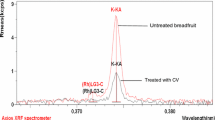
Abstract
In this paper, Barbary fig skin was used which is a natural adsorbent and is low cost, without any chemical or physical activation that causes loss of energy and mass and use of large amount of water for rinsing. The product was characterized by SEM, energy-dispersive X-ray, NMR\(^{13}\)C, and FTIR spectroscopy. In addition, physicochemical parameters to know the iodine value, \(\hbox {pH}_{ (\mathrm{ZPC})}\), and distribution of functional groups on the surface were determined. The material obtained was then used for the treatment of water contaminated by zinc released by zinc plating industrial activities and by an acid red dye (CR) from the textile industry. The results obtained after the study of kinetics, sorption isotherms, the effect of pH, mass and thermodynamic quantities show good efficiency in view of the large sorption capacity to the two treated pollutants (\({Q}_{\mathrm{max}\hbox {-}\mathrm{Zn}}=129.87\) mg g\(^{-1}\), \({Q}_{\mathrm{max}\hbox {-}\mathrm{CR}}=151.51\) mg g\(^{-1}\)). The results found in the course of this study show that the surface of the prepared material has an amphoteric character with the presence of cavities. Freundlich parameters confirm that the adsorption of both pollutants was favorable. The sorption capacity results were compared with those of other adsorbents cited in the literature.
Similar content being viewed by others
References
Booth, S.C.; Workentine, M.L.; Weljie, A.M.; Turner, R.J.: Metabolomics and its application to studying metal toxicity. Metallomics 3, 1142–1152 (2011)
Morris, K.; Zhao, H.J.; John, R.: Ferricyanide-mediated microbial reactions for environmental monitoring. Aust. J. Chem. 58(4), 237–245 (2005)
Pasco, N.F.; Goonerate, R.; Daniel, R.M.; Czollner, A.; Scott, A.J.: Toxicity assessment of chlorophenols using a mediated microbial toxicity assay. Int. J. Environ. Anal. Chem. 88, 1063–1075 (2008)
Tizzard, A.; Webber, J.; Gooneratne, R.; John, R.; Hay, J.; Pasco, N.: MICREDOX: application for rapid biotoxicity assessment. Anal. Chim. Acta 522, 197–205 (2004)
Liu, C.; Sun, T.; Xu, X.; Dong, S.: Direct toxicity assessment of toxic chemicals with electrochemical method. Anal. Chim. Acta 641, 59–63 (2009)
Pasco, N.; Baronian, K.B.; Jeffries, C.; Webber, J.; Hay, J.: MICREDOX\(^{\textregistered }\)—development of a ferricyanide-mediated rapid biochemical oxygen demand method using an immobilised Proteus vulgaris biocomponent Biosens. Biosens. Bioelectron. 20, 524–532 (2004)
Pasco, N.; Hay, J.; Webber, J.: Biosensors MICREDOX—a new biosensor technique for rapid measurement of BOD and toxicity. Biomarkers 6, 83–89 (2001)
Liu, C.; Yong, D.; Yu, D.; Dong, S.: Cell-based biosensor for measurement of phenol and nitrophenols toxicity. Talanta 84, 766–770 (2011)
Yong, D.; Liu, C.; Yu, D.; Dong, S.: A sensitive, rapid and inexpensive way to assay pesticide toxicity based on electrochemical biosensor. Talanta 84, 7–12 (2011)
Yong, D.; Liu, L.; Yu, D.; Dong, S.: Development of a simple method for biotoxicity measurement using ultramicroelectrode array under non-deaerated condition. Anal. Chim. Acta 701, 164–168 (2011)
El Haddad, M.; Slimani, R.; Mamouni, R.; ElAntri, S.; Lazar, S.: Removal of two textile dyes from aqueous solutions onto calcined bones. J Assoc Arab Univ Basic Appl Sci 14, 51–59 (2013)
Mc Kay, G.: Adsorption of dyes tuffs from aqueous solutions with activated carbon. Part l: Equilibrium and batch contact time studies. J. Chem. Technol. Biotechnol. 32, 759–772 (1982)
Mc Kay, G.: Waste colour removal from textile effluents. Am. Dyest. Rep. 68, 29–34 (1979)
Peric, J.; Trgo, M.; Vukojevic Medvidovic, N.: Removal of zinc, copper and lead by natural zeolite—a comparison of adsorption isotherms. Water Res. 38, 1893–1899 (2004)
Rengaraj, S.; Yeon, K.H.; Kang, S.Y.; Lee, J.U.; Kim, K.W.; Moon, S.H.: Studies on adsorptive removal of Co(II), Cr(III) and Ni(II) by IRN77 cation-exchange resin. J. Hazard. Mater. 92, 185–198 (2002)
Zhao, X.; Holl, W.H.; Yun, G.: Elimination of cadmium trace contaminations from drinking water. Water Res. 36, 851–858 (2002)
Gouda, A.A.; Al Ghannam, S.M.: Impregnated multiwalled carbon nanotubes as efficient sorbent for the solid phase extraction of trace amounts of heavy metal ions in food and water samples. Food Chem. 202, 409–416 (2016)
Ihsanullah, Abbas A.; Al-Amer, A.M.; Laoui, T.; Al-Marri, M.J.; Nasser, M.S.; Khraisheh, M.; Atieh, M.A.: Heavy metal removal from aqueous solution by advanced carbon nanotubes: critical review of adsorption applications. Sep. Purif. Technol. 157, 141–161 (2016)
Khare, P.; Yadav, A.; Ramkumar, J.; Verma, N.: Microchannel-embedded metal–carbon–polymer nanocomposite as a novel support for chitosan for efficient removal of hexavalent chromium from water under dynamic conditions. Chem. Eng. J. 293, 44–54 (2016)
Larous, S.; Meniai, A.H.: Removal of copper(II) from aqueous solution by agricultural by-products-sawdust. Energy Proc. 18, 915–923 (2012)
Liu, H.; Ning, W.; Cheng, P.; Zhang, J.; Wang, Y.; Zhang, C.: Evaluation of animal hairs-based activated carbon for sorption of norfloxacin and acetaminophen by comparing with cattail fiber-based activated carbon. Anal. Appl. Pyrolysis 101, 156–165 (2013)
Nowicki, P.; Kazmierczak, J.; Pietrzak, R.: Comparison of physicochemical and sorption properties of activated carbons prepared by physical and chemical activation of cherry stones. Powder Technol. 269, 312–319 (2015)
Liu, H.; Gao, Q.; Dai, P.; Zhang, J.; Zhang, C.; Bao, N.: Preparation and characterization of activated carbon from lotus stalk with guanidine phosphate activation: sorption of Cd(II). Anal. Appl. Pyrolysis 102, 7–15 (2013)
Liu, H.; Wang, X.; Zhai, G.; Zhang, J.; Zhang, C.; Bao, N.; Cheng, C.: Preparation of activated carbon from lotus stalks with the mixture of phosphoric acid and pentaerythritol impregnation and its application for Ni(II) sorption. Chem. Eng. J. 209, 155–162 (2012)
Zhu, J.; Deng, B.; Yang, J.; Gang, D.: Modifying activated carbon with hybrid ligands for enhancing aqueous mercury removal. Carbon 47, 2014–2025 (2009)
Esmi Ferreira, C.; Guevar, C.; Lanie Dhoury, M.; Schrive, L.; Barre, Y.; Palmeri, J.; Deratani, A.: Evaluating the use of activated carbon felts to remove Co\(^{2+}\), Ni\(^{2+}\) and Sr\(^{2+}\) from wastewater. Environ. Chem. Eng. 2, 1705–1712 (2014)
Monday, M.; Adubakar, B.-Y.U.; Zaki, F.U.; Udoji, I.A.: Adsorption of Ni(II) and Zn(II) ions onto activated carbon derived from agricultural waste. Rep. Opin. 3(5), 41–45 (2011)
Aguayo-Villareal, I.A.; Bonilla-Petriciolet, A.; Hernandez-Montrya, V.; Montes-Moran, M.; Reynel-Avila, H.E.: Batch and column studies of Zn\(^{+2}\) ions removal from aqueous solution using chicken feathers as sorbents. Chem. Eng. J. 167, 67–76 (2010)
Issabayeva, G.; Aroua, M.K.: Removal of copper and zinc ions onto biomodified palm shell activated carbon. World Acad. Sci. Eng. Technol. 5(4), 279–279 (2011)
Jamil, R.M.: Adsorption of methyl parathion pesticide from water using watermelon peels as a low cost adsorbent. Chem. Eng. J. 138, 616–621 (2008)
Mahmoodi, N.M.; Salehi, R.; Arami, M.: Binary system dye removal from colored textile wastewater using activated carbon: kinetic and isotherm studies. Desalination 272, 187–195 (2011)
Belaroui, K.; Seghier, A.; Hadjel, M.: Synthesis of activated carbon based on apricot stones for wastewater treatment. Desalin. Water Treat. 52, 1422–1433 (2014)
Namane, A.; Mekarzia, A.; Benrachedi, K.; Bensemra, B.N.; Hellal, A.: Determination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl\(_2\) and H\(_3\)PO\(_4\). J. Hazard. Mater. B 119, 189–194 (2005)
Khan, M.N.; Wahab, M.F.: Characterization of chemically modified corncobs and its application in the removal of metal ions from aqueous solution. J. Hazard. Mater. 141, 237–244 (2007)
Khormaei, M.; Nasernejed, B.; Edrisi, M.; Eslamzadeh, T.: Copper bio sorption from aqueous solution by sour orange residue. J. Hazard. Mater. 149, 269–274 (2007)
Boehm, H.P.: Chemical identification of surface groups. Adv. Catal. 16, 179–274 (1966)
Annadurai, A.; Juang, R.S.; Lee, D.J.: Adsorption of heavy metals from water using banana and orange peels. Water Sci. Technol. 47, 185–190 (2002)
Sakr, F.; Sennaoui, A.; Elouardi, M.; Tamimi, M.; Assabbane, A.: Adsorption study of methylene blue on biomaterial using cactus. J. Mater. Environ. Sci. 6(2), 397–406 (2015)
Daoud, M.; Benturki, O.: Activation d’un charbon à base de noyaux de jujubes et application à l’environnement. Adsorption d’un colorant de textile. Revue des Energies Renouvelables SIENR Ghardaïa, vol. 14, pp. 155–162 (2014)
Li, F.T.; Yang, H.; Zhao, Y.; Xu, R.: Novel modified pectin for heavy metal adsorption. Chin. Chem. Lett. 18, 325–328 (2007)
Farinella, N.V.; Matos, G.D.; Arruda, M.A.Z.: Grape bagasse as a potential biosorbent of metals in effluent treatments. Bioresour. Technol. 98, 1940–1946 (2007)
Ibarra, J.V.; Moliner, R.: Coal characterization using pyrolysis-FTIR. J. Anal. Appl. Pyrolysis 20, 171–184 (1991)
Barka, N.; Ouzaouit, K.; Abdennouri, M.; Makhfouk, M.: Dried prickly pear cactus (Opuntia ficus indica) cladodes as a low-cost and eco-friendly biosorbent for dyes removal from aqueous solutions. J. Taiwan Inst. Chem. Eng. 44, 52–60 (2013)
Morais, S.A.L.; Nascimentoa, E.A.; Queiro, C.R.A.A.; Pilo-Veloso, D.; Drumond, M.G.: Studies on polyphenols and lignin of Astronium urundeuva wood. J. Braz. Chem. Soc. 10, 447–452 (1999)
Aziz, A.; Oualia, M.S.; Elandaloussia, E.; De Menorval, L.C.; Lindheimer, M.: Chemically modified olive stone: a low-cost sorbent for heavy metals and basic dyes removal from aqueous solutions. J. Hazard. Mater. 163, 441–447 (2009)
Ho, Y.S.; McKay, G.: Pseudo-second order model for sorption processes. Process Biochem. 34, 451–465 (1999)
Tsai, W.T.; Yangm, J.M.; Laim, C.W.; Cheng, Y.H.; Lin, C.C.; Yeh, C.W.: Characterization and adsorption properties of eggshells and eggshell membrane. Bioresour. Technol. 97, 488–493 (2006)
Shrestha, S.; Son, G.; Lee, S.H.; Lee, T.G.: Isotherm and thermodynamic studies of Zn(II) adsorption on lignite and coconut shell-based activated carbon fiber. Chemosphere 92, 1053–1061 (2013)
Amran, M.B.; Zulfikar, M.A.: Removal of Congo Red dye by adsorption onto phyrophyllite. Int. J. Environ. Stud. 67(6), 911–921 (2010)
Chatterjee, S.; Lee, D.S.; Lee, M.W.; Woo, S.H.: Enhance adsorption of Congo red from aqueous solutions by chitosan hydrogel beads impregnated with cetyl trimethyl ammonium bromide. Bioresour. Technol. 100, 2803–2809 (2009)
Wang, L.; Wang, A.: Adsorption properties of Congo red from aqueous solution onto surfactantmodified montmorillonite. J. Hazard. Mater. 160, 173–180 (2008)
Patil, A.K.; Shrivastava, V.S.: Alternanthera bettzichiana plant powder as low cost adsorbent for removal of Congo red from aqueous solution. Int. J. ChemTech Res. 2(2), 842–850 (2010)
Namasivayam, C.; Kavitha, D.: Removal of Congo red from water by adsorption onto activated carbon prepared from choir pith, an agricultural solid waste. Dyes Pigm. 54, 47–58 (2002)
Zhang, Z.; Moghaddam, L.; O’Hara, I.M.; Doherty, W.O.S.: Congo red adsorption by ball-milled sugarcane bagasse. Chem. Eng. J. 178, 122–128 (2011)
Binupriya, A.R.; Sathishkumar, M.; Swaminathan, K.; Ku, C.S.; Yun, S.E.: Comparative studies of removal of Congo red by native and modified mycelial pellets of Trametes versicolor in various reactor modes. Bioresour. Technol. 99, 1080–1088 (2008)
Smaranda, C.; Gavrilescu, M.; Bulgariu, D.: Studies on sorption of Congo red from aqueous solution onto soil. Int. J. Environ. Res. 5(1), 177–188 (2011)
Chen, H.; Zhao, J.: Adsorption study for removal of Congo red anionic dye using organo-attapulgite. Adsorption 15, 381–389 (2009)
Mumin, M.A.; Khan, M.M.R.; Akhter, K.F.; Uddin, M.J.: Potentially of open burnt clay as an adsorbent for the removal of Congo red from aqueous solution. Int. J. Environ. Sci. Technol. 4(4), 525–532 (2007)
Jayaraj, R.; Thanaraj, P.J.; Natarajan, S.T.; Prasath, P.M.D.: Removal of Congo red dye from aqueous solution using acid activated eco-friendly low cost carbon prepared from marine algae Valoria bryopsis. J. Chem. Pharm. Res. 3(3), 389–396 (2011)
Zulfikar, M.A.; Setiyanto, H.: Adsorption of Congo red from aqueous solution using powdered eggshell. Int. J. ChemTech Res. 5, 1532–1540 (2013)
Galiatsatou, P.; Metaxas, M.; Vasilia, K.R.: Adsorption of zinc by activated carbons prepared from solvent extracted olive pulp. J. Hazard. Mater. 91(1–3), 187–203 (2002)
Markovska, L.T.; Meshko, V.D.; Marinkovski, M.S.: Modeling of the adsorption kinetics of zinc onto granular activated carbon and natural zeolite. J. Serb. Chem. Soc. 71(8–9), 957–967 (2006)
Kalyani, G.; Rao, G.B.; Saradhi, B.V.; Kumar, Y.P.: Equilibrium and kinetic studies on biosorption of zinc onto Gallus domesticus shell powder. J. Eng. Appl. Sci. 4(1), 39–49 (2009)
Lu, C.; Chiu, H.: Adsorption of zinc(II) from water with purified carbon nanotubes. Chem. Eng. Sci. 61, 1138–1145 (2006)
Hasar, H.; Cuci, Y.; Obek, E.; Dilekoglu, M.F.: Removal of zinc(II) by activated carbon prepared from almond husks under different conditions. Adsorpt. Sci. Technol. 21(9), 799 (2003)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seghier, A., Hadjel, M. & Benderdouche, N. Adsorption Study of Heavy Metal and Acid Dye on an Amphoteric Biomaterial Using Barbary Fig Skin. Arab J Sci Eng 42, 1487–1496 (2017). https://doi.org/10.1007/s13369-016-2360-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-016-2360-7