Abstract
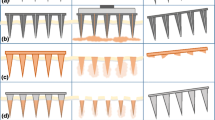
Over the years, scientists have been focused on the development of microneedle coating process to coat a broad range of therapeutic agents onto the surface of the solid microneedles for effective drug delivery. The precise dose control, content uniformity as well as large-scale production of coated microneedles are still the core issues that have been the interest of researchers in this topic. To this end, a repeatable method that involved a micro-molding process was demonstrated for mass fabrication of coated microneedles with homogeneous and controllable drug loading under mild conditions. In this system, the dissolvable drug carriers with precise dosage were first mounted onto the solid microneedles and then exposed to the high moisture condition to finally obtain the coated microneedle with uniform and precise drug loading. Using the microneedle molds with the volume of 4.71 nL, 8.24 nL, 10.47 nL, and 12.56 nL per cavity, the drug loadings were precisely controlled at 4.8 ng, 6.4 ng, 9.3 ng, and 13.5 ng per needle, with the standard deviation of 0.09, 0.01, 0.07, and 0.53%, respectively. Mechanical property tests showed that the coated microneedles are strong enough for reliable skin insertion, and with in vivo trials in diabetic mice, we further confirmed the similar hypoglycaemic effect of insulin-coated microneedles to subcutaneous injection. Taken together, the micro-molding-based fabrication process has practical merits in the mass production of coated microneedles with homogeneous and controllable drug loading, facilitating the clinical translation of the microneedle technique.
Graphical abstract









Similar content being viewed by others
Data availability
The data are available from the corresponding author on reasonable request.
References
Ingrole R, Gill H. Microneedle coating methods: a review with a perspective. J Pharmacol Exp Ther. 2019;370:555–69.
Gill HS, Prausnitz MR. Coated microneedles for transdermal delivery. J Control Release. 2007;117(2):227–37.
Chen BZ, Ashfaq M, Zhu DD, Zhang XP, Guo XD. Controlled delivery of insulin using rapidly separating microneedles fabricated from genipin-crosslinked gelatin. Macromol Rapid Commun. 2018;39(20):1800075.
Kim YC, Park JH, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64(14):1547–68.
Chen BZ, Zhang LQ, Xia YY, Zhang XP, Guo XD. A basal-bolus insulin regimen integrated microneedle patch for intraday postprandial glucose control. Sci Adv. 2020;6(28):eaba7260.
Jin X, Zhu DD, Chen BZ, Ashfaq M, Guo XD. Insulin delivery systems combined with microneedle technology. Adv Drug Deliv Rev. 2018;127:119–37.
Ito Y, Hirono M, Fukushima K, Sugioka N, Takada K. Two-layered dissolving microneedles formulated with intermediate-acting insulin. Int J Pharm. 2012;436(1–2):387–93.
Feng YH, Zhang XP, Zhao ZQ, Guo XD. Dissipative particle dynamics aided design of drug delivery systems: a review. Mol Pharm. 2020;17(6):1778–99.
Prausnitz MR. Microneedles for transdermal drug delivery. Adv Drug Deliv Rev. 2004;56(5):581–7.
Larraneta E, Lutton REM, Woolfson AD, Donnelly RF. Microneedle arrays as transdermal and intradermal drug delivery systems: materials science manufacture and commercial development. Mater Sci Eng R. 2016;104:1–32.
Lee JW, Prausnitz MR. Drug delivery using microneedle patches: not just for skin. Expert Opin Drug Deliv. 2018;15(6):541–3.
Choi HJ, Yoo DG, Bondy BJ, Quan FS, Compans RW, Kang SM, Prausnitz MR. Stability of influenza vaccine coated onto microneedles. Biomaterials. 2012;33(14):3756–69.
Li S, Li W, Prausnitz M. Individually coated microneedles for co-delivery of multiple compounds with different properties. Drug Deliv Transl Res. 2018;8(5):1043–52.
Vrdoljak A, McGrath MG, Carey JB, Draper SJ, Hill AV, O’Mahony C, Crean AM, Moore AC. Coated microneedle arrays for transcutaneous delivery of live virus vaccines. J Control Release. 2012;159(1):34–42.
McGrath MG, Vrdoljak A, O’Mahony C, Oliveira JC, Moore AC, Crean AM. Determination of parameters for successful spray coating of silicon microneedle arrays. Int J Pharm. 2011;415(1):140–9.
DeMuth PC, Li AV, Abbink P, Liu J, Li H, Stanley KA, Smith KM, Lavine CL, Seaman MS, Kramer JA, Miller AD, Abraham W, Suh H, Elkhader J, Hammond PT, Barouch DH, Irvine DJ. Vaccine delivery with microneedle skin patches in nonhuman primates. Nat Biotechnol. 2013;31(12):1082–5.
Khan H, Mehta P, Msallam H, Armitage D, Ahmad Z. Smart microneedle coatings for controlled delivery and biomedical analysis. J Drug Target. 2014;22(9):790–5.
Haj-Ahmad R, Khan H, Arshad MS, Rasekh M, Hussain A, Walsh S, Li X, Chang M-W, Ahmad Z. Microneedle coating techniques for transdermal drug delivery. Pharmaceutics. 2015;7(4):486–502.
Caudill CL, Perry JL, Tian S, Luft JC, DeSimone JM. Spatially controlled coating of continuous liquid interface production microneedles for transdermal protein delivery. J Control Release. 2018;284:122–32.
Uddin MJ, Scoutaris N, Klepetsanis P, Chowdhry B, Prausnitz MR, Douroumis D. Inkjet printing of transdermal microneedles for the delivery of anticancer agents. Int J Pharm. 2015;494(2):593–602.
Liang L, Chen Y, Zhang BL, Zhang XP, Liu JL, Shen CB, Cui Y, Guo XD. Optimization of dip-coating methods for the fabrication of coated microneedles for drug delivery. J Drug Deliv Sci Technol. 2020;55:101464.
Park SC, Kim MJ, Baek S-K, Park J-H, Choi S-O. Spray-formed layered polymer microneedles for controlled biphasic drug delivery. Polymers. 2019;11(2):369.
Larraneta E, McCrudden MTC, Courtenay AJ, Donnelly RF. Microneedles: a new frontier in nanomedicine delivery. Pharm Res. 2016;33(5):1055–73.
Ross S, Scoutaris N, Lamprou D, Mallinson D, Douroumis D. Inkjet printing of insulin microneedles for transdermal delivery. Drug Deliv Transl Res. 2015;5(4):451–61.
Zhang Y, Yu J, Kahkoska AR, Wang J, Buse JB, Gu Z. Advances in transdermal insulin delivery. Adv Drug Deliv Rev. 2019;139:51–70.
Chen BZ, Yang Y, Wang BB, Ashfaq M, Guo XD. Self-implanted tiny needles as alternative to traditional parenteral administrations for controlled transdermal drug delivery. Int J Pharm. 2019;556:338–48.
Saurer EM, Flessner RM, Sullivan SP, Prausnitz MR, Lynn DM. Layer-by-layer assembly of DNA- and protein-containing films on microneedles for drug delivery to the skin. Biomacromol. 2010;11(11):3136–43.
O’Mahony C, Hilliard L, Kosch T, Bocchino A, Sulas E, Kenthao A, O’Callaghan S, Clover AJP, Demarchi D, Bared G. Accuracy and feasibility of piezoelectric inkjet coating technology for applications in microneedle-based transdermal delivery. Microelectron Eng. 2017;172:19–25.
He MC, Chen BZ, Ashfaq M, Guo XD. Assessment of mechanical stability of rapidly separating microneedles for transdermal drug delivery. Drug Deliv Transl Res. 2018;8(5):1034–42.
Thakur RR, Tekko IA, Al-Shammari F, Ali AA, McCarthy H, Donnelly RF. Rapidly dissolving polymeric microneedles for minimally invasive intraocular drug delivery. Drug Deliv Transl Res. 2016;6(6):800–15.
Chen BZ, Liu JL, Li QY, Wang ZN, Zhang XP, Shen CB, Cui Y, Guo XD. Safety evaluation of solid polymer microneedles in human volunteers at different application sites. ACS Appl Bio Mater. 2019;2(12):5616–25.
Wang QL, Zhu DD, Chen Y, Guo XD. A fabrication method of microneedle molds with controlled microstructures. Mater Sci Eng C. 2016;65:135–42.
Chen BZ, Ashfaq M, Zhang XP, Zhang JN, Guo XD. In vitro and in vivo assessment of polymer microneedles for controlled transdermal drug delivery. J Drug Target. 2018;26(8):720–9.
Chu LY, Prausnitz MR. Separable arrowhead microneedles. J Control Release. 2011;149(3):242–9.
Kim M, Jung B, Park JH. Hydrogel swelling as a trigger to release biodegradable polymer microneedles in skin. Biomaterials. 2012;33(2):668–78.
Funding
This work was financially supported by the National Natural Science Foundation of China (51873015), the Joint Project of BRC-BC (Biomedical Translational Engineering Research Center of BUCT-CJFH) (XK2020-05, RZ2020-01), and the long-term subsidy mechanism from the Ministry of Finance and the Ministry of Education of PRC.
Author information
Authors and Affiliations
Contributions
B.Z.C. and M.C.H. performed experimental measurements. B.Z.C. and M.C.H. drafted the paper. B.Z.C., X.P.Z and W.M.F. contributed to the data interpretation. Y. C. and X.D.G. directed the research and provided financial support.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was approved by the institutional animal care committee of China-Japan Friendship Hospital, and all procedures performed were in accordance with the ethical standards of our institution.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Bo Zhi Chen and Meng Chan He contributed equally.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 2557 KB)
Rights and permissions
About this article
Cite this article
Chen, B.Z., He, M.C., Zhang, X.P. et al. A novel method for fabrication of coated microneedles with homogeneous and controllable drug dosage for transdermal drug delivery. Drug Deliv. and Transl. Res. 12, 2730–2739 (2022). https://doi.org/10.1007/s13346-022-01123-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-022-01123-8