Abstract
Japanese farmers who grow Oriental persimmon, Diospyros kaki Thunb. (Ericales: Ebenaceae), occasionally introduce Apis mellifera L. (Hymenoptera: Apidae) hives into their orchards during the flowering season, mainly because cultivars such as ‘Fuyu’ require pollination for stable fruit set. However, wild pollinators might serve this function. Thus, we aimed at identifying the most important pollinators of D. kaki in Japan by surveying visitation frequency across ten prefectures and pollination efficiency. The predominant flower visitors were Bombus ardens ardens Smith (Hymenoptera: Apidae) and A. mellifera, with similar pollination efficiencies. The estimated threshold number of pollen grains for fruit set of D. kaki ‘Fuyu’ was 27, which was comparable to the number deposited by a single visit of either species, meaning one visit of these dominant visitors probably induces fruit set. However, more pollen grains deposited by multiple visits further ensured fruit set. To achieve > 80% fruit set, more than about 70 pollen grains were needed. These results indicate that several visits by the bees would be sufficient for fruit set of D. kaki ‘Fuyu’.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The European honeybee, Apis mellifera L. (Hymenoptera: Apidae), is the representative domesticated pollinator in fruit production worldwide (Garratt et al. 2016; Rollin and Garibaldi 2019). Many farmers have introduced honeybee hives into their orchards to increase and ensure pollination. An increasing body of evidence, however, has revealed that a greater diversity of wild pollinators provide pollination service to crops (Garibaldi et al. 2013). Introducing numerous honeybee hives into orchards regardless of the presence of native wild pollinators could incur an unnecessary cost to farmers and even harm populations of wild flower visitors (Eeraerts et al. 2017; Huryn 1997; Sugden and Pyke 1991; Valido et al. 2019). Farmers cultivating Oriental (or Japanese) persimmon, Diospyros kaki Thunb. (Ericales: Ebenaceae), have also conventionally introduced hives of A. mellifera into their orchards to alleviate the burden of hand-pollination and to guarantee a good crop yield every year, though the honeybees’ effect has been controversial (Fukae et al. 1987; Nakamura et al. 2020; Yokozawa 1952; Yokozawa et al. 1968).
Some cultivars of D. kaki, such as 'Fuyu', require pollination to ensure fruit set. ‘Fuyu’ is the most widely grown in Japan (29.9% in 2018, including ‘Matsumotowase-Fuyu’, the early ripening bud sport of ‘Fuyu’; Statistics Bureau, Ministry of Internal Affairs and Communications, Japan 2021). Since it produces only female flowers, they need other pollinizer cultivars to receive pollen. Although ‘Fuyu’ sets seedless fruits without pollination, those fruits easily drop early, and the number of matured seedless fruits in a tree fluctuates greatly between years (Yamada et al. 1987). Furthermore, seedless fruits are slightly flatter than seeded ones (Kitajima et al. 1993). Thus, pollination of ‘Fuyu’ decreases physiological fruit drop and stabilizes fruit yield (Kajiura 1941; Yamada et al. 1987). After a certain number of seeds, however, the production of more seeds does not always reduce the risk of early fruit drop. Yakushiji and Hase (1991) reported that two or three seeds were enough to avoid fruit abscission. Increased seed number per fruit, to a maximum of eight, is not correlated with the size and quality of ‘Fuyu’ (Kitajima et al. 1993; Woodburn and Andersen 1996). In addition, a fruit with many seeds is less favored by consumers. Therefore, fruit containing a couple of seeds would be ideal for constant production of high-quality ‘Fuyu’ fruit. As for other cultivars, necessity of pollination for increasing fruit set and/or improving the quality of fruit vary depending on the cultivars (Moriguchi et al. 2006; Suzuki and Niikawa 2018; Woodburn and Andersen 1996; Yamamura 1982; Yamamura and Osaki 1982).
Our previous studies suggested that a bumblebee, Bombus ardens ardens Smith (Hymenoptera: Apidae), was a common wild pollinator of Oriental persimmon at some study sites in western Japan, whereas the dominance of A. mellifera as a pollinator fluctuated among years (Nakamura et al. 2020; Nikkeshi et al. 2019). Miyamoto (1961) also reported that B. ardens ardens was a persimmon flower visitor. However, since these research sites are limited to western Japan (i.e., Hiroshima, Hyogo, and Shimane prefectures), more field research across a wider area of Japan is needed to clarify the common visitors to D. kaki flowers in Japan.
In addition to visitation frequency, pollination efficiency is another critical criterion for identifying the most important pollinator of the crop (Ne’eman et al. 2010). Nakamura et al. (2020) showed that the number of pollen grains on the body surface of A. mellifera was far less than that of B. ardens ardens. In addition, the number of matured seeds was positively correlated with the number of visitations by B. ardens ardens but not by A. mellifera. These findings suggest that A. mellifera is less efficient at transporting pollen. However, the authors did not precisely survey pollination efficiency. The best measure of pollination efficiency of a species is the number of pollen grains deposited on a stigma of the crop by a single visit by an individual of the species (King et al. 2013). Therefore, to clarify whether pollination service is sufficient in an orchard or not, we must first evaluate both visitation frequency and pollination efficiency of the major flower visitors, and calculate the necessary number of pollen grains on a stigma for guaranteed fruit set.
Our main objective here was to identify the most important visitors to D. kaki over a wide area of Japan to understand the natural pollination system and environmentally sustainable management of this crop. First, we assessed the species assemblage of flower-visiting insects and their visitation frequencies by monitoring and capturing them on D. kaki flowers in ten orchards in different prefectures. Second, to evaluate the pollination efficiency of each major flower-visiting species, we compared the number of pollen grains deposited on a stigma by a single visit between major visitor species. Third, we evaluated the effectiveness of a single visit of the major pollinator on fruit set by comparing the fruit-set probability among pollination treatments. Finally, we estimated the minimum and sufficient numbers of pollen grains required for fruit set by analyzing the relationship between the number of pollen grains deposited on a stigma and the following fruit set.
Methods
Study sites
We monitored flower-visiting insects in six experimental fields in Fukuoka (33.50° N, 130.57° E), Gifu (35.44° N, 136.70° E), Hiroshima (34.33° N, 132.82° E), Kagawa (34.30° N, 133.93° E), Kumamoto (32.64° N, 130.72° E), and Wakayama (34.28° N, 135.41° E) prefectures and at four farms in Fukushima (37.82° N, 140.44° E), Ibaraki (36.27° N, 140.25° E), Shimane (34.69° N, 131.87° E), and Shizuoka (34.86° N, 137.74° E) prefectures. We monitored cultivars ‘Aizu-mishirazu’ (Fukushima), ‘Fuyu’ (Hiroshima), ‘Fuyu’ and ‘Matsumotowase-Fuyu’ (Ibaraki), ‘Fuyu’ and ‘Taishuu’ (Fukuoka and Kagawa), ‘Saijo’ (Shimane), ‘Soshu’ (Gifu), ‘Taishuu’ (Kumamoto and Wakayama), and ‘Tateishi’ (Shizuoka). These cultivars were selected for monitoring of flower-visitors because enough flowers were available in the study sites. No domesticated pollinators were placed in any of the fields except for the farm in Ibaraki, where two hives of A. mellifera were placed. For detailed information about the study sites, see Table S1.
We performed pollination experiments in the experimental field in Hiroshima (Site A) and the farm in Ibaraki (Site B), which were also used for the monitoring of flower-visiting insects, and at a farm in Shimane (Site C; 35.46° N, 132.80° E). ‘Fuyu’ was used for the pollination experiments at Sites A and B and ‘Matsumotowase-Fuyu’ at Site C. Detailed information about Sites A–C is given in Table S1.
Identification of flower-visiting species
We captured 453 flower-visiting insects in the 10 orchards; each was captured in a 5-mL vial placed on the flower that it had entered. Insects were captured on different trees from those used for monitoring if the tasks were conducted simultaneously. We recorded the sex of the flowers on which insects were captured.
The captured insects were morphologically and genetically analyzed. We first identified collected insects to the species level on the basis of morphological traits, and then confirmed if necessary by COI DNA barcoding. The primer combination used for PCR amplification and sequencing of COI of the bees was BarbeeF (forward) and MtD9 (reverse) (Françoso and Arias 2013; Simon et al. 1994). That used for the others was LCO1490 (forward) and HCO2198 (reverse) (Folmer et al. 1994). The PCR products were treated with ExoSAP-IT (USB Corp., Cleveland, OH, USA) and directly sequenced in both directions. Similar sequences were searched using “All Barcode Records on BOLD” (http://www.boldsystems.org/index.php/IDS_OpenIdEngine) in BOLD (the Barcode of Life Data Systems) or the Basic Local Alignment Search Tool (BLAST; https://blast.ncbi.nlm.nih.gov/Blast.cgi) in GenBank (NCBI, National Centre for Biotechnology Information). Species were identified on the basis of highly similar best matches (> 98% identity). The sequences were deposited in the DNA Data Bank of Japan (DDBJ accession numbers LC682290–LC682345; see Table S2). For details of the procedure, see Nikkeshi et al. (2019).
Monitoring flower visitors
Visits by insects to D. kaki flowers were recorded in 2019, with the exception of the farm in Ibaraki that was monitored in 2021. The ten surveyed prefectures all ranked in the upper half of persimmon-producing prefectures in Japan (Statistics Bureau, Ministry of Internal Affairs and Communications, Japan 2021). Monitoring was performed at the peak blooming date or a few days earlier. We set several monitoring plots in a study site. Each monitoring plot measured approximately 2 m width × 2 m height, which we found to be the maximum area in which any researcher could adequately track all visitors in a plot. Tree height was approximately 3 to 4 m. We set each monitoring plot at eye level, because it was difficult to monitor the flowers on the upper branches. After the numbers of male and female flowers in a plot had been recorded, visits by insects to the flowers in the plot were recorded for 30 min. Every flower-visiting insect was observed and classified into five species that can be identified by appearance: Apis cerana japonica Radoszkowski, A. mellifera, B. ardens ardens, Bombus diversus diversus Smith, and Xylocopa appendiculata circumvolans Smith (all Hymenoptera: Apidae); the others were classified by functional group: medium-sized bees, small bees, dipterans including syrphid flies, and beetles. Consecutive visitations to a flower by a single individual were counted independently. Each monitoring plot was surveyed several times.
Annual fluctuations of flower visitation frequency and fruit set
To examine the range of annual fluctuations of fruit set, the frequency of flower visitors to D. kaki and fruit set were monitored over 4 years (2018–2021) in Hiroshima, where pollination experiments were also carried out (Site A; see Table S1). The flower visitors were monitored as above. To assess fruit set, we conducted pollination experiments on flowers, with a no pollination (NP) treatment in which female flower buds were covered with paper bags before and during anthesis and an open-pollination (OP) treatment in which flower buds were tagged and kept uncovered. Each year, the female flower buds were thinned beforehand to one per shoot. In 2021, for example, we selected five ‘Fuyu’ trees adjacent to at least one tree onto which ‘Zenjimaru’, a pollinizer cultivar, had been top-grafted, and set up six replications of each treatment on a single tree (n = 30 for each treatment). Different trees were selected for assessing fruit set each year. Treatment dates also varied among years owing to the variation in peak blooming dates (when 80% of flowers had bloomed) of ‘Fuyu’ in this field: 21 May 2018, 23 May 2019, 27 May 2020, and 22 May 2021. In 2021, for example, the NP female flower buds were covered with paper bags on 18 May, and visitor insects were monitored on 18 and 19 May. Fruit set in each pollination treatment was assessed on 29 July, when physiological fruit drop in the early development stage had finished. The numbers of female flowers used for the NP and OP treatments were 25 and 25, respectively, in 2018, 50 and 19 in 2019, 30 and 30 in 2020, and 30 and 29 (one tug was lost) in 2021. The fruit-set probability was compared between treatments and among years.
Evaluating pollination efficiency by a single visit
To evaluate pollination efficiency of major visitor species, we performed five sets of field experiments to limit the number of visits at Sites A–C from 2019 to 2021. The number of pollen grains on a stigma and the fruit-set probability were compared among the following five pollination treatments: (1) NP; (2) single visitation by A. mellifera (SV-Am); (3) single visitation by B. ardens ardens (SV-Baa); (4) OP; and (5) hand-pollination (HP). At Site C in 2021, for example, on 21 May we covered flowers with paper bags to prevent insect pollination; after confirming that the bagged buds had bloomed, we removed the bags and used these flowers for the SV-Am and SV-Baa treatments on 23, 24, and 26 May. We re-bagged the flowers immediately after we confirmed the visitation to prevent additional pollination. For the HP treatment, we used pollen collected from the male flower buds of ‘Zenjimaru’. On 24 May, male flower buds from which the sepals and petals had been removed were dried at 25 °C overnight. On the following day, we dipped a fingertip in the dried pollen in a Petri dish and attached the pollen to the stigma of a female flower. After hand-pollination, the flowers were left open. The procedures in the NP and OP treatments were as described in the previous subsection. In this set of pollination experiments, we used 30 flowers in the NP treatment, 26 in SV-Am, 40 in SV-Baa, 30 in OP, and 30 in HP. The other four sets of a sequence of pollination experiments (at Site A in 2019 and 2021, at Site B in 2021, and at Site C in 2020) were conducted as described above, but the number of treatments (3–5) depended on the set. At Site A, we omitted SV-Am owing to the extremely rare flower visitation by A. mellifera. At Site B, we omitted SV-Am, and also omitted HP owing to the limited number of female flowers available for our experiment.
The stigmas of the female flowers used for the pollination treatments were collected to count the number of pollen grains deposited; they were collected 24 h after pollination or later in the SV-Am, SV-Baa, and HP treatments, or 3 days after anthesis in the NP and OP treatments. This procedure is harmless to ovary growth, because it has been confirmed that removal of stigmas 10 h after hand-pollination does not reduce persimmon fruit set and seed formation (Kawagoe and Inoue 1958). The collected stigmas were taken to our laboratory and stored individually in tubes at − 30 °C in the freezer until measurement. Under a microscope (Eclipse E200; Nikon, Tokyo, Japan), we counted the numbers of pollen grains of D. kaki deposited on each stigma without dyeing by continuously shifting the focal distance. We compared the number of pollen grains deposited by a single visit between A. mellifera and B. ardens ardens using the data obtained at Site C in 2020 and 2021.
We then compared the fruit-set probabilities between pollination treatments using the data from the pollination experiment conducted at Site B in 2021, where the NP treatment had the lowest fruit-set probability among our five datasets. The effect of a single visit by a pollinator species on the fruit-set probability was analyzed as the difference between the NP and SV-Baa treatments.
Relationship between the number of pollen grains on a stigma and yields
To test the relationship between the number of pollen grains deposited on a stigma and the resulting fruit set and seed numbers, we used fruits that had developed from the flowers from which the stigmas were collected for counting pollen grains. Fruit set was assessed as in the previous subsection. The number of seeds was counted in each immature fruit harvested in late July every year. To analyze the relationship between the number of pollen grains on a stigma and fruit sets or the number of seeds, we used the data set of the pollination experiment conducted at Site B in 2021, where the NP treatment had the lowest fruit-set probability among our five datasets. Forty-five female flowers were used for this experiment.
Statistical analysis
To compare the fruit-set probability between the OP and NP treatments, we used a generalized linear model (GLM) in which the response variable was the fruit-set success, assumed to follow a binomial distribution (with a logit link function), and the explanatory variables were treatment and year. In addition, we estimated the fruit-set probability distribution using a Bayesian model in which we assumed that the fruit-set probability followed a Bernoulli probability distribution. To compare the number of pollen grains deposited on a stigma by a single visit between A. mellifera and B. ardens ardens at Site C in 2020 and 2021, we used a GLM in which the response variable was the number of pollen grains on a stigma and the explanatory variables were year, visitor species, and their interaction. In this model, the response variable was assumed to follow a quasi-Poisson distribution because the residual deviance was far greater than the degree of freedom when a Poisson distribution was assumed in a preliminary analysis. To compare the fruit-set probability between the OP, NP, and SV-Baa treatments, we used a Bayesian model similar to that above. Then we used a generalized linear mixed model (GLMM) to examine the relationship between the number of pollen grains on a stigma and the fruit-set success. In the model, the response variable was the fruit-set success, assumed to follow a binomial distribution (with a logit link function), and the explanatory variable was the number of pollen grains deposited on a stigma and a random effect was the trees. In the GLMM assessing the relationship between the number of pollen grains on a stigma and the number of seeds in a fruit, the response variable was the number of seeds, of which the maximum is eight, assumed to follow a binomial distribution, and the explanatory variable was the number of pollen grains on a stigma and a random effect was the trees. Data from both developed and dropped fruits (all assumed to have no seed) were used to assess the relationship between the number of pollen grains on a stigma and the “potential” number of seeds. All statistical analyses were performed in R v. 4.1.1 software (R Development Core Team 2021). For Bayesian inferences, we used Stan v. 2.21.2 software (Stan Development Team 2020). To fit a GLMM, we used “lme4” package (Bates et al. 2015).
Results
Flower-visiting species
Morphological identification indicated that the visitors captured in the 10 orchards consisted of at least 18 species in female flowers and 22 species in male flowers. The 453 captured individuals belonged to the Coleoptera (56 individuals), Diptera (4), Hymenoptera (392), and Lepidoptera (1) (Table S3). Of the Hymenoptera, B. ardens ardens (183) and A. mellifera (130) were the predominant species (69.1% of all individuals), along with other taxa such as Xylocopa appendiculata circumvolans (18), Andrena spp. (Andrenidae; 8 individuals of 3 species), and Lasioglossum spp. (Halictidae; 31 individuals of 7 species). Bombus ardens ardens was the only bumblebee species captured in this survey. Species belonging to other orders, such as the Coleoptera, Diptera, and Lepidoptera, were relatively minor.
Frequency of flower visitations
Apis mellifera and B. ardens ardens were the predominant visitors to the female flowers (Fig. 1), consistent with the results of the insect capture (Table S3). The visitation frequency of A. mellifera far exceeded those of the other species in five orchards (Fukushima, Gifu, Kumamoto, Shimane, and Shizuoka), whereas B. ardens ardens was the most frequent visitor in four (Hiroshima, Ibaraki, Kagawa, and Wakayama). In the orchard in Fukuoka, both species were major visitors to female flowers.
Frequency of visitations to the female flowers of Diospyros kaki in Japan. Flower-visiting individuals were classified into five species (Apis cerana japonica, A. mellifera, Bombus ardens ardens, B. diversus diversus, and Xylocopa appendiculata circumvolans) or four functional groups (medium-sized bees, small bees, dipterans including syrphid flies, and beetles). The numbers of visitations by insects (N) and to the female flowers in total used for the monitoring are noted for each research site. Consecutive visitations to a flower by a single individual were counted independently. The prefectures in gray ranked in the upper half of persimmon-producing prefectures in 2020
Although A. mellifera was most abundant in the five orchards, no hives of European honeybee were introduced into these orchards. Within a 1-km radius around each site in Gifu and Kumamoto, we found seven orchards with managed hives around the site in Gifu but no such orchard around that in Kumamoto. In contrast, European honeybee hives were introduced into the orchard in Ibaraki, but the predominant visitor to the flowers in this orchard was B. ardens ardens. For the frequency of the visitors to the male flowers, see Fig. S1. Several functional groups, such as medium-sized bees, small bees, and beetles, were found more frequently on the male flowers than on the female ones, but A. mellifera and B. ardens ardens were also the major visitors to the male flowers.
Frequency of flower visitations and fruit set in consecutive years
Predominant visitations by B. ardens ardens to female flowers of D. kaki were confirmed in 4 consecutive years in Hiroshima, although the visitation frequency fluctuated between years, from 1.20 visitations/h/flower in 2021 to 5.84 in 2018 (Table 1). Other minor visitors, such as A. mellifera, X. appendiculata circumvolans, medium-sized bees, and small bees, occasionally visited the flowers, but they remained marginal compared with B. ardens ardens.
The estimated fruit-set probability distribution differed between the OP and NP treatments in 2018, 2019, and 2020, but not in 2021 (Fig. 2). The mean fruit-set probabilities were 0.888 (Bayesian 95% confidence interval, 0.745–0.975) in OP and 0.111 (0.0236–0.258) in NP in 2018, 0.952 (0.826–0.999) and 0.440 (0.319–0.573) in 2019, 0.843 (0.696–0.946) and 0.0312 (0.00105–0.116) in 2020, and 0.749 (0.591–0.880) and 0.719 (0.549–0.861) in 2021. The Bayesian 95% confidence intervals of the OP treatment overlapped among the 4 years, whereas those of the NP treatment did not; and the intervals of the NP treatment in 2018 and 2020 did not overlap with those in 2019 and 2021. Thus, fruit set was more variable in NP (i.e., parthenocarpic fruiting) than in OP among years.
Pollination efficiency by a single visit
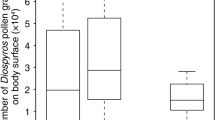
The average number of pollen grains deposited on a stigma by a single visit of A. mellifera was 20.7 ± 14.8 (mean ± SD) and that of B. ardens ardens was 24.4 ± 21.7 (Fig. 3). The average of these two bee species was 9.4 ± 11.8 in 2020 and 28.9 ± 18.9 in 2021. GLM detected a significant effect of research year (F = 25.24, p < 0.0001) but not of visitor species (F = 1.15, p = 0.29) or the interaction effect (F = 0.02, p = 0.89).
The number of pollen grains on a stigma deposited by a single visit of Apis mellifera (gray circle and box) and Bombus ardens ardens (white circle and box). The top, middle, and bottom lines of each box indicate the third, second (median), and first quartiles, respectively. The top and bottom end of whiskers indicate the third quartile + 1.5 interquartile range (IQR) and the first − 1.5 IQR, respectively. Statistical summary of GLM is written on the upper left (N.S.: not significant, **: p < 0.01)
The mean estimated fruit-set probability by a single visit of B. ardens ardens (0.483, 95% CI = 0.313–0.662) was higher than that of NP (0.195, 95% CI = 0.081–0.350) (p = 0.991) and lower than that of OP (0.844, 95% CI = 0.699–0.945) (p = 0.999) (Fig. 4).
Relationship between number of pollen grains deposited on a stigma and yield
Fruit set increased as the number of pollen grains on a stigma increased (z = 3.903, p < 0.0001; Fig. 5a). The estimated values of the intercept and coefficient in the estimated logistic curve were 0.0353 and − 0.957, respectively, such that the number of pollen grains at the inflection point of the curve was 27.1. To achieve a successful fruit set of > 80%, the model suggested that > 66.5 pollen grains are required.
a Fruit-set success: open circles represent the numbers of pollen grains deposited on a stigma, and the solid line is the fitted logistic curve. b The number of mature seeds developed in a fruit when the given number of pollen grains was deposited on the stigma. Open and gray circles indicate developed fruits and dropped fruits at the early development stage, respectively. The number of seeds in dropped fruits is assumed to be 0 in this analysis. Solid line shows a fitted curve estimated by GLMM with a binomial distribution. For details, see text
The GLMM detected a significant effect of the number of pollen grains on the number of mature seeds in a fruit (z = 9.865, p < 0.0001; Fig. 5b). The fitted GLMM predicted that when 27.1 and 66.5 pollen grains were deposited on a stigma, the seeding success rate per seed was 0.115 and 0.235, respectively. Then when 27.1 pollen grains are given, a fruit will produce one or more seeds with a 62.3% probability. When 66.5 pollen grains are given, a fruit will do so with a 88.3% probability. The number of pollen grains attached on a stigma, fruit set, and number of seeds in the seven sets of pollination experiments are summarized in Table 2.
Discussion
Flower visitors and their pollination efficiencies
Both B. ardens ardens and A. mellifera were the major flower visitors of Oriental persimmon over a wide area in Japan (Fig. 1). Both field monitoring and capture indicated that either of these species was the most dominant visitor at all monitoring sites, accounting for more than 90% of total observed individuals, though which species was the most dominant was site dependent. These species were also found on the flowers of both sexes, suggesting that they potentially carried pollen to the female flowers. Frequent visits by several functional groups to the male flowers could be due to the difference in the shape of flowers and the presence/absence of pollen. Species composition of flower visitors possibly varies between cultivars of D. kaki, but B. ardens ardens and A. mellifera predominated over the others as far as we investigated.
Pollination efficiencies of the two major flower visitors, B. ardens ardens and A. mellifera, did not differ significantly in each year (Fig. 3). This study is the first to exactly evaluate pollination efficiency by comparing the number of pollen grains deposited on a stigma per visit between major flower visitors to D. kaki. Our measurements indicate that the pollination efficiency of honeybees on D. kaki was considerably higher than previous studies suggested (Fukae et al. 1987; Nakamura et al. 2020; Yokozawa 1952; Yokozawa et al. 1968). The lower estimation by those studies reflects the smaller number of pollen grains attached to the body surface of honeybees. Our results highlight the importance of directly surveying the number of pollen grains deposited on a stigma during a single visit. Therefore, it seems that the European honeybee may serve as a pollinator as well as the bumblebee, although its pollination effectiveness could be more influenced by environmental factors, such as the presence of groundcover flowers (Nikkeshi et al. 2021).
The number of pollen grains given by a single visit of a bee differed between years, although the reason was unclear (Fig. 3). The difference might be due to fluctuating density of bees or the amount of pollen resources available in the orchard. In addition to Site C, we performed similar experiments at Sites A and B and found that the average number of pollen grains deposited by a single visit of these bee species fell in the range of 10 to 35. These results indicate that even a single visit induces seed formation, as described below, and increases fruit set in the year in which physiological fruit drop is large. But considering that the fruit-set probability in the SV-Baa treatment was smaller than that in OP, multiple visits by bees should be necessary to ensure sufficient fruit set (Fig. 4).
Other wild bees were also observed as the flower visitors of Oriental persimmon. Table 3 summarizes the flower visitors to D. kaki that have been recorded in more than two locations in previous studies and here. Among the relatively minor species, the Japanese honeybee (A. cerana japonica) and carpenter bee (X. appendiculata circumvolans) were more frequently observed than the other 15 species in our flower-visitor survey. They were also recorded as major flower visitors to D. kaki in past studies (Miura 1982; Yokozawa 1951). The pollination efficiency of these 17 species has not been evaluated, but we observed that a single visit by A. cerana japonica and by a small bee resulted in fruit set (data not shown), suggesting that such wild bees in general can pollinate D. kaki. Thus, A. cerana japonica and X. appendiculata circumvolans would be second to B. ardens ardens as natural pollinators of Oriental persimmon in Japan.
It appears that the role of coleopterans and dipterans, both of which were occasional visitors, in pollinating this crop is marginal, for a couple of reasons. First, our field observation revealed that coleopterans tend to remain on the same flower for a long time (data not shown), which indicates that cross-visitations between male and female flowers are rare. Second, one of the most common syrphid species found on D. kaki flowers, Eristalis cerealis Fabricius (Diptera: Syrphidae), was reported to be extremely poor at transporting the pollen (Ueda et al. 1976).
Fruit set and seed formation
A fitted logistic curve showed a positive relationship between the number of pollen grains deposited on a stigma and subsequent fruit set in our pollination experiment (Fig. 5a). The threshold number of pollen grains for fruit set was estimated to be 27, comparable to the average number of pollen grains deposited by a single visit of the major insect visitors. To achieve > 80% fruit set, however, more than about 70 pollen grains were needed. These results indicate that several visits by the bees would be necessary for assured fruit set. Because a female flower remains highly fertile for approximately 3 days after anthesis (Mori and Hamaguchi 1950; Yokozawa 1951), it could receive enough pollen grains for assured fruit set via a few visits by pollinators per day.
We found a loose but significantly positive relationship between the number of pollen grains deposited on a stigma and the resulting seed number (Fig. 5b). According to our model prediction, a fruit given 66.5 pollen grains, which ensures fruit set (> 80%), develops one or more seeds with a 88.3% probability. These predictions reasonably agree with a report that two to three seeds are sufficient to inhibit physiological fruit drop (Yakushiji and Hase 1991).
Our analysis demonstrated that the ratio of fruit set without pollination varied between years, from 0.031 to 0.719 (Fig. 2), which agrees with the report that the fruiting ratio ranged from 0 to 59% in the negative control using ‘Fuyu’ (Yamada et al. 1987). In contrast, fruit set in the OP treatment remained high, between 0.749 and 0.952, suggesting that pollination of ‘Fuyu’ ensures a stable crop yield and that sufficient pollination was provided throughout the 4 years despite the annual fluctuation of visitation frequency (Table 1).
Introduction of honeybee hives
The European honeybee, A. mellifera, is one of the major pollinators of this crop. Because it is not established in the natural ecosystem in Japan, it can be observed in an orchard if hives are placed in the surrounding area. Its arrival on D. kaki flowers, however, has been somewhat uncertain, because the introduction of hives into orchards does not necessarily increase its visitation frequency to the focal crop (Fukae et al. 1987; Nakamura et al. 2020). This uncertainty was also seen here. The most dominant visitor species was B. ardens ardens in the orchard where hives of A. mellifera were placed (i.e., Ibaraki), while A. mellifera was dominant in orchards where no hives were placed (Fig. 1). In the latter case, workers might have come from neighboring orchards. We confirmed in Gifu that hives were introduced in the surrounding orchards, but we could find no hives around the orchard in Kumamoto. The uncertainty may be caused by the species’ long-range foraging, which occasionally exceeds 5 km (Beekman and Ratnieks 2000), and its preference for herbaceous flowers to tree ones (Hagler et al. 2011; Lau et al. 2019; Nakamura et al. 2020; Nikkeshi et al. 2021). Introduction of A. mellifera hives into an orchard of Oriental persimmon might be unnecessary if flower visits by wild bees are sufficient every year. As described above, several visits by the major bees per female flower are enough to ensure fruit set, so the introduced hives might contribute less to fruit production than expected, or European honeybee workers might hinder native wild bee populations by competing for floral resources (e.g., Huryn 1997). In addition, as honeybee workers often fly for a long distance to forage, neighboring farmers should cooperate in deciding how many hives should be placed in the whole persimmon production area to maximize the effectiveness of introduced hives on their orchards.
Wild pollinators of Oriental persimmon
Our data suggest that B. ardens ardens is a predominant wild pollinator of Oriental persimmon over a wide area in Japan. It was previously recorded as a flower visitor to D. kaki in western Honshu: in Hyogo (Miyamoto 1961), Shimane (Miura 1982; Nakamura et al. 2020), and Hiroshima (Nikkeshi et al. 2019). Here, it was also recorded in Kyushu, Shikoku, and the Kanto and southern Tohoku districts, implying that it functions as an effective pollinator of D. kaki almost everywhere in Japan, provided that nesting habitats are available nearby. This is reasonable, because B. ardens ardens is distributed throughout Japan, except on Hokkaido, Tsushima Island, and the Okinawa Islands (Suzuki-Ohno et al. 2017; Tadauchi and Murao 2014). In addition, the period of its seasonal appearance, from early April until early July (Katayama 1996), fully covers the flowering period of D. kaki, which spans from early May in southwestern Japan until early June in northeastern Japan. Furthermore, B. ardens ardens relies on tree flowers rather than herbaceous ones (Osada et al. 2003; Ushimaru et al. 2008). Therefore, it is conceivable that this bumblebee has long served as the principal pollinator of Oriental persimmon since the introduction of this crop into Japan more than a thousand years ago (Yamada 2000).
Which bee species plays the principal role in pollination of Oriental persimmon in other countries, especially in East Asia? Oriental persimmon originated in southern China, and it was introduced into Korea and Japan in ancient times (Sobajima 2000). Because B. ardens ardens is also native and is one of the common bumblebee species in Korea (Byun et al. 2009; Lim and Lee 2012), it is possible that this bee species is the major pollinator of D. kaki in Korea as in Japan. However, B. ardens is not found in China (Huang and An 2018). Thus, other Bombus species or other bees would pollinate Oriental persimmon in China, probably together with honeybees, but there is no information available on this matter. Identifying the major pollinators in areas where D. kaki is native and non-native will improve our understanding of the natural pollination system and environmentally sustainable management of this crop.
References
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Beekman M, Ratnieks FLW (2000) Long-range foraging by the honey-bee, Apis mellifera L. Funct Ecol 14:490–496. https://doi.org/10.1046/j.1365-2435.2000.00443.x
Byun BK, Lee BW, Park SY, Lee YM, Jo DG (2009) Insect fauna of Mt. Nam-san in Seoul, Korea. J Korean Nat 2:137–153. https://doi.org/10.1016/S1976-8648(14)60050-5
Eeraerts M, Meeus I, Van Den Berge S, Smagghe G (2017) Landscapes with high intensive fruit cultivation reduce wild pollinator services to sweet cherry. Agric Ecosyst Environ 239:342–348. https://doi.org/10.1016/j.agee.2017.01.031
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299. https://www.mbari.org/wp-content/uploads/2016/01/Folmer_94MMBB.pdf. Accessed 27 Jan 2022
Françoso E, Arias MC (2013) Cytochrome c oxidase I primers for corbiculate bees: DNA barcode and mini-barcode. Mol Ecol Resour 13:844–850. https://doi.org/10.1111/1755-0998.12135
Fukae Y, Hamachi F, Tsujikawa Y (1987) A detailed analysis of the effect of honeybee introduction on the pollination of persimmons. Honeybee Sci 8:167–171 (in Japanese with English abstract)
Garibaldi LA, Steffan-Dewenter I, Winfree R, Aizen MA, Bommarco R et al (2013) Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 339:1608–1611. https://doi.org/10.1126/science.1230200
Garratt MPD, Breeze TD, Boreux V, Fountain MT, McKerchar M et al (2016) Apple pollination: demand depends on variety and supply depends on pollinator identity. PLoS ONE 11:e0153889. https://doi.org/10.1371/journal.pone.0153889
Hagler JR, Mueller S, Teuber LR, Machtley SA, Van Deynze A (2011) Foraging range of honey bees, Apis mellifera, in alfalfa seed production fields. J Insect Sci 11:144. https://doi.org/10.1673/031.011.14401
Huang J, An J (2018) Species diversity, pollination application and strategy for conservation of the bumblebees of China. Biodiv Sci 26:486–497. https://doi.org/10.17520/biods.2018068 (in Chinese with English abstract)
Huryn VMB (1997) Ecological impacts of introduced honey bees. Q Rev Biol 72:275–297. https://doi.org/10.1086/419860
Kajiura M (1941) Studies on physiological dropping of fruits in the Japanese persimmon II. Relationships among dropping of fruits, pollination and parthenocarpy. J Jpn Soc Hortic Sci 12:247–283. https://doi.org/10.2503/jjshs.12.247 (In Japanese)
Katayama E (1996) Survivorship curves and longevity for workers of Bombus ardens Smith and Bombus diversus Smith (Hymenoptera, Apidae). Jpn J Ent 64:111–121
Kawagoe H, Inoue J (1958) Studies on the pollination in Japanese persimmon II. On the time required for fertilization and increase efficiency in artificial pollination. Special Bull Okayama Pref Agric Exp Stn 56:153–162 (in Japanese)
King C, Ballantyne G, Willmer PG (2013) Why flower visitation is a poor proxy for pollination: measuring single-visit pollen deposition, with implications for pollination networks and conservation. Methods Ecol Evol 4:811–818. https://doi.org/10.1111/2041-210X.12074
Kitajima A, Ohshita Y, Nakano M, Ishida M (1993) Comparisons of fruit set and quality between seeded and parthenocarpic kaki fruits cv. Fuyu in relation to the location of the fruiting shoot. J Jpn Soc Hortic Sci 62:505–512. https://doi.org/10.2503/jjshs.62.505 (in Japanese with English abstract)
Lau P, Bryant V, Ellis JD, Huang ZY, Sullivan J et al (2019) Seasonal variation of pollen collected by honey bees (Apis mellifera) in developed areas across four regions in the United States. PLoS ONE 14:e0217294. https://doi.org/10.1371/journal.pone.0217294
Lim HM, Lee DH (2012) Insect fauna of Is. Ulleung-do (Prov. Gyeongsangbuk-do) in Korea. J Korean Nat 5:243–250. https://doi.org/10.7229/jkn.2012.5.3.243
Miura T (1982) On the hourly change of the pollinator association found in the Japanese persimmon, var. Saijo orchard in day-time. Bull Fac Agric Shimane Univ 16:166–171 (In Japanese with English abstract)
Miyamoto S (1961) Flower-visiting habits of bumblebees (Biological studies on Japanese bees XIX). Jpn J Appl Entomol Zool 5:28–39. https://doi.org/10.1303/jjaez.5.28 (in Japanese with English abstract)
Mori H, Hamaguchi K (1950) Some experiments on the commercial hand pollination of Kaki (Japanese persimmon). J Jpn Soc Hortic Sci 19:98–106. https://doi.org/10.2503/jjshs.19.98 (in Japanese with English abstract)
Moriguchi K, Matsushita T, Matsumoto H, Ohmasa Y, Sakimoto T (2006) Cultivation of the Japanese persimmons ‘Shinsyuu’ and ‘Taishuu’ under heated greenhouse conditions. Bull Ehime Fruit Tree Exp Sta Jpn 20:7–17 (in Japanese with English abstract)
Nakamura S, Yamamoto S, Sawamura N, Nikkeshi A, Kishi S, Kamo T (2020) Pollination effectiveness of European honeybee, Apis mellifera (Hymenoptera: Apidae), in an Oriental persimmon, Diospyros kaki (Ericales: Ebenaceae), orchard. Appl Entomol Zool 55:405–412. https://doi.org/10.1007/s13355-020-00696-5
Ne’eman G, Jürgens A, Newstrom-Lloyd L, Potts SG, Dafni A (2010) A framework for comparing pollinator performance: effectiveness and efficiency. Biol Rev 85:435–451. https://doi.org/10.1111/j.1469-185X.2009.00108.x
Nikkeshi A, Inoue H, Arai T, Kishi S, Kamo T (2019) The bumblebee Bombus ardens ardens (Hymenoptera: Apidae) is the most important pollinator of Oriental persimmon, Diospyros kaki (Ericales: Ebenaceae), in Hiroshima, Japan. Appl Entomol Zool 54:409–419. https://doi.org/10.1007/s13355-019-00637-x
Nikkeshi A, Inoue H, Arai T, Kishi S, Kamo T (2021) The bumblebee Bombus ardens ardens (Hymenoptera: Apidae) visits white clover in orchards before Oriental persimmon blooms. Entomol Sci 24:12–17. https://doi.org/10.1111/ens.12449
Osada N, Sugiura S, Kawamura K, Cho M, Takeda H (2003) Community-level flowering phenology and fruit set: comparative study of 25 woody species in a secondary forest in Japan. Ecol Res 18:711–723. https://doi.org/10.1111/j.1440-1703.2003.00590.x
R Development Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org
Rollin O, Garibaldi LA (2019) Impacts of honeybee density on crop yield: a meta-analysis. J Appl Ecol 56:1152–1163. https://doi.org/10.1111/1365-2664.13355
Simon C, Frati F, Beckenbach A, Crespi B, Liu H, Flook P (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701. https://doi.org/10.1093/aesa/87.6.651
Sobajima Y (2000) In: Rural Culture Association Japan (ed.) Kaju Engei Dai Hyakka 6, Kaki (Encyclopedia of Fruit Trees 6, Oriental persimmon), Rural Culture Association Japan, Tokyo, pp 5–6
Stan Development Team (2020) RStan: the R interface to Stan. R Package Version 2(21):2
Statistics Bureau, Ministry of Internal Affairs and Communications, Japan (2021) The e-Stat (a portal site for Japanese Government Statistics) of the Statistics Bureau, Ministry of Internal Affairs and Communications. Tables ‘p001-30-015’ and ‘syukaku_senasi_20’ (In Japanese). https://www.e-stat.go.jp/. Accessed 5 Oct 2021
Sugden EA, Pyke GH (1991) Effects of honey bees on colonies of Exoneura asimillima, an Australian native bee. Aust J Ecol 16:171–181. https://doi.org/10.1111/j.1442-9993.1991.tb01044.x
Suzuki T, Niikawa T (2018) Effects of artificial pollination and grafting of pollen cultivar on physiological fruit drop in ‘Soshu’ persimmon. Hort Res (japan) 17:19–26. https://doi.org/10.2503/hrj.17.19 (in Japanese with English abstract)
Suzuki-Ohno Y, Yokoyama J, Nakashizuka T, Kawata M (2017) Utilization of photographs taken by citizens for estimating bumblebee distributions. Sci Rep 7:11215. https://doi.org/10.1038/s41598-017-10581-x
Tadauchi O, Murao R (2014) An illustrated guide to Japanese bees. Bun-ichi Shuppan, Tokyo (In Japanese)
Ueda H, Notsu Y, Miura T (1976) Use of insect pollinators in kaki ‘Saijo’ orchard. Pollinating Ability of Eristalis cerealis Fabricious. Bull Fac Agric Shimane Univ 10:39–42 (In Japanese with English abstract)
Ushimaru A, Ishida C, Sakai S, Shibata M, Tanaka H, Niiyama K, Nakashizuka T (2008) The effects of human management on spatial distribution of two bumble bee species in a traditional agro-forestry Satoyama landscape. J Apic Res 47:296–303. https://doi.org/10.1080/00218839.2008.11101478
Valido A, Rodríguez-Rodríguez MC, Jordano P (2019) Honeybees disrupt the structure and functionality of plant-pollinator networks. Sci Rep 9:4711. https://doi.org/10.1038/s41598-019-41271-5
Woodburn KR, Andersen PC (1996) Pollination and pollen source influence fruit of Oriental persimmon ‘Fuyu’ and ‘Tanenashi.’ Hort Sci 31:218–221. https://doi.org/10.21273/HORTSCI.31.2.218
Yakushiji H, Hase Y (1991) Influence of number of seeds and tree shading on June drop and phytohormone content of Japanese persimmon ‘Fuyu’ fruit. Bull Fruit Tree Res Stn 19:49–59 (in Japanese with English abstract)
Yamada M, Kurihara A, Sumi T (1987) Varietal differences in fruit bearing in Japanese persimmon (Diospyros kaki Thunb.) and their yearly fluctuations. J Jpn Soc Hortic Sci 56:293–299. https://doi.org/10.2503/jjshs.56.293 (In Japanese with English abstract)
Yamada M (2000) In: Rural Culture Association Japan (ed.) Kaju Engei Dai Hyakka 6, Kaki (Encyclopedia of Fruit Trees 6, Oriental persimmon), Rural Culture Association Japan, Tokyo, pp 109–114
Yamamura H (1982) Effects of pollination and fertilization on fruit set in “Saijo” Japanese persimmon. Bull Fac Agric Shimane Univ 16:8–13 (In Japanese with English abstract)
Yamamura H, Osaki Y (1982) Histological studies on the ovule development and fertilization ability in “Saijo” Japanese persimmon. Bull Fac Agric Shimane Univ 16:14–19 (In Japanese with English abstract)
Yokozawa Y (1951) Insect visitors on the flowers of Japanese persimmon. J Jpn Soc Hortic Sci 20:58–64. https://doi.org/10.2503/jjshs.20.58 (In Japanese)
Yokozawa Y (1952) Insect visitors on the flowers of Japanese persimmon (The second report). J Jpn Soc Hortic Sci 21:25–28. https://doi.org/10.2503/jjshs.21.25 (In Japanese)
Yokozawa Y, Yasui A, Morimoto M (1968) Studies on the transport of pollen of Japanese persimmons and peaches by insects. 1. The efficiency of honeybee colonies. Bull Nara Agric Exp Sta 2:1–9 (in Japanese with English abstract)
Acknowledgements
We appreciate the kind cooperation of Ms. Natsuko Kado (Shimane Agricultural Technology Center), Dr. Hisatomo Taki (Forest Research and Management Organization), Dr. Soichi Kugimiya, Dr. Masayoshi K. Hiraiwa, and Mr. Tomoya Katsuno (NARO) in the field experiments. We are grateful to the Fukuoka Agricultural and Forestry Research Center, Gifu Prefectural Agricultural Technology Center, Kagawa Prefectural Agricultural Experiment Station, Kumamoto Prefectural Agricultural Research Center, and Wakayama Fruit Tree Experiment Station for our use of their experimental fields. We thank the persimmon growers in Fukushima, Ibaraki, Shimane, and Shizuoka prefectures for their kind cooperation. The technical assistance of Ms. Satomi Kohara and Mr. Ryohei Hoshino is also greatly acknowledged. This work was supported financially by the Ministry of Agriculture, Forestry and Fisheries, Japan, through a research project entitled “Monitoring and enhancement of pollinators for crop production (JPJ006239).”
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kamo, T., Nikkeshi, A., Inoue, H. et al. Pollinators of Oriental persimmon in Japan. Appl Entomol Zool 57, 237–248 (2022). https://doi.org/10.1007/s13355-022-00784-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-022-00784-8