. Yang, P. Ma, Y. Li, Y. Wu, X. Chen, X. Deng, T. Yang, X. Mao, L. Qiu, W. Meng, B. Zhang, Z. Wang, J. Han, Claudin-2 promotes colorectal cancer growth and metastasis by suppressing NDRG1 transcription. Clin. Transl. Med. 11(12), e667 (2021)
E.S. Villodre, Y. Gong, X. Hu, L. Huo, E.C. Yoon, N.T. Ueno, W.A. Woodward, D. Tripathy, J. Song, B.G. Debeb, NDRG1 Expression Is an Independent Prognostic Factor in Inflammatory Breast Cancer. Cancers (Basel) 12(12), 3711 (2020)
M. Weiler, J. Blaes, S. Pusch, F. Sahm, M. Czabanka, S. Luger, L. Bunse, G. Solecki, V. Eichwald, M. Jugold, S. Hodecker, M. Osswald, C. Meisner, T. Hielscher, P. Rubmann, P.N. Pfenning, M. Ronellenfitsch, T. Kempf, M. Schnolzer, A. Abdollahi, F. Lang, M. Bendszus, A. von Deimling, F. Winkler, M. Weller, P. Vajkoczy, M. Platten, W. Wick, mTOR target NDRG1 confers MGMT-dependent resistance to alkylating chemotherapy. Proc. Natl. Acad. Sci. U.S.A. 111(1), 409–414 (2014)
Z. Kovacevic, S. Chikhani, G.Y. Lui, S. Sivagurunathan, D.R. Richardson, The iron-regulated metastasis suppressor NDRG1 targets NEDD4L, PTEN, and SMAD4 and inhibits the PI3K and Ras signaling pathways. Antioxid. Redox Signal 18(8), 874–887 (2013)
K.M. Dixon, G.Y. Lui, Z. Kovacevic, D. Zhang, M. Yao, Z. Chen, Q. Dong, S.J. Assinder, D.R. Richardson, Dp44mT targets the AKT, TGF-beta and ERK pathways via the metastasis suppressor NDRG1 in normal prostate epithelial cells and prostate cancer cells. Br. J. Cancer 108(2), 409–419 (2013)
A.Y. Wu, Y. Hu, W. Cang, D. Li, W.J. Wang, Q. Tian, L.Y. Gu, N. Zhang, F. Ji, L.H. Qiu, Suppressive effect of microRNA-449a on the NDRG1/PTEN/AKT axis regulates endometrial cancer growth and metastasis. Exp. Cell Res. 382(2), 111468 (2019)
X. Dai, Y. Fu, Y. Ye, Increased NDRG1 expression suppresses angiogenesis via PI3K/AKT pathway in human placental cells. Pregnancy Hypertens 21, 106–110 (2020)
J.B. Wu, L. Yin, C. Shi, Q. Li, P. Duan, J.M. Huang, C. Liu, F. Wang, M. Lewis, Y. Wang, T.P. Lin, C.C. Pan, E.M. Posadas, H.E. Zhau, L.W.K. Chung, MAOA-Dependent Activation of Shh-IL6-RANKL Signaling Network Promotes Prostate Cancer Metastasis by Engaging Tumor-Stromal Cell Interactions. Cancer Cell 31(3), 368–382 (2017)
Acknowledgements
We thank Dr. Xue-Li Zhang, Dr. Dong-Xue Li, Dr. Lei Zhu, Dr. Lin-Li Yao, Dr. Yan-Li Zhang, Dr. Hui-Zhen Nie, Dr. Xiao-Mei Yang for assistance with our experiments. We also would like to thank members of the Department of Pathology, Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University (Shanghai, China) for providing assistance.
Funding
This work was supported by the National Natural Science Foundation of China (ID 82103348, to Y.-Y. Wang; ID 82073023, 81871923, to J. Li; ID 82002485, to Q. Li; ID 82103357, to L.-P. Hu), the Shanghai Municipal Education Commission—Gaofeng Clinical Medicine Grant Support (ID 20191809, to J. Li), Shanghai Sailing Program (ID 21YF1445200, to L.-P. Hu), Natural Science Foundation of Shanghai (ID 21ZR1461300, to L.-P. Hu).
Author information
Authors and Affiliations
Contributions
Y.-Y.W. and J.L. designed and supervised the overall study, analyzed data, and drafted the manuscript. Y.-Y.W., Y.-Q.Z., J.-X.X. and X.Z. performed immunohistochemical staining of gastric cancer and normal tissues, quantitative realtime PCR and Western blotting. Q.L., L.-P.H., S.-H.J. and S.-Q.Y. technically assisted with experiments and analyzed data. S.-C.W. Collected patient data and handled patient follow-up. J.X., H.C. and E.-H.Z. provided human gastric cancer and normal tissues. J.L. and Y.-Y.W. supervised this study and edited the manuscript.
Corresponding authors
Ethics declarations
Ethical approval
Written informed consent was obtained from all enrolled patients prior to their inclusion in the study, and the studies were conducted in accordance with Declaration of Helsinki ethical guidelines. The present study was approved by the Ethics Committee of Renji Hospital, Shanghai Jiao Tong University School of Medicine.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, YY., Zhou, YQ., Xie, JX. et al. MAOA suppresses the growth of gastric cancer by interacting with NDRG1 and regulating the Warburg effect through the PI3K/AKT/mTOR pathway. Cell Oncol. 46, 1429–1444 (2023). https://doi.org/10.1007/s13402-023-00821-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-023-00821-w