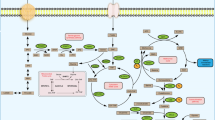
Abstract
Background
Gastric cancer (GC) is the fifth most common cancer worldwide and the most commonly diagnosed cancer in Iran. The nervous system provides proximity to tumor cells by releasing neurotransmitters such as dopamine and presenting them to the corresponding receptor-bearing tumors. While nerve fibers infiltrate the tumor microenvironment, little is known about the expression levels of dopamine (DA), dopamine receptors (DRs), and catechol-O-methyltransferase (COMT) in GC patients.
Methods
DRs and COMT expression were analyzed in 45 peripheral blood mononuclear cells (PBMCs) and 20 paired tumor and adjacent tissue of GC patients by quantitative polymerase chain reaction. DA was measured in plasma specimens using enzyme-linked immunosorbent assay. Protein-protein interaction analysis was carried out to identify GC-related hub genes.
Results
Increased expression of DRD1-DRD3 was found in tumor specimens compared with adjacent non-cancerous specimens (P < 0.05). A positive correlation was found between DRD1 and DRD3 expression (P = 0.009); DRD2 and DRD3 expression (P = 0.04). Plasma levels of dopamine were significantly lower in patients (1298 pg/ml) than in controls (4651 pg/ml). DRD1–DRD4 and COMT were up-regulated in PBMCs of patients compared with controls (P < 0.0001). Bioinformatic analyses showed 30 hub genes associated with Protein kinase A and extracellular signal-regulated kinase signaling pathways.
Conclusions
The findings indicated dysregulation of DRs and COMT mRNA expression in GC and suggest that the brain- gastrointestinal axis may mediate gastric cancer development. Network analysis revealed that combination treatments could be considered for optimizing and improving the precision treatment of GC.






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin 71:209–249
Global Cancer Observatory (2021) Iran-Islamic-republic-of-fact-sheets: the global cancer observatory. International agency for research on cancer. Available from https://gco.iarc.fr/today/data/factsheets/populations/364-iran-islamic-republic-of-fact-sheets
Janbabai G, Yaghoubi Ashrafi M, Mousavi RS, Shekarriz R, Eslami Jouybari M, Zaboli E, Alizadeh-Navaei R, Hedayatizadeh-Omran A, Aarabi M, Qazizadeh Z (2019) Epidemiology of hematopoietic cancers in north of Iran: results of Mazandaran population based cancer registry. WCRJ. https://doi.org/10.32113/wcrj_20194_1270
Hui Y, Tu C, Liu D, Zhang H, Gong X (2023) Risk factors for gastric cancer: a comprehensive analysis of observational studies. Front Public Health. https://doi.org/10.3389/fpubh.2022.892468
De Couck M, Caers R, Spiegel D, Gidron Y (2018) The role of the vagus nerve in cancer prognosis: a systematic and a comprehensive review. J Oncol. https://doi.org/10.1155/2018/1236787
Kamiya A, Hiyama T, Fujimura A, Yoshikawa S (2021) Sympathetic and parasympathetic innervation in cancer: therapeutic implications. Clin Auton Res 31:165–178
Hoffman C, Aballay A (2019) Role of neurons in the control of immune defense. Curr Opin Immunol 60:30–36
Xu H, Shi X, Li X et al (2020) Neurotransmitter and neuropeptide regulation of mast cell function: a systematic review. J Neuroinflamm 17:356
Brougher J, Aziz U, Adari N et al (2021) Self-administration of right vagus nerve stimulation activates midbrain dopaminergic nuclei. Front Neurosci 15:782786
Osinga TE, Links TP, Dullaart RPF et al (2017) Emerging role of dopamine in neovascularization of pheochromocytoma and paraganglioma. Faseb J 31:2226–2240
Craddock N, Owen MJ, O’Donovan MC (2006) The catechol-O-methyl transferase (COMT) gene as a candidate for psychiatric phenotypes: evidence and lessons. Mol Psychiatr 11:446–458
Wang X, Wang Z-B, Luo C et al (2019) The prospective value of dopamine receptors on bio-behavior of tumor. J Cancer 10:1622–1632
Mishra A, Singh S, Shukla S (2018) Physiological and functional basis of dopamine receptors and their role in neurogenesis: possible implication for Parkinson’s disease. J Exp Neurosci 12:1179069518779829
Peters MA, Walenkamp AM, Kema IP, Meijer C, de Vries EG, Oosting SF (2014) Dopamine and serotonin regulate tumor behavior by affecting angiogenesis. Drug Resist Updates 17:96–104
Ahangari GPM, Aminzadeh S, Bakhtou H, Ahmadkhaniha HR (2015) Significant association between catechol amine o-methyl transferase (COMT) gene expression changes and breast cancer pathogenesis. J Carcinog Mutagen 6:219
Zahalka AH, Frenette PS (2020) Nerves in cancer. Nat Rev Cancer 20:143–157
Warde-Farley D, Donaldson SL, Comes O et al (2010) The GeneMANIA prediction server: biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res 38:W214–W220
Hamashima C (2020) The burden of gastric cancer. Ann Translat Med 8:734–734
Sobczuk P, Łomiak M, Cudnoch-Jędrzejewska A (2020) Dopamine D1 receptor in cancer. Cancers (Basel). https://doi.org/10.3390/cancers12113232
Yan Y, Pan J, Chen Y et al (2020) Increased dopamine and its receptor dopamine receptor D1 promote tumor growth in human hepatocellular carcinoma. Cancer Commun (London England) 40:694–710
Zhu S, Soutto M, Chen Z et al (2019) Activation of IGF1R by DARPP-32 promotes STAT3 signaling in gastric cancer cells. Oncogene 38:5805–5816
Mu J, Huang W, Tan Z et al (2017) Dopamine receptor D2 is correlated with gastric cancer prognosis. Oncol Lett 13:1223–1227
Yong M, Yu T, Tian S et al (2017) DR2 blocker thioridazine: a promising drug for ovarian cancer therapy. Oncol Lett 14:8171–8177
Pierce SR, Fang Z, Yin Y et al (2021) Targeting dopamine receptor D2 as a novel therapeutic strategy in endometrial cancer. J Exp Clin Cancer Res 40:61
Liu H, Yang J, Zhang Y et al (2021) Psychologic stress drives progression of malignant tumors via DRD2/HIF1α signaling. Cancer Res 81:5353
Williford SE, Libby CJ, Ayokanmbi A et al (2021) Novel dopamine receptor 3 antagonists inhibit the growth of primary and temozolomide resistant glioblastoma cells. PLoS ONE 16:e0250649
He L, Bhat K, Ioannidis A et al (2021) Effects of the DRD2/3 antagonist ONC201 and radiation in glioblastoma. Radiother Oncol. https://doi.org/10.1016/j.radonc.2021.05.027
Czerwiński M, Bednarska-Czerwińska A, Zmarzły N, Boroń D, Oplawski M, Grabarek BO (2021) miRNAs in the expression regulation of dopamine-related genes and proteins in endometrial cancer. J Clin Med 10:4939
Ptácek R, Kuzelová H, Stefano GB (2011) Dopamine D4 receptor gene DRD4 and its association with psychiatric disorders. Med Sci Monit Int Med J Exp Clin Res 17:RA215–RA220
Rosas-Cruz A, Salinas-Jazmín N, Velázquez MAV (2021) Dopamine receptors in cancer: are they valid therapeutic targets? Technol Cancer Res Treat 20:15330338211027912
Pornour M, Ahangari G, Hejazi SH, Ahmadkhaniha HR, Akbari ME (2014) Dopamine receptor gene (DRD1-DRD5) expression changes as stress factors associated with breast cancer. Asian Pac J Cancer Prev 15:10339–10343
Prabhu VV, Madhukar NS, Gilvary C et al (2019) Dopamine receptor D5 is a modulator of tumor response to dopamine receptor D2 antagonism. Clin Cancer Res 25:2305–2313
Qian X, Zhang D, Cao Z, Ma H (2021) Dopamine pathway mediated by DRD5 facilitates tumor growth via enhancing Warburg effect in esophageal cancer. Front Oncol 11:655861
Leng ZG, Lin SJ, Wu ZR et al (2017) Activation of DRD5 (dopamine receptor D5) inhibits tumor growth by autophagic cell death. Autophagy 13:1404–1419
Kline CLB, Ralff MD, Lulla AR et al (2018) Role of dopamine receptors in the anticancer activity of ONC201. Neoplasia 20:80–91
Bai R, Mei J, Hu W (2020) Hypermethylation of DRD5 promoter is a Biomarker Across 12 Cancer types. DNA Cell Biol 39:2052–2058
Freedman ND, Ahn J, Hou L et al (2009) Polymorphisms in estrogen- and androgen-metabolizing genes and the risk of gastric cancer. Carcinogenesis 30:71–77
Akbarian F, Abolhasani M, Dadkhah F, Asadi F, Ahangari G (2019) Novel insight into differential gene expression and clinical significance of dopamine receptors, COMT, and IL6 in BPH and prostate cancer. Curr Mol Med 19:605–619
Zhang X, Liu Q, Liao Q, Zhao Y (2017) Potential roles of peripheral dopamine in tumor immunity. J Cancer 8:2966–2973
Ahmed Abdelsalam KE, Asad M, Ahmed MAI et al (2021) A case control study on serum levels of potential biomarkers in male breast cancer patients. Int J Environ Res Public Health 18:4852
Basu S, Dasgupta PS (1999) Decreased dopamine receptor expression and its second-messenger cAMP in malignant human colon tissue. Dig Dis Sci 44:916–921
Chakroborty D, Sarkar C, Mitra RB, Banerjee S, Dasgupta PS, Basu S (2004) Depleted dopamine in gastric cancer tissues: dopamine treatment retards growth of gastric cancer by inhibiting angiogenesis. Clin Cancer Res 10:4349–4356
Chakroborty D, Sarkar C, Mitra RB, Banerjee S, Dasgupta PS, Basu S (2004) Depleted dopamine in gastric cancer tissues. Clin Cancer Res 10:4349
Basu S, Nagy JA, Pal S et al (2001) The neurotransmitter dopamine inhibits angiogenesis induced by vascular permeability factor/vascular endothelial growth factor. Nat Med 7:569–574
Chakroborty D, Chowdhury UR, Sarkar C, Baral R, Dasgupta PS, Basu S (2008) Dopamine regulates endothelial progenitor cell mobilization from mouse bone marrow in tumor vascularization. J Clin Investig 118:1380–1389
Kurnik-Łucka M, Pasieka P, Łączak P, Wojnarski M, Jurczyk M, Gil K (2021) Gastrointestinal dopamine in inflammatory bowel diseases: a systematic review. Int J Mol Sci. https://doi.org/10.3390/ijms222312932
Zhu Y, Yuan M, Liu Y et al (2022) Association between inflammatory bowel diseases and Parkinson’s disease: systematic review and meta-analysis. Neural Regen Res 17:344–353
Dai D, Liu H (2022) The nervous system contributes to the tumorigenesis and progression of human digestive tract cancer. J Immunol Res 2022:9595704
Channer B, Matt SM, Nickoloff-Bybel EA et al (2023) Dopamine, immunity, and disease. Pharmacol Rev 75:62–158
Wang K, Zhao XH, Liu J, Zhang R, Li JP (2020) Nervous system and gastric cancer. Biochim Biophys Acta Rev Cancer 1873:188313
Acknowledgements
Our Sincere thanks to all participants for donating samples and Mazandaran University of Medical Sciences and National Institute of Genetic Engineering and Biotechnology for financial support and also Professor Lira Sergio A from Precision Immunology Institute, Icahn School of Medicine at Mount Sinai, New York, the USA for scientific support and critical reading of the manuscript.
Funding
The current research was supported by Mazandaran University of Medical Sciences (Grant No. MAZUMS.7870) and the National Institute of Genetic Engineering and Biotechnology (Grant No.772).
Author information
Authors and Affiliations
Contributions
Conception and design: GA. Data acquisition: EZ. Writing manuscript and performing experiments: OA. Analysis and interpretation of data: OA and EZ. Providing primary tumor and adjacent samples: MMG. Review and/or revision of the manuscript: GJ and AH. Study supervision: GA. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study protocol was approved by the Ethics Committee of Mazandaran University of Medical Sciences (IR.MAZUMS.REC.1400.7870).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Patients voluntarily signed informed consent regarding publishing their data.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Amjadi, O., Hedayatizadeh-Omran, A., Zaboli, E. et al. Dopamine receptors gene overexpression in the microenvironment of invasive gastric cancer and its potential implications. Mol Biol Rep 50, 6529–6542 (2023). https://doi.org/10.1007/s11033-023-08541-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08541-y