Abstract
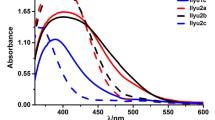
Tetrahedral structures comprising Sn-cored materials with five different types of substituents were synthesized. For the substituents, we employed methyl and tert-butyl as aliphatic groups, and naphthyl and phenyl as aromatic groups. The bandgap is in the range of 3.28 – 3.56 eV. The All the compounds with substituents showed bathochromical photoluminescence characteristics and exhibited aggregation-induced emission characteristics. Specifically, the compounds with aromatic substituents prohibited side-chain packing and π-π stacking. The energy levels of the highest occupied and lowest unoccupied molecular orbitals were measured to be 5.5 – 5.75 and 2.0 – 2.37 eV, respectively. The maximum luminance efficiencies and power efficiencies of the Sn-cored compound-based organic light-emitting diodes (OLEDs) were 0.38 – 0.71 cd/A and 0.15 – 0.28 lm/W. Therefore, it is expected that Sn-cored compounds with a tetrahedral structure, especially those containing aromatic substituents, can be used as an active material in blue OLEDs for prohibiting side-chain packing and π-π stacking.

Similar content being viewed by others
References
H. Sasabe and J. Kido, J. Mater. Chem. C 1, 1699 (2013).
J. Wang, F. Zhang, J. Zhang, W. Tang, A. Tang, H. Peng, Z. Xu, F. Teng, and Y. Wang, J. Photoch. Photobio. C 17, 69 (2013).
J. H. Kim, J.-Y. Cho, J. Park, B. K. Lee, K.-H. Baek, H. Lee, and L.-M. Do, Electron. Mater. Lett. 10, 27 (2014).
G.-P. Kim, B.-M. Park, and H.-J. Chang, Electron. Mater. Lett. 10, 491 (2014).
N.-C. Seong, Y.-M. Jeon, T.-H. Lim, J.-W. Kim, C.-W. Lee, E.-J. Lee, J.-G. Jang, H.-J. Jang, J.-Y. Lee, and M.-S. Gong, Synthetic Met. 157, 421 (2007).
L. Xiao, Z. Chen, B. Qu, J. Luo, S. Kong, Q. Gong, and J. Kido, Adv. Mater. 23, 926 (2011).
K. S. Choi, H. Jo, K. Park, S. Y. Kim, B. H. Koo, K. Hong, and J.-L. Lee, J. Phys. Chem. C 115, 9767 (2011).
Y. Wei and C.-T. Chen, J. Am. Chem. Soc. 129, 7478 (2007).
H.-C. Yeh, R.-H. Lee, L.-H. Chan, T.-Y. J. Lin, C.-T. Chen, E. Balasubramaniam, and Y.-T. Tao, Chem. Mater. 13, 2788 (2001).
S. Wang, W. J. Oldham, R. A. Hudack, and G. C. Bazan, J. Am. Chem. Soc. 122, 5695 (2000).
S.-K. Kim, Y.-I. Park, I.-N. Kang, and J.-W. Park, J. Mater. Chem. 17, 4670 (2007).
S. Liu, F. He, H. Wang, H. Xu, C. Wang, F. Li, and Y. Ma, J. Mater. Chem. 18, 4802 (2008).
S.-C. Lo and P. L. Burn, Chem. Rev. 107, 1097 (2007).
P. Furuta, J. Brooks, M. E. Thompson, and J. M. J. Fréchet, J. Am. Chem. Soc. 125, 13165 (2003).
X.-M. Liu, C. He, X.-T. Hao, L.-W. Tan, Y. Li, and K. S. Ong, Macromolecules 37, 5965 (2004).
Q. He, H. Huang, Q. Sun, H. Lin, J. Yang, and F. Bai, Polym. Advan. Technol. 15, 43 (2004).
Y.-Y. Lyu, J. Kwak, W. S. Jeon, Y. Byun, H. S. Lee, D. Kim, C. Lee, and K. Char, Adv. Funct. Mater. 19, 420 (2009).
Y. Park, K. S. Choi, S. Y. Kim, H. Jo, and K. Park, Displays 33, 55 (2012).
Z. Zhao, S. Chen, X. Shen, F. Mahtab, Y. Yu, P. Lu, J. W. Lam, H. S. Kwok, and B. Z. Tang, Chem. Commun. 46, 686 (2010).
W. Z. Yuan, P. Lu, S. Chen, J. W. Lam, Z. Wang, Y. Liu, H. S. Kwok, Y. Ma, and B. Z. Tang, Adv. Mater. 22, 2159 (2010).
Y. Hong, J. W. Y. Lam, and B. Z. Tang, Chem. Soc. Rev. 40, 5361 (2011).
R. Hu, N. L. Leung, and B. Z. Tang, Chem. Soc. Rev. 43, 4494 (2014).
J. Mei, Y. Hong, J. W. Lam, A. Qin, Y. Tang, and B. Z. Tang, Adv. Mater. 26, 5429 (2014).
C. Kim, T. P. Nguyen, Q. V. Le, J.-M. Jeon, H. W. Jang, and S. Y. Kim, Adv. Funct. Mater. 25, 4512 (2015).
K. Guo, Z. Gao, J. Cheng, Y. Shao, X. Lu, and H. Wang, Dyes Pigments 115, 166 (2015).
C. Liu, W. He, G. Shi, H. Luo, S. Zhang, and Z. Chi, Dyes Pigments 112, 154 (2015).
K. C. Kwon, C. Kim, Q. V. Le, S. Gim, J.-M. Jeon, J. Y. Ham, J.-L. Lee, H. W. Jang, and S. Y. Kim, ACS Nano 9, 4146 (2015).
Z. Zhang, Y. Zhao, R. Zhang, L. Zhang, W. Cheng, and Z. H. Ni, Dyes Pigments 118, 95 (2015).
H. Shi, W. Zhang, X. Dong, X. Wu, Y. Wu, L. Fang, Y. Miao, and H. Wang, Dyes Pigments 104, 34 (2014).
L. Chen, Y. Jiang, H. Nie, P. Lu, H. H. Y. Sung, I. D. Williams, H. S. Kwok, F. Huang, A. Qin, Z. Zhao, and B. Z. Tang, Adv. Funct. Mater. 24, 3621 (2014).
C. Y. Lee, Q. V. Le, C. Kim, and S. Y. Kim, Phys. Chem. Chem. Phys. 17, 9369 (2015).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kim, C., Yoon, MJ., Hong, S.H. et al. Aromatic substituents for prohibiting side-chain packing and π-π stacking in tin-cored tetrahedral stilbenoids. Electron. Mater. Lett. 12, 388–398 (2016). https://doi.org/10.1007/s13391-016-6002-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13391-016-6002-8