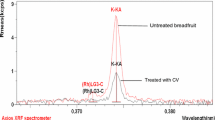
Abstract
The objective of the present paper is to remove Congo red (CR) from wastewater onto Barbary fig skin (BFS) using binary sorption system (\(\hbox {CR}/\hbox {Zn}^{++}\)) at ambient temperature and study the variance of contact time and sorption capacity to that of the singular system. The SEM images, the iodine value (436 mg/g) and the specific surface area (\(368.49\,\hbox {m}^{2}/\hbox {g}\)) show that the used sorbent has a high surface area. The characterization of the surface sorbent by \(^{13}\hbox {CNMR}\), energy-dispersive X-ray, FTIR spectroscopy and Boehm titration test shows the availability of carbonyl, carboxyl, ester and hydroxyl functional groups on this surface as. The Boehm titration test indicates the presence of these functions with almost equivalent amounts (acid groups \(=\) 047 meq \(\hbox {g}^{-1}\), basic groups \(=\) 0.52 meq \(\hbox {g}^{-1}\)). The study of the sorption kinetics indicates a gain of 45 min of contact time in the binary system. The sorption isotherms are very well described by the Langmuir model. According to the Langmuir model, the maximum sorption capacity for CR in binary system (\(Q_{\max } = 161.21 \, \hbox {mg}/\hbox {g}\)) is higher than in singular system (\(Q_{\max } = 151.51 \, \hbox {mg}/\hbox {g}\)). These results show that the sorption of CR onto BFS presents a synergistic phenomenon, not competitive sorption in an aqueous solution which already contains \(\hbox {Zn}^{++}\) (multi-solute system \(\hbox {CR}/\hbox {Zn}^{++}\)).
Similar content being viewed by others
References
Kadrivelu, K.; Kavipriya, M.; Karthika, C.; Radhika, M.; Vennilamani, N.; Pattabhi, S.: Utilization of various agricultural wastes for activated carbon preparation and application for the removal of dyes and metal ions from aqueous solutions. Bioresour. Technol. 87(1), 129–32 (2003)
Jain, A.K.; Gupta, V.K.; Bhatnagar, A.: Suhas.: utilization of industrial waste products as adsorbents for the removal of dyes. J. Hazard. Mater. 101(1), 31–42 (2003)
Aziz, A.; Oualia, M.S.; Elandaloussia, E.; De Menorval, L.C.; Lindheimer, M.: Chemically modified olive stone: a low-cost sorbent for heavy metals and basic dyes removal from aqueous solutions. J. Hazard. Mater. 163, 441–447 (2009)
Natt-Meroug, A.; Benjaballah, A.; Guellati, O.: Préparation et caractérisation d’un charbon actif à base d’un déchet agricole. In: Third International Conference on Energy, Materials, Applied Energetics and Pollution ICEMAEP2016, Constantine, Algeria, October 30–31. (2016)
Esmaili, S.; Nasseri, S.R.; Atash-Dehghan, R.: Adsorption of lead and zinc ions from aqueous solutions by volcanic ash soil (VAS). In: Proceedings of the 8th International Conference Environmental Science and Technology, pp. 8–10 (2003)
Belaroui, K.; Seghier, A.; Hadjel, M.: Synthesis of activated carbon based on apricot stones for wastewater treatment. Desalin. Water Treat. 52, 1422–1433 (2014)
Kovalova, L.; Hansruedi, S.; Von Gunten, U.; Eugster, J.; Hagenbuch, M.; Wittmer, A.; Moser, R.; McArdell, C.S.: Elimination of micropollutants during post-treatment of hospital wastewater with powdered activated carbon, ozone, and UV. Environ. Sci. Technol. 47, 7899–7908 (2013)
Ruhl, A.S.; Zietzschmann, F.; Hilbrandt, I.; Meinel, F.; Altmann, J.; Sperlich, A.; Jekel, M.: Targeted testing of activated carbons for advanced wastewater treatment. Chem. Eng. J. 257, 184–190 (2014)
Khandegar, V.; Saroha, A.K.: Electrocoagulation for the treatment of textile industry effluente a review. J. Environ. Manag. 128, 949–963 (2013)
Liang, C.Z.; Sun, S.P.; Li, F.Y.; Ong, Y.K.; Chung, T.S.: Treatment of highly concentrated wastewater containing multiple synthetic dyes by a combined process of coagulation/flocculation and nanofiltration. J. Membr. Sci. 469, 306–315 (2014)
Cañizares, P.; Martínez, F.; Jiménez, C.; Lobato, J.; Rodrigo, M.A.: Coagulation and electrocoagulation of wastes polluted with dyes. Environ. Sci. Technol. 40(20), 6418–6424 (2006)
Cañizares, P.; Martínez, F.; Rodrigo, M.A.; Jiménez, C.; Sáez, C.; Lobato, J.: Modelling of wastewater electrocoagulation processes: part II: application to dye-polluted wastewaters and oil-in-water emulsions. Sep. Purif. Technol. 60(2), 147–154 (2008)
Ge, Q.; Wang, P.; Wan, C.; Chung, T.S.: Polyelectrolyte-promoted forward osmosisemembrane distillation (FOeMD) hybrid process for dye wastewater treatment. Environ. Sci. Technol. 46, 6236–6243 (2012)
Zhao, P.; Gao, B.; Xu, S.; Kong, J.; Ma, D.; Shon, H.K.; Yue, Q.; Liu, P.: Polyelectrolyte-promoted forward osmosis process for dye wastewater treatment exploring the feasibility of using polyacrylamide as draw solute. Chem. Eng. J. 264, 32–38 (2015)
Punzi, M.; Nilsson, F.; Anbalagan, A.; Svensson, B.M.; Jeonsson, K.; Mattiasson, B.; Jonstrup, M.: Combined anaerobiceozonation process for treatment of textile wastewater: removal of acute toxicity and mutagenicity. J. Hazard. Mater. 292, 52–60 (2015)
Aouni, A.; Fersi, C.; Cuartas-Uribe, B.; Bes-Pía, A.; Alcaina-Miranda, M.I.; Dhahbi, M.: Reactive dyes rejection and textile effluent treatment study using ultrafiltration and nanofiltration processes. Desalination 297, 87–96 (2012)
Ong, Y.K.; Li, F.Y.; Sun, S.P.; Zhao, B.W.; Liang, C.Z.; Chung, T.S.: Nanofiltration hollow fiber membranes for textile wastewater treatment: lab-scale and pilotscale studies. Chem. Eng. Sci. 114, 51–57 (2014)
Zhang, L.; Xu, L.; He, J.; Zhang, J.: Preparation of Ti/\(\text{ SnO }_2\)–Sb electrodes modified by carbon nanotube for anodic oxidation of dye wastewater and combination with nanofiltration. Electrochim. Acta. 117, 192–201 (2014)
Gupta, V.K.; Jain, R.; Mittal, A.; Saleh, T.A.; Nayak, A.; Agarwal, S.; Sikarwar, S.: Photocatalytic degradation of toxic dye amaranth on \(\text{ TiO }_2\)/UV in aqueous suspensions. Mater. Sci. Eng. C 32, 12–17 (2012)
Wang, A.; Li, X.; Zhao, Y.; Wu, W.; Chen, J.; Meng, H.: Preparation and characterizations of \(\text{ Cu }_2\)O/reduced graphene oxide nanocomposites with high photo-catalytic performances. Powder Technol. 261, 42–48 (2014)
Bhatnagar, R.; Joshi, H.; Mall, I.D.; Srivastava, V.C.: Electrochemical treatment of acrylic dye-bearing textile wastewater: optimization of operating parameters. Desalin. Water Treat. 52, 111–122 (2014)
Aquino, J.M.; Rocha-Filho, R.C.; Ruotolo, L.A.; Bocchi, N.; Biaggio, S.R.: Electrochemical degradation of a real textile wastewater using b-\(\text{ Pbo }_2\) and DSA® anodes. Chem. Eng. J. 251, 138–145 (2014)
Ghosh, A.; Dastidar, M.G.; Sreekrishnan, T.R.: Biosorption and biodegradation of chromium complex dye using Aspergillus species. J Hazard. Toxic Radioact. Waste 18(4), 4014–4022 (2014)
Choi, Y.; Park, B.; Cha, D.K.: Enhancing biological treatment of dye wastewater with zero-valent iron. J. Chem. Eng. 32, 1812–1817 (2015)
Bilal, M.; Asgher, M.; Parra Saldivar, S.; Hu, H.; Wang, W.; et al.: Immobilized ligninolytic enzymes: an innovative and environmental responsive technology to tackle dye-based industrial pollutants-a review. Sci. Total Environ. 576, 646–659 (2017)
Chatha, SAS.; Asgher, M.; Iqbal, H.M.N.: Enzyme-based solutions for textile processing and dye contaminant biodegradation-a review. Environ. Sci. Pollut. Res. 24, 14005–14018 (2017)
Bilal, M.; Asgher, M.; Iqbal, H.M.N.; Hu, H.; Zhang, X.: Bio-based degradation of emerging endocrine-disrupting and dye-based pollutants using cross-linked enzyme aggregates. Environ. Sci. Pollut. Res. doi:10.1007/s11356-017-8369-y
Bilal, M.; Iqbal, H.M.N.; Hu, H.; Wang, W.; Zhang, X.: Development of horseradish peroxidase-based cross-linked enzyme aggregates and their environmental exploitation for bioremediation purposes. J. Environ Manag. 188, 137–143 (2017)
Bilal, M.; Iqbal, H.M.N.; Hu, H.; Wang, W.; Zhang, X.: Enhanced biocatalytic performance and dye degradation potential of chitosan encapsulated horseradish peroxidase in a packed bed reactor system. Sci. Total Environ. 575, 1352–1360 (2017)
Asgher, M.; Shah, S.A.H.; Iqbal, H.M.N.: Statistical correlation between ligninolytic enzymes secretion and remazol brilliant yellow-3GL dye degradation potential of Trametes versicolor IBL-04. Water Environ. Res. 88, 338–345 (2016)
Chatha, S.A.S.; Asgher, M.; Iqbal, H.M.N.; Ali, S.: A novel enzymatic stripping of CI reactive black B dyed knitted textiles fabric as an environmentally responsible technology. Electron. J. Biol. 12, 276–281 (2016)
Asgher, M.; Yasmeen, K.; Iqbal, H.M.N.: Enhanced decolorization of Solar brilliant red 80 textile dye by an indigenous white rot fungus Schizophyllum commune IBL-06. Saudi J. Biol. Sci. 20, 347–352 (2013)
Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A.: Adsorption of methylene blue on low-cost adsorbents: a review. J Hazard Mater. 177, 70–80 (2010)
Amran, M.B.; Zulfikar, M.A.: Removal of congo red dye by adsorption onto phyrophyllite. Int. J. Environ. Stud. 67(6), 911–921 (2010)
Jayaraj, R.; Thanaraj, P.J.; Natarajan, S.T.; Prasath, P.M.D.: Removal of congo red dye from aqueous solution using acid activated eco-friendly low cost carbon prepared from marine algae Valoria bryopsis. J. Chem. Pharm. Res. 3(3), 389–396 (2011)
Ghaedi, M.; Shojaeipour, E.; Ghaedi, A.M.; Sahraei, R.: Isotherm and kinetics study of malachite green adsorption onto copper nanowires loaded on activated carbon: artificial neural network modeling and genetic algorithm optimization. Spectroch. Acta Part A Mol Biomol. Spectrosc. 142, 135–149 (2015)
Benderdouche, N.; Bestani, B.; Benstaali, B.; Derrichen, Z.: Enhancement of the adsorptive properties of a desert Salsola vermiculata species. Adsorpt. Sci. Technol. 21(8), 739–750 (2003)
Zou, W.; Liu, L.; Li, H.; Han, X.: Investigation of synergistic adsorption between methyl orange and Cd (II) from binary mixtures on magnesium hydroxide modified clinoptilolite. Korean J. Chem. Eng 33(7), 2073–2083 (2016). doi:10.1007/s11814-016-0048-z
Zou, W.; Bai, H.; Gao, S.: Competitive adsorption of neutral red and \(\text{ Cu }^{2+}\) onto pyrolytic char: isotherm and kinetic study. J. Chem. Eng. Data 57, 2792–2801 (2012)
Kacha, S.; Derriche, Z.; Elmaleh, S.: Equilibrium and kinetics of color removal from dye solutions with bentonite and polyaluminum hydroxide. Water Environ. Res. 75(1), 15–20 (2003)
Malkoc, E.; Nuhoglu, Y.: Removal of Ni(II) ions from aqueous solutions using waste of tea factory: adsorption on a fixed-bed column. J. Hazard. B135, 328–336 (2006)
Sakr, F.; Sennaoui, A.; Elouardi, M.; Tamimi, M.; Assabbane, A.: Adsorption study of Methylene Blue on biomaterial using cactus. J. Mater. Environ. Sci. 6(2), 397–406 (2015)
Zulfikar, M.A.; Setiyanto, H.: Adsorption of congo red from aqueous solution using powdered eggshell. Int. J. ChemTech Res. 5, 1532–1540 (2013)
Seghier, A.; Hadjel, M.; Benderdouche, N.: Adsorption study of heavy metal and acid dye on an amphoteric biomaterial using barbary fig skin. Arab J. Sci. Eng. doi:10.1007/s13369-016-2360-7 (2016)
ASTM D4607-94, Standard Test Method for Determination of Iodine Number of Activated Carbon, (1999)
Boehm, H.P.: Chemical identification of surface groups. Adv. Catal. 16, 179–274 (1966)
Liu, H.; Ning, W.; Cheng, P.; Zhang, J.; Wang, Y.; Zhang, C.: Evaluation of animal hairs-based activated carbon for sorption of norfloxacin and acetaminophen by comparing with cattail fiber-based activated carbon. Anal. Appl. Pyrol. 101, 156–165 (2013)
Bestani, B.; Benderdouche, N.; Benstaali, B.; Belhakem, M.; Addou, A.: Methylene blue and iodine adsorption onto an activated desert plant. Bioresour. Technol. 99, 8441–8444 (2008)
Noszko, L.; Bota, A.; Simay, A.; Nagy, L.: Preparation of activated carbon from the by-products of agricultural industry. Period Polytech. Chem. Eng. 28, 293–297 (1984)
Galiatsatou, P.; Metaxas, M.; Vasilia, K.R.: Adsorption of zinc by activated carbons prepared from solvent extracted olive pulp. J. Hazard. Mater. 91(1–3), 187–203 (2002)
Kobya, M.; Demirbas, M.; Senturk, E.; Ince, M.: Adsorption of heavy metal ions from aqueous solutions by activated carbon prepared from apricot stone. Bioresour. Technol. 96, 1518–1521 (2005)
Namane, A.; Mekarzia, A.; Benrachedi, K.; Bensemra, B.N.; Hellal, A.: Determination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl2 and H3PO4. J. Hazard. Mater. B119, 189–194 (2005)
Shrestha, S.; Son, G.; Lee, S.H.; Lee, T.G.: Isotherm and thermodynamic studies of Zn (II) adsorption on lignite and coconut shell-based activated carbon fiber. Chemosphere 92, 1053–1061 (2013)
Davis, R.D.; Jarrett, W.L.; Mathias, L.J.: Solution \(^{13}\text{ C }\) NMR spectroscopy of pol-yamide homopolymers (nylons 6, 11, 12, 66, 69, 610 and 612) and several commercial co-polymers. Polymer 42, 2621–2626 (2001)
Liua, H.; Gaoa, Q.; Daib, P.; Zhanga, J.; Zhanga, C.; Baoa, N.: Preparation and characterization of activated carbon from lotus stalk with guanidine phosphate activation: sorption of Cd(II). J. Anal. Appl. Pyrolysis. 102, 7–15 (2013)
Li, K.; Zheng, Z.; Huang, X.; Zhao, G.; Feng, J.; Zhang, J.: Equilibrium, kinetic and thermodynamic studies on the adsorption of 2-nitroaniline onto activated carbon prepared from cotton stalk fibre. J. Hazard. Mater. 166, 213–220 (2009)
Knupp, S.L.; Li, W.; Paschos, O.; Murray, T.M.; Snyde, r J.; Haldar, P.: The effect of experimental parameters on the synthesis of carbon nanotube/nanofiber supported platinum by polyol processing techniques. Carbon 46, 1276–1284 (2008)
Morais, S.A.L.; Nascimentoa, E.A.; Queiro, C.R.A.A.; Pilo-Veloso, D.; Drumond, M.G.: Studies on polyphenols and lignin of astronium urundeuvawood. J. Braz. Chem. Soc. 10, 447–452 (1999)
Freundlich, H.M.F.: Over the adsorption in solution. J. Phys. Chem. 57, 385–470 (1906)
Pollard, S.J.T.; Sollars, C.J.; Perry, R.: A low cost adsorbent from spent bleaching earth. I-the selection of an activation procedure. J. Chem. Technol. Biotechnol. 50(2), 265–275 (1991)
Yao, Y.; Xu, F.; Chen, M.; Xu, Z.; Zhu, Z.: Adsorption behavior of methylene blue on carbon nanotubes. Bioresour. Technol. 101, 3040–3046 (2010)
Haghseresht, F.; Lu, G.: Adsorption characteristics of phenolic compounds onto coal-reject-derived adsorbents. Energy Fuels 12, 1100–1107 (1998)
Langmuir, I.: The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 40, 1361–1403 (1918)
Hall, K.; Eagleton, L.; Acrivos, A.; Vermeulen, T.: Pore-and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Ind. Eng. Chem. Fundam. 5, 212–223 (1966)
Murray, J.W.: The surface chemistry of hydrous manganese dioxide. J. Colloid Interface Sci. 46, 357 (1974)
Lackovic, K.; Angove, M.J.; Wells, J.D.; Johnson, B.B.: Modeling the adsorption of Cd (II) onto goethite in the presence of citric acid. J. Colloid Interface Sci. 269, 37–45 (2004)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seghier, A., Hadjel, M. & Benderdouche, N. Comparative Study of the Sorption Capacity and Contact Time of Congo Red Removal in a Binary and Singular System. Arab J Sci Eng 43, 2319–2327 (2018). https://doi.org/10.1007/s13369-017-2722-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-017-2722-9