Abstract
‘Agricultural pests’ is a locution used to indicate those wild species considered as dangerous to crops. The crested porcupine Hystrix cristata is a semi–fossorial rodent considered as one of the main agricultural pests by farmers in Italy. Assessing the role of agriculture in shaping the porcupine’s space use is of great importance to develop proper management and conservation strategies. The goal of this study was to quantify the degree of habitat selection by and suitability for porcupines in Central Italy to assess the potential influence of agricultural fields on the ecology of this rodent. The habitat suitability analysis was realized using presence–only data implemented in the MAXENT Software, while the habitat selection analysis was carried out using the Jacobs’ selectivity index. Our research revealed that croplands have an important role in the overall habitat suitability for porcupines. However, during the warm period, all habitats were used according to their availability. Conversely, orchards, vineyards, and olive groves were preferred during the cold period. Our findings suggest that the use of agricultural crops in accordance with their availability during the warm season and the preference shown for orchards, vineyards, and olive groves during the cold one, highlight that porcupine–famer negative interactions may occur in the area. The implementation of proper prevention measures is therefore strongly suggested, especially during the cold period, to reduce the potential impacts on agriculture.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
‘Human–wildlife conflict’ is a locution commonly used to refer to the negative interactions involving people and wild species (Woodroffe et al. 2005). These includes: (i) actions pursued by wildlife or humans that lead to an adverse effect on the other category (Conover 2002), (ii) real threats imposed by wildlife to human lives (e.g., direct attacks) and/or economic security (Treves and Karanth 2003), or (iii) perceptions that wildlife may constitute a threat for human safety, food, and/or property (Peterson et al. 2011; Dressel et al. 2015; Franchini et al. 2021). Some of the most important drivers of negative interactions between wildlife and humans include land use change to be devoted to livestock practices, and agriculture (Nyhus et al. 2016). Furthermore, ecological interactions between sympatric species relying on the same (or similar) food resources may further increase the likelihood of human–wildlife conflicts (e.g., Polisar et al. 2003; Odden et al. 2010; Franchini and Guerisoli 2023). Large predators are commonly used as an example when discussing human–wildlife negative interactions. Nevertheless, also species defined as ‘agricultural pests’ (i.e., animals that are considered as dangerous to crops) (Waterfield and Zilberman 2012) can cause substantial economic damages (Nyhus et al. 2016).
Although crested porcupine Hystrix cristata (hereafter, porcupine) damages to croplands are negligible compared to other species (Laurenzi et al. 2016), this large rodent is considered as one of the main agricultural pests by farmers in Tuscany (Laurenzi et al. 2016; Cerri et al. 2017; Lovari et al. 2017). In European law, the porcupine has been listed in Appendix II of the Bern Convention and Appendix IV of the ‘Habitat’ Directive since 1978 and 1982, respectively. In Italy, the species has been protected since 1977 (National Law 968/1977) and is listed among the ‘particularly protected species’ in National Law 157/1992. Despite this level of legal protection, this rodent is still heavily poached for its tasty meat, and because of (real or perceived) damages to croplands (Laurenzi et al. 2016; Lovari et al. 2017). The highest damages have been reported to occur primarily in private unprotected vegetable gardens (Laurenzi et al. 2016), leading to resentment among the owners. Moreover, local intolerance towards porcupines has increased due to damages to riverbanks caused by burrow digging (Lovari et al. 2017) and wounds provoked on hunting hounds (Mori et al. 2014a).
‘Habitat suitability’ has been defined as the ability of a habitat to support a viable animal population over an ecological time–scale (Kellner et al. 1992). Conversely, ‘Habitat selection’ has been commonly used to indicate the use of a certain habitat in relation to its local availability (Johnson 1980). Studying habitat selection and suitability is a way of assessing the importance of specific habitats for species expansion and conservation (Mayor et al. 2009; Mori et al. 2021a; Torretta et al. 2021; Frangini et al. 2022). Moreover, it may provide useful information about the role of cultivated fields for a species, thereby allowing to explore the intensity of human–wildlife negative interactions. So far, in Italy, researches on porcupine habitat suitability have been mainly focused on assessing both the current and potential range of expansion for the species (Mori et al. 2021a; Torretta et al. 2021). As for the habitat selection, studies were carried out in small areas of Southern Tuscany and Northern Latium (Sonnino et al. 1998; Mori et al. 2014b; Lovari et al. 2017; Luzi et al. 2021). Due to hostilities towards the species expressed by farmers in Central Italy (Laurenzi et al. 2016; Cerri et al. 2017; Lovari et al. 2017), exploring porcupine habitat selection and suitability at a regional level is necessary to ascertain the species’ habitat requirements and to examine the (potential) impacts on farmlands. This is essential for developing appropriate prevention strategies. The presence of food resources and undisturbed areas are key determinants influencing habitat selection (Mori et al. 2014b; Lovari et al. 2017) and suitability (Mori et al. 2021; Torretta et al. 2021) for this rodent. The porcupine is a ‘generalist species’ feeding not only on underground storage organs (e.g., Cyclamen spp. L., Rumex spp. L., Ruscus aculeatus L.) (Mori et al. 2017), but also on fruits and stems (Lovari et al. 2017; Viviano et al. 2022). Underground storage organs constitute the staple of the diet of porcupines throughout the year (Mori et al. 2017). Conversely, fruits and agricultural products are mainly consumed in the warm season (Mori et al. 2014b, 2017). In fact, during summer, the Mediterranean ‘macchia’ of Central Italy is relatively poor in terms of food (Massei et al. 1997; Lovari et al. 2013), hence prompting porcupines to travel into agricultural lands searching for profitable food resources (Mori et al. 2014b). This behaviour may increase the odds of negative interactions with farmers (Laurenzi et al. 2016; Lovari et al. 2017).
Based on these considerations, the purposes of this study were (i) to assess the impact of agricultural fields on the habitat suitability for porcupines in Central Italy, and (ii) to explore the extent of habitat selection by porcupines, accounting for seasonal variations, to determine whether croplands were preferred, avoided, or used in proportion to their availability in comparison to other habitats. We assumed that cultivated fields constitute an important source of food, especially during the warm season when food resources in natural habitats are more rarefied. Therefore, we predicted to observe a different degree of habitat selection between warm and cold period. In particular, we would expect to observe a higher preference for agricultural areas during the warm period, and a lower preference during the cold one.
Materials and methods
Study area and data collection
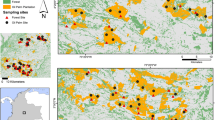
The study was realized in Tuscany (Central Italy) (Fig. 1), where agriculture predominates across the region (Franchini et al. 2022). Depending on the latitude, during the warm season cereals, sunflowers and lucerne represent the most abundant crops (Mori et al. 2014b, 2017). Conversely, in the cold months, only cereals and few cabbage are cultivated.
To conduct the research, we considered porcupine positive locations opportunistically collected from 2012 to 2014 across the region using different data sources: camera–traps (n = 134), road–kills (n = 23), and live captures (n = 12). Although opportunistic records may provide accurate predictions of a species occurrence (Tiago et al. 2017), given the unknown or unbalanced sampling effort (van Strien et al. 2013), they are often discontinuous and/or spatially auto–correlated (Boitani et al. 2011). To reduce those potential biases, we used the ‘spThin’ R package (Aiello-Lammens et al. 2015) to spatially thin our initial dataset and retain the maximum number of locations that were at least 1 km apart. The decision to use a 1 km resolution was made following filtering experiments. This choice aimed to strike a balance between retaining an adequate number of representative samples and ensuring the inclusion of smaller habitat categories that could hold relevance for the species. Moreover, a 1 km distance between locations aligns closely with the average home–range size observed for porcupines in Central Italy (see Habitat suitability and selection analyses—Lovari et al. 2013). The final dataset was composed by n = 93 camera–traps, n = 13 road–kills, and n = 1 live capture, covering an area of about 17,551 km2 and defined through a 100% Minimum Convex Polygon (100% MCP) implemented starting from the coordinates of each location (i.e., camera–traps, road–kills, live capture) (Fig. 1).
Given the absence of seasonal dispersal patterns in the study area (Mori and Fattorini 2019), we conducted the habitat suitability and selection analyses only for adult individuals, presuming their resident movement behaviour, rather than filtering for dispersal periods. The age class of road–kill and captured individuals was determined based on body measurements and maxillary teeth eruption (Mori and Lovari 2014) whereas, as for camera–trapping data, they were all classified as adults based on their body size (Franchini et al. 2022).
Carcases of road–kill porcupines were opportunistically collected from 2012 to 2014 in the Provinces of Arezzo (n = 3), Grosseto (n = 4), and Siena (n = 6) (Supplementary material 1). Captures were carried out within a 24–year research project (1990–2014) aimed at exploring the behavioural ecology of porcupines in Central Italy (Lovari et al. 2013; Mori et al. 2014b, 2015), with captures conducted throughout ten years. During this period, a total of 54 individuals were captured: ten in the Province of Grosseto and 44 in the Province of Siena (Mori et al. 2015). Ten home–made metal box traps (55 × 75 × 100 cm) were used. Traps were baited with vegetables and fruits (apples, carrots, corn, pears, plums, potatoes), activated for at least seven nights per month and checked at sunrise and sunset during each trapping session (five per year, respectively, in 1990, 1991, 1992, 1995, 1997, 1998, 1999, 2012, 2013 and 2014). When an individual was trapped, it was sedated before manipulation following the standard protocol elaborated by Massolo et al. (2003).
As for camera–trapping, we used independent presence–only data obtained from previous researches realized in Tuscany in 2013. Because these studies aimed to explore the ecology of several species (data available on www.inaturalist.org and published works: e.g., Mori et al. 2014c; Franchini et al. 2017; Mori and Menchetti 2019; Viviano et al. 2021), camera–traps were randomly placed in all habitat types (excepting water bodies). Random points were selected through the QGIS (v. 2.0) Software within a regular grid, following the habitat classification obtained from the 2013 land cover map of the Tuscany region (DB UCS 1:10000) (see Habitat suitability and selection analyses). Since cameras were distributed across the entire region (Fig. 1), the average distance among them was approximately 77.3 m (Standard Deviation [SD] = 41.5 m; minimum [min] = 1,030 m; maximum [max] = 228,340 m). Distance parameters (i.e., mean, SD, min, max) were calculated using the distance matrix tool in QGIS (v. 3.28). Cameras were placed along trails and/or near den sites at a height variable from 30 to 50 cm above the ground (tied with ropes and chains, to reduce the likelihood of thefts and/or vandalisms), activated 24 h per day throughout the year to take one video/event and maximize the detection probability, and checked every 10–15 days to download data and replace batteries. Cameras were rotated amongst stations at least once every 40 days, to ensure that each camera trap station was sampled for at least 30 days per season. The overall sampling effort was 4,319 camera–trap nights, during which an average of 38 (SD = 22) independent porcupine events were recorded. A time span of 30 min between pictures at the same site was considered to define an independent event (Meek et al. 2014).
Habitat suitability and selection analyses
Based on 2013 land cover map of the Tuscany region (DB UCS 1:10000), habitats were re–classified into 18 different percentage share of land cover classes (PLAND—Table 1).
Since our goal was to investigate the role of agriculture in shaping the porcupine habitat selection and suitability, we first evaluated its influence in the habitat suitability model. Because this environmental covariate made a substantial contribution (see Supplementary material 2 and Results), the habitat selection analysis was carried out. As a first step, we built a MAXENT model (Phillips et al. 2006) focusing on two Software outputs, i.e., the variable importance score and the jackknife test plot of variable importance (see Supplementary material 2). These two metrics enabled us to evaluate the contribution of agricultural fields to the model construction. We used the 18 environmental covariates representing the percentage share of the re–classified land cover categories (Table 1) as 1.5 × 1.5 km raster layers, to reproduce the average home–range for a porcupine in Central Italy (Lovari et al. 2013). Multicollinearity among land cover categories was checked through the Variance Inflation Factor (VIF) and considering VIF ≥ 5 to indicate the presence of covariates presenting severe collinearity issues (Akinwande et al. 2015). We run the MAXENT model through the standalone Software using default settings and a 10–fold cross validation as model evaluation (Phillips 2017). Presence data (represented by porcupine positive locations) were compared with 10,000 background locations.
To realize the habitat selection analysis, a buffer with a radius of 849 m was applied to each porcupine location based on the average home–range for an individual in Central Italy (Lovari et al. 2013). The porcupine seasonal habitat selection was investigated using the Jacobs’ selectivity index (Jacobs 1974):
where r is the proportion of porcupines captured/road–killed/camera–trapped in each habitat, and p is the proportion of habitats available within the buffer areas (i.e., sum of each habitat calculated within each buffer).
The index ranges from − 1 (complete avoidance) to + 1 (exclusive use) with preference or underuse declared only at values > 0.3 or < -0.3, respectively (Laurenzi et al. 2016; Lovari et al. 2017).
As for the seasonal habitat selection, we compared warm (spring, summer) vs. cold (autumn, winter) season classified as follows: autumn (October, November, December), winter (January, February, March), spring (April, May, June), summer (July, August, September). Before using the index, the significant difference in habitat use compared to the available habitats was assessed using Fisher’s exact test (Fisher 1922) due to all values in the contingency table being < 5.
The analyses were carried out using the QGIS (v. 3.28) and R Software (v. 4.3.1) (R Development Core Team 2023) and the level of significance was set at 0.05.
Results
Following PLAND values calculated for each buffer, the habitat was mainly covered by broad–leaved forests, cultivated fields and urban areas, respectively (Table 2). The total number of porcupine positive locations was higher during the warm season (n = 69, 64.5%) and lower during the cold one (n = 38, 35.5%) (Table 2).
All the 18 environmental covariates showed VIF ≤ 5. Therefore, none was discarded in the MAXENT model. The model performance was poor (AUC = 0.63 ± 0.08 (SE), as values typically indicating good model performance are ≥ 0.7 (Greiner et al. 2000; Vanagas 2004). However, the percent contribution of each environmental covariate and jackknife plots (see Supplementary material 2) highlighted that agricultural fields hold relevance in the model construction, thus empirically justifying the habitat selection analysis realized through the Jacobs’ selectivity index.
During the warm season, all habitats were used according to their availability (Fisher’s test, p = 0.1). Conversely, during the cold one, habitat use differed from the expected (Fisher’s test, p = 0.01). Arboriculture, cliff areas, coastal areas, moors and shrublands, and open pastures and grasslands were totally avoided (D = -1); broad–leaved forests (D = -0.4) and cultivated fields (D = -0.5) were underused; while agroforestry (D = 0.7), coniferous forests (D = 0.5), lagoons and swamps (D = 0.4), mixed forests (D = 0.4), orchards, vineyards, and olive groves (D = 0.3), and urban areas (D = 0.6) were preferred. Areas with woody and shrubby vegetation in evolution (D = 0.1) where instead used according to their availability (Fig. 2).
Habitat selection (Jacobs’ selectivity index) by porcupines during the cold season (autumn, winter). Positive (> 0.3) or negative (< -0.3) values (horizontal red lines) indicate preference or underuse, respectively, towards a certain habitat. Areas with sparse vegetation as well as sclerophyllous vegetation, were not included because they were absent within the calculated buffers
Discussion
Habitat suitability
The contribution given by urban areas and cultivated fields in the habitat suitability model contrasts with Mori’s et al. (2021a) research where they observed that an increase in farmlands and urbanization may limit the range of expansion of porcupines. In fact, dry areas (like urban ones) are generally considered as unsuitable for this rodent since they impede digging and burrowing behaviours (Mori et al. 2013). Conversely, this finding is in accordance with the study conducted by Torretta et al. (2021) where they have shown that cultivars are frequently highly heterogeneous and intermixed with patches of natural or semi–natural habitats, thus providing abundant and diversified food resources. Moreover, peri–urban environments are used by the species long as they are linked to cultivated fields for both feeding and denning (Mori et al. 2022a). Given that natural habitats in Tuscany are highly fragmented, interspersed with agricultural and urban areas (Franchini et al. 2022), we consider that the region’s diverse habitat supports ample shelter and food resources for porcupines. This heterogeneous landscape context thus likely contributes to the suitability of the area for the species.
Seasonal habitat selection
During the warm season, all habitats were used according to their availability, thus rejecting our initial assumption. Although not considered optimal habitats for porcupines (Mori et al. 2014b; Torretta et al. 2021), croplands are used during the warm months (a period coinciding with reduced food availability in natural habitats— Mori et al. 2014b, 2017; Lovari et al. 2017), being rich in food resources (e.g., cereals, lucerne, figs, sunflowers, watermelons) (Mori et al. 2017). The use of agricultural areas in accordance with their availability during the warm season might be attributed to seasonal shifts in territorial behaviour exhibited by this rodent (Mori et al. 2014b). An ecological process called ‘seasonal contraction’ dictates that seasonal variations in food availability force porcupines to increase or decrease their home–range size when trophic resources are near or far from the den (Lovari et al. 2013). Specifically, during the warm period, underground plant storage organs may be difficult to reach due to sun–baked soil. This could force porcupines to expand their home–ranges in search of more accessible food resources (Lovari et al. 2013; Mori et al. 2014b). Consequently, this expanded roaming behaviour may drive the species to explore and use a wide spectrum of habitats. An alternative explanation lies in the porcupine’s generalist feeding behaviour, allowing the species to consume a wide array of vegetables and organs like fruits, roots, and stems (Bruno and Riccardi 1995; Mori et al. 2021b, for a review) based on their local seasonal availability.
As for the cold period and with reference to agricultural habitats, the results obtained revealed complete avoidance for arboriculture, a lower than expected use of cultivated fields, and a preference for orchards, vineyards, and olive groves, thus partially matching our initial assumption. As stated above, the use of these habitats depends on the amount of food available in natural environments (e.g., woodlands), which varies depending on the season (Mori et al. 2014b, 2017; Lovari et al. 2017). During the cold season, porcupines mainly forage on underground storage organs of woody plants (Mori et al. 2017; Viviano et al. 2022). However, cultivars could also be consumed during this period when prevalence of different parasite species tend to be higher, in turn leading to higher parasite infections. Therefore, those plants (including those producing fruits) having antiparasitic medical properties are frequently consumed (Viviano et al. 2022). This may explain why, during the cold months, certain agricultural crops are avoided or underused, while others (like orchards, vineyards, and olive groves) are preferred. Furthermore, vineyards and fruit orchards provide easily accessible food resources, since fruits fallen on the ground and/or those growing on low branches constitute an important part of the porcupine’s diet (Mori et al. 2017; Lovari et al. 2017). The preference shown for agroforestry areas may find explanation in the feeding ecology of this rodent, searching for underground storage organs in canopy–covered areas (Mori et al. 2017; Viviano et al. 2022) regardless of their origin (i.e., wild or cultivated). Nevertheless, another possible explanation may be linked to the ongoing climatic crisis which may alter the species’ feeding behaviour (Pettorelli et al. 2007; Lovari et al. 2020). During cold periods characterized by severe droughts, porcupines may encounter difficulty in foraging for food through digging, in turn prompting the species to resort to typically avoided food sources, such as tree barks (Laurenzi et al. 2016; Mori et al. 2022b). Tree debarking is mainly oriented towards plant species (e.g., Quercus pubescens Willd., Robinia pseudoacacia L., Sambucus nigra L.) presenting soft barks with medical properties (Mori et al. 2022b). Therefore, the positive or negative selection of cultivated forestry areas may also depend on the characteristics of the cultivated tree species. The work carried out by Mori et al. (2022b) was realized using data collected between March 2021 and February 2022, when Central Italy was affected by a severe drought. Our research included data collected from 2012 to 2014, years in which Tuscany (and other Italian regions) experienced drought events (ARPAT—Agenzia Regionale per la Protezione Ambientale in Toscana). In over 30 years of studies conducted on the spatial ecology and/or feeding behaviour of porcupines (e.g., Corsini et al. 1995; Mori et al. 2014a, b, c, 2017, 2022a; Laurenzi et al. 2016; Lovari et al. 2013, 2017), stem debarking has been only rarely observed (Laurenzi et al. 2016). However, field surveys conducted in Tuscany during the winter of 2012–2013 (Mori et al. 2014d) indicated that two snowfall events, with snow depth ranging from 20 to 40 cm, might have stimulated the debarking behaviour observed in porcupines (Laurenzi et al. 2016). Moreover, most of the above studies were mainly conducted in Central/Southern Tuscany. Therefore, the likelihood of porcupines engaging in tree debarking behaviour across other areas of the region (in connection with the severity of seasonal drought) might have been underestimated.
The preference observed for coniferous forests contrasts with Lovari’s et al. (2017) study, where it was found that porcupines typically avoid coniferous woodlands due to limited shelters and/or food resources. However, fruits of some hard epicarp species (e.g., pine nuts) are consumed during the cold season (Mori et al. 2017), in turn explaining the higher occurrence of porcupine in coniferous forests during this period. The preference shown for lagoons and wetlands does not match the results achieved by Mori et al. (2014b), where they found that these habitats are generally underused by the species. Nevertheless, occasional visits may occur by certain individuals, particularly sub–adults, for the creation of temporary or seasonal burrows (Pigozzi and Patterson 1990; Mori and Assandri 2019). The preference for mixed forests might be attributed to the porcupines’ inclination to feed on underground storage organs (e.g., Cyclamen spp., Rumex spp., Ruscus aculeatus L.) or fruits of understory vegetal species. These resources are commonly found in areas where both coniferous and deciduous plants coexist (Mori et al. 2017). The preference shown for urban areas is in contrast with other studies (Lovari et al. 2017; Mori et al. 2017; Viviano et al. 2020; Luzi et al. 2021) where a general avoidance of heavily human–altered environments was reported. However, despite habitat degradation, urban areas may host a high species diversity (e.g., Collins et al. 2000; Chace and Walsh 2004; Magle et al. 2012). Furthermore, the use of urban areas by porcupines has notably expanded in recent years (Manenti et al. 2020; Mori et al. 2022a), particularly when these areas are connected to cultivated lands (Mori et al. 2022a). In fact, porcupines exhibit a preference for thorny thickets in these settings for denning purposes and to exploit seasonal food resources (Lovari et al. 2017; Mori et al. 2022a). For instance, figs (especially during summer) are particularly attractive for porcupines, which in turn get close to human infrastructures to feed on them (Lovari et al. 2017). Nevertheless, because this represents a ‘high risk, high gain’ strategy, porcupines aim to minimize their time spent within or near human areas to mitigate the risk of poaching (Lovari et al. 2017).
The total avoidance of cliff and coastal areas aligns with findings from Mori et al. (2014b), indicating these habitats are typically avoided due to their scarcity of resources, including food and shelters. The total avoidance of moors and shrublands is in contrast with Mori et al. (2017) and Lovari et al. (2017). Shrub woods are generally positively selected by porcupines for both feeding (Lovari et al. 2017) and denning (Mori et al. 2017). Nevertheless, positive or negative selection depends on the plant species occurring in the area (Lovari et al. 2017). For instance, Rubus ulmifolius Schott., Prunus spinosa L., and Spartium junceum L. are positively selected. Digging dens within these dense thorny bushes, might indeed serve as a strategy to minimize the risk of poaching due to the challenging accessibility of these habitats (Lovari et al. 2017). On the other hand, garigue habitats are generally avoided as they are deemed unsuitable for porcupines (Mori et al. 2014b). We speculate that the total avoidance of shrub habitats, as observed, might be due to the unsuitable conditions associated with the plant species occurring in the area. Additional researches conducted at a more detailed landscape scale are necessary to offer comprehensive insights. The total avoidance of open pastures and grasslands might indicate the species’ preference for habitats (such as scattered landscapes) that offer a more diverse array of food resources and denning sites (Lovari et al. 2013; Mori et al. 2014b, 2017; Luzi et al. 2021).
The underuse of broad–leaved forests is in contrast with other studies realized in Central Italy (Lovari et al. 2017; Mori et al. 2014b, 2022b). Underground vegetal organs constitute the staple of the diet of porcupines and are mostly abundant in deciduous woodlands (Mori et al. 2017, 2021). The generalist feeding behaviour of the rodent allow it to feed on a large variety of plant materials exploiting a wide spectrum of habitats (Bruno and Riccardi 1995; Mohamed 2011), may underlie it.
Conclusions
To our knowledge, this study marks the initial endeavour to evaluate the extent of habitat suitability and selection by porcupines across the whole Tuscany region, investigating the significance of cultivated fields for this species, in turn exploring the potential patterns of negative human–porcupine interactions in Tuscany.
Despite porcupines may feed on cultivars during the warm months (when food resources in more natural habitats are limited—Mori et al. 2014b, 2017; Lovari et al. 2017), this cannot be considered as a ‘rule of thumb’ since our findings revealed that, during the warm period, all habitats are used according to their availability. On the contrary, orchards, vineyards and olive groves were positively selected during the cold season. In Tuscany, agricultural fields cover about 50% of the total available habitats, and from 1750 to 1855 porcupines have been estimated in the area (Franchini et al. 2022). Despite our findings revealed moderate use of cultivars during the warm season, the implementation of proper prevention measures is strongly recommended, especially during the cold season, to reduce the odds of potential damages.
Despite we recognize the importance of the findings achieved in the exploration of human–porcupine negative interactions in Central Italy, we also recognize the potential limits of our work: (i) despite relying on few monitored individuals, the research carried out by Sonnino et al. 1998; Mori et al. 2014b; Lovari et al. 2017 included radio–tagged individuals, a method which can provide more detailed information about habitat use/selection as well as individual movements. For instance, the observed preference for coniferous forests during the cold season, a habitat typically deemed unsuitable for porcupines (Lovari et al. 2017), might also be associated with using this habitat as a transitional area; (ii) diet analyses (not realized in this research) may have provided additional useful information about the role of cultivars (and other habitats) in shaping the species habitat use/preference. In fact, the species’ presence (or preference) in/towards a certain habitat, does not necessarily imply occurrence of damages; (iii) the avoidance of moors and shrublands might be influenced by the availability of food and plant species within those areas (Mori et al. 2014b, 2017; Lovari et al. 2017). Similarly, certain cultivated fields (e.g., cereals, lucerne, figs, sunflowers, watermelons) might experience greater consumption compared to others. However, the level of landscape detail necessary to differentiate between these distinctions is not provided in the habitat classification outlined in the 2013 land cover map of the Tuscany region. These findings imply that investigating the habitat use and selection by porcupines should occur on a finer scale, also considering the species’ feeding behaviour. This approach would better delineate the role of each habitat in influencing the porcupine’s ecology and intensity of negative interactions with local farmers.
Data availability
The dataset generated and analysed during this study is available from the corresponding author on reasonable request.
References
Agenzia Regionale per la Protezione Ambientale in Toscana (2024) (ARPAT)—https://www.arpat.toscana.it/ (last accessed January 14th
Aiello-Lammens ME, Boria RA, Radosavljevic A, Vilela B, Anderson RP (2015) spThin: an R package for spatial thinning of species occurrence records for use in ecological niche models. Ecography 38(5):541–545. https://doi.org/10.1111/ecog.01132
Akinwande MO, Dikko HG, Samson A (2015) Variance inflation factor: as a condition for the inclusion of sup-pressor variable(s) in regression analysis. Open J Stat 05:754–767. https://doi.org/10.4236/ojs.2015.57075
Boitani L, Maiorano L, Baisero D, Falcucci A, Visconti P, Rondinini C (2011) What spatial data do we need to develop global mammal conservation strategies? Philos Trans R Soc Lond B Biol Sci 366(1578):2623–2632. https://doi.org/10.1098/rstb.2011.0117
Bruno E, Riccardi C (1995) The diet of the crested porcupine Hystrix cristata L., 1758 in a Mediterranean rural area. Z Säugetierk 60:226–236
Cerri J, Mori E, Vivarelli M, Zaccaroni M (2017) Are wildlife value orientations useful tools to explain tolerance and illegal killing of wildlife by farmers in response to crop damage? Eur J Wildl Res 63:70. https://doi.org/10.1007/s10344-017-1127-0
Chace JF, Walsh JJ (2004) Urban effects on native avifauna: a review. Landsc Urb Plann 74:46–69. https://doi.org/10.1016/j.landurbplan.2004.08.007
Collins JP, Kinzing A, Grimm NB, Fagan WF, Hope D, Wu J, Borer ET (2000) A new urban ecology. Modeling human communities as integral parts of ecosystems poses special problems for the development and testing of ecological theory. Am Sci 88:416–424. https://www.jstor.org/stable/27858089
Conover MR (2002) Resolving Human-Wildlife conflicts: the Science of Wildlife damage management. CRC, Boca Raton, FL
Corsini MT, Lovari S, Sonnino S (1995) Temporal activity patterns of crested porcupines Hystrix cristata. J Zool 236:43–54. https://doi.org/10.1111/j.1469-7998.1995.tb01783.x
R Development Core Team (2023) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Dressel S, Sandström C, Ericsson G (2015) A meta-analysis of studies on attitudes toward bears and wolves across Europe 1976–2012. Conserv Biol 29(2):565–574. https://doi.org/10.1111/cobi.12420
Fisher RA (1922) On the Interpretation of χ2 from Contingency Tables, and the Calculation of P. J R Stat Soc 85(1):87–94. https://pdfs.semanticscholar.org/0c15/62db88551e9412b314616995c7bc94a6d65b.pdf
Franchini M, Guerisoli MM (2023) Interference competition driven by co-occurrence with tigers Panthera tigris may increase livestock predation by leopards Panthera pardus: a first step meta-analysis. Mamm Rev. https://doi.org/10.1111/mam.12323
Franchini M, Fazzi P, Lucchesi M, Mori E (2017) Diet of adult and juvenile wildcats in Southern Tuscany (Central Italy). Folia Zool 66(2):147–151. https://doi.org/10.25225/fozo.v66.i2.a1.2017
Franchini M, Corazzin M, Bovolenta S, Filacorda S (2021) The return of large carnivores and extensive farming systems: a review of stakeholders’ perception at an EU level. Animals 11:1735. https://doi.org/10.3390/ani11061735
Franchini M, Viviano A, Frangini L, Filacorda S, Mori E (2022) Crested porcupine (Hystrix cristata) abundance estimation using bayesian methods: first data from a highly agricultural environment in Central Italy. Mamm Res 67:187–197. https://doi.org/10.1007/s13364-022-00622-w
Frangini L, Sterrer U, Franchini M, Pesaro S, Rüdisser J, Filacorda S (2022) Stay home, stay safe? High habitat suitability and environmental connectivity increases road mortality in a colonizing mesocarnivore. Landsc Ecol 37:2343–2361. https://doi.org/10.1007/s10980-022-01491-z
Greiner M, Pfeiffer D, Smith RD (2000) Principles and practical application of the receiver-operating characteristic analysis for diagnostic tests. Prev Vet Med 45:23–41. https://doi.org/10.1016/S0167-5877(00)00115-X
Jacobs J (1974) Quantitative measurement of food selection: a modification of the forage ratio and Ivlev’s electivity index. Oecologia 14:413–417. https://doi.org/10.1007/BF00384581
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resource preference. Ecology 61:65–71. https://doi.org/10.2307/1937156
Kellner CJ, Brawn JD, Karr JR (1992) What is habitat suitability and how should it be measured? In: McCullough DR, Barrett RH (eds) Wildlife 2001: populations. Springer, Dordrecht, pp 476–488
Laurenzi A, Bodino N, Mori E (2016) Much ado about nothing: assessing the impact of a problematic rodent on agriculture and native trees. Mammal Res 61:65–72. https://doi.org/10.1007/s13364-015-0248-7
Lovari S, Sforzi A, Mori E (2013) Habitat richness affects home range size in a monogamous large rodent. Behav Proc 99:42–46. https://doi.org/10.1016/j.beproc.2013.06.005
Lovari S, Corsini MT, Guazzini B, Romeo G, Mori E (2017) Suburban ecology of the crested porcupine in a heavily poached area: a global approach. Eur J Wildl Res 63:10. https://doi.org/10.1007/s10344-016-1075-0
Lovari S, Franceschi S, Chiatante G, Fattorini L, Fattorini N, Ferretti F (2020) Climatic changes and the fate of mountain herbivores. Clim Change 162:2319–2337. https://doi.org/10.1007/s10584-020-02801-7
Luzi G, Mori E, Puddu G, Zapparoli M (2021) Does the crested porcupine select coppice forest? Habitat preference and activity patterns of a large rodent in the Lago Di Vico Natural Reserve. Mammalia 85(4):355–361. https://doi.org/10.1515/mammalia-2020-0143
Magle SB, Hunt VM, Vernon M, Crooks KR (2012) Urban wildlife research: past, present, and future. Biol Cons 155:23–32. https://doi.org/10.1016/j.biocon.2012.06.018
Manenti R, Mori E, Di Canio V, Mercurio S, Picone M, Caffi M, Brambilla M, Ficetola GF, Rubolini D (2020) The good, the bad and the ugly of COVID-19 lockdown effects on wildlife conservation: insights from the first European locked down country. Biol Cons 249:108728. https://doi.org/10.1016/j.biocon.2020.108728
Massei G, Genov PV, Staines BW, Gorman ML (1997) Factors influencing home range and activity of wild boar (Sus scrofa) in a Mediterranean coastal area. J Zool 242:411–423. https://doi.org/10.1111/j.1469-7998.1997.tb03845.x
Massolo A, Sforzi A, Lovari S (2003) Chemical immobilization of crested porcupine with tiletamine HCl and zolazepam HCl (Zoletil) under field condition. J Wildl Dis 39:727–731. https://doi.org/10.7589/0090-3558-39.3.727
Mayor SJ, Schneider DC, Schaefer JA, Mahoney SP (2009) Habitat selection at multiple scales. Ecoscience 16:238–247. https://doi.org/10.2980/16-2-3238
Mohamed WF (2011) The crested porcupine, Hystrix cristata (Linnaeus, 1758) in Misurata, Libya. J J Ecol Nat Environm 3:228–231. https://doi.org/10.5897/JENE.9000103
Mori E, Assandri G (2019) Coming back home: recolonisation of abandoned dens by crested porcupine Hystrix cristata and European badgers Meles meles after wood-cutting and riparian vegetation mowing events. Hystrix 30:39–43. https://doi.org/10.4404/hystrix-00176-2019
Mori E, Fattorini N (2019) Love getaway: dispersal pattern and distance of the crested porcupine. Mamm Res 64:529–534. https://doi.org/10.1007/s13364-019-00438-1
Mori E, Lovari S (2014) Sexual size monomorphism in the crested porcupine (Hystrix cristata). Mammal Biol 79:157–160. https://doi.org/10.1016/j.mambio.2013.07.077
Mori E, Menchetti M (2019) Living with roommates in a shared den: spatial and temporal segregation among semifossorial mammals. Behav Processes 164:48–53. https://doi.org/10.1016/j.beproc.2019.04.013
Mori E, Sforzi A, Di Febbraro M (2013) From the Apennines to the Alps: recent range expansion of the crested porcupine Hystrix cristata L., 1758 (Mammalia: Rodentia: Hystricidae) in Italy. Ital J Zool 80:469–480. https://doi.org/10.1080/11250003.2013.857729
Mori E, Maggini I, Menchetti M (2014a) When quills kill: the defense strategy of the crested porcupine Hystrix cristata L., 1758. Mammalia 78:229–234. https://doi.org/10.1515/mammalia-2013-0126
Mori E, Lovari S, Sforzi A, Romeo G, Pisani C, Massolo A, Fattorini L (2014b) Patterns of spatial overlap in a monogamous large rodent, the crested porcupine. Behav Processes 107:112–118. https://doi.org/10.1016/j.beproc.2014.08.012
Mori E, Menchetti M, Dondini G, Biosa D, Vergari S (2014c) Theriofauna of site of community importance Poggi Di Prata (Grosseto, Central Italy): terrestrial mammals and preliminary data on Chiroptera. Check List 10(4):718–723. https://doi.org/10.15560/10.4.718
Mori E, Menchetti M, Dartora F (2014d) Evidence of the carrion consumption behaviour in the long-eared owl Asio otus (Aves: Strigiformes: Strigidae). Ital J Zool 81:471–475. https://doi.org/10.1080/11250003.2014.920928
Mori E, Sforzi A, Menchetti M, Mazza G, Lovari S, Pisanu B (2015) Ectoparasite load in the crested porcupine Hystrix cristata Linnaeus, 1758 in Central Italy. Parasitol Res 114(6):2223–2229. https://doi.org/10.1007/s00436-015-4413-3
Mori E, Bozzi R, Laurenzi A (2017) Feeding habits of the crested porcupine Hystrix cristata L. 1758 (Mammalia, Rodentia) in a Mediterranean area of Central Italy. Eur Zoological J 84(1):261–265. https://doi.org/10.1080/24750263.2017.1329358
Mori E, Ficetola GF, Bartolomei R, Capobianco G, Varuzza P, Falaschi M (2021a) How the South was won: current and potential range expansion of the crested porcupine in Southern Italy. Mamm Biol 101:11–19. https://doi.org/10.1007/s42991-020-00058-2
Mori E, Di Gregorio M, Mazza G, Ficetola GF (2021b) Seasonal consumption of insects by the crested porcupine in Central Italy. Mammalia 85:231–235. https://doi.org/10.1515/mammalia-2020-0131
Mori E, Molteni R, Ancillotto L, Ficetola GF, Falaschi M (2022a) Spatial ecology of crested porcupine in a metropolitan landscape. Urban Ecosyst 25:1797–1803. https://doi.org/10.1007/s11252-022-01264-1
Mori E, Viviano A, Scarfò M, Preti F, Pucci C, Ciuti F, Giovannelli A, Traversi ML, Senserini D, Mazza G (2022b) Bark stripping behaviour by a large-sized rodent, the crested porcupine, as an adaptation to climate change. Biologia 77:3525–3529. https://doi.org/10.1007/s11756-022-01243-z
Nyhus PJ (2016) Human–wildlife conflict and coexistence. Annu Rev Environ Resour 41:143–171. https://doi.org/10.1146/annurev-environ-110615-085634
Odden M, Wegge P, Fredriksen T (2010) Do tigers displace leopards? If so. why? Ecol Res 25:875–881. https://doi.org/10.1007/s11284-010-0723-1
Peterson MN, Birckhead JL, Leong K, Peterson MJ, Peterson TR (2011) Rearticulating the myth of human–wildlife conflict. Conserv Lett 3:74–82. https://doi.org/10.1111/j.1755-263X.2010.00099.x
Pettorelli N, Pelletier F, von Hardenberg A, Festa-Bianchet M, Cote SD (2007) Early onset of vegetation growth vs. rapid green-up: impacts on juvenile mountain ungulates. Ecology 88:381–390. https://doi.org/10.1890/06-0875
Phillips SJ (2017) A Brief Tutorial on Maxent. Available from url: http://biodiversityinformatics.amnh.org/open_source/maxent/. Accessed on 2023-07-01
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190(3–4):231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Pigozzi G, Patterson IJ (1990) Movements and diet of crested porcupines in the Maremma National Park, Central Italy. Acta Theriol 35:173–180. https://doi.org/10.4098/AT.ARCH.90-21
Polisar J, Maxit I, Scognamillo D, Farrel L, Sunquist ME, Eisenberg JF (2003) Jaguars, pumas, their prey base, and cattle ranching: ecological interpretations of a management problem. Biol Conserv 109:297–310. https://doi.org/10.1016/S0006-3207(02)00157-X
Sonnino S (1998) Spatial activity and habitat use of crested porcupine, Hystrix cristata L., 1758 (Rodentia, Hystricidae) in Central Italy. Mammalia 62(2):775–759. https://doi.org/10.1515/mamm.1998.62.2.175
Tiago P, Pereira HM, Capiñha C (2017) Using citizen science data to estimate climatic niches and species distributions. Basic Appl Ecol 20:75–85. https://doi.org/10.1016/j.baae.2017.04.001
Torretta E, Orioli V, Bani L, Mantovani S, Dondina O (2021) En route to the North: modelling crested porcupine habitat suitability and dispersal flows across a highly anthropized area in northern Italy. Mamm Biol. https://doi.org/10.1007/s42991-021-00155-w
Treves A, Karanth KU (2003) Human-Carnivore conflict and perspectives on Carnivore management worldwide. Conserv Biol 17:1491–1499. https://doi.org/10.1111/j.1523-1739.2003.00059.x
van Strien AJ, van Swaay CAM, Termaat T (2013) Opportunistic citizen science data of animal species produce reliable estimates of distribution trends if analysed with occupancy models. J Appl Ecol 50(6):1450–1458. https://doi.org/10.1111/1365-2664.12158
Vanagas G (2004) Receiver operating characteristic curves and comparison of cardiac surgery risk stratification systems. Interact Cardiovasc Thorac Surg 3:319–322. https://doi.org/10.1016/j.icvts.2004.01.008
Viviano A, Amori G, Luiselli L, Oebel H, Bahleman F, Mori E (2020) Blessing the rains down in Africa: spatiotemporal behaviour of the crested porcupine Hystrix cristata (Mammalia: Rodentia) in the rainy and dry seasons, in the African savannah. Trop Zool 33(4):113–124. https://doi.org/10.4081/tz.2020.80
Viviano A, Mori E, Fattorini N, Mazza G, Lazzeri L, Panichi A, Strianese L, Mohamed WF (2021) Spatiotemporal overlap between the European brown hare and its potential predators and competitors. Animals 11:562. https://doi.org/10.3390/ani11020562
Viviano A, Huffman MA, Senini C, Mori E (2022) Do porcupines self–medicate? The seasonal consumption of plants with antiparasitic properties coincides with that of parasite infections in Hystrix cristata of Central Italy. Eur J Wildl Res 68:72. https://doi.org/10.1007/s10344-022-01620-8
Waterfield G, Zilberman D (2012) Pest management in food systems: an economic perspective. Annu Rev Environ Resour 37:223–245. https://doi.org/10.1146/annurev-environ-040911-105628
Woodroffe R, Thirgood S, Rabinowitz A (2005) People and Wildlife: conflict or coexistence? Cambridge, UK. Cambridge University Press
Acknowledgements
We are very grateful to prof. Sandro Lovari (University of Siena) for realizing the capture sessions and providing the original data.
Funding
As for captures, the financial support was provided by the Provincial Councils of Siena and Grosseto.
Open access funding provided by Università degli Studi di Udine within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
MF, LF, SF and EM conceived the study; EM collected data in the field; LF conducted the habitat suitability analysis; MF conducted the habitat selection and statistical analyses; MF wrote the first draft of the manuscript. All authors participated in writing, review and editing the final version of the manuscript and approved it before submission.
Corresponding author
Ethics declarations
Ethical approval
The ethical approval was obtained by the National Institute for Environmental Protection and Research (ISPRA). Permits for captures were released by both the Environment Ministry and the Provincial Councils of Grosseto and Siena. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
On behalf of all authors, the corresponding author states that there are no conflicts of interest.
Additional information
Communicated by Magdalena Niedziałkowska.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Franchini, M., Frangini, L., Viviano, A. et al. Get out from my field! The role of agricultural crops in shaping the habitat selection by and suitability for the crested porcupine in Central Italy. Mamm Res 69, 411–421 (2024). https://doi.org/10.1007/s13364-024-00744-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13364-024-00744-3