Abstract
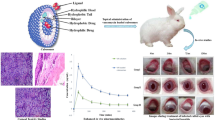
The objective of the present study was to enhance ocular antifungal activity of fluconazole (FCZ) in treatment of keratomycosis through incorporation into cubosomal nanoparticles. FCZ-loaded cubosomal dispersions were prepared by emulsification method according to 23 full factorial design. Design-Expert® software was used to study the effects of different formulation factors on properties of FCZ-loaded cubosomal dispersions and select the optimal formulation. Eight FCZ-loaded cubosomal dispersions were prepared and were in vitro and in vivo evaluated. In vitro, the results revealed that the optimum formula exhibited a mean particle size of 48.17 ± 0.65 nm and entrapped 85.70 ± 2.56% of FCZ. The ex vivo permeation study confirmed a two-fold enhancement in FCZ permeation through rabbit cornea compared to aqueous FCZ solution. Furthermore, in vivo ocular tolerance and histopathological studies proved the efficacy and safety FCZ-loaded cubosomal dispersion in treatment of induced keratomycosis in rats compared to aqueous FCZ solution after topical ocular application. In conclusion, the obtained results indicated that cubosomes could be promising ocular drug delivery system for enhancing antifungal activity of FCZ in treatment of fungal keratitis in rats.

Graphical abstract








Similar content being viewed by others
References
Iyer SA, Tuli SS, Wagoner RC. Fungal keratitis: emerging trends and treatment outcomes. Eye Contact Lens. 2006;32(6):267–71. https://doi.org/10.1097/01.icl.0000249595.27520.2e.
Gower EW, Keay LJ, Oechsler RA, Iovieno A, Alfonso EC, Jones DB, et al. Trends in fungal keratitis in the United States, 2001 to 2007. Ophthalmology. 2010;117(12):2263–7. https://doi.org/10.1016/j.ophtha.2010.03.048.
Kalkanci A, Ozdek S. Ocular fungal infections. Curr Eye Res. 2011;36(3):179–89. https://doi.org/10.3109/02713683.2010.533810.
Chien YW. Ocular drug delivery and delivery systems. In: Novel drug delivery systems. New York: Marcel Dekker; 1996. p. 269–70.
Meisner D, Mezei M. Liposome ocular delivery systems. Adv Drug Deliv Rev. 1995;16:75–93.
Zimmer A, Kreuter J. Microspheres and nanoparticles used in ocular delivery systems. Adv Drug Deliv Rev. 1995;16:61–73.
Yamaguchi M, Yasueda S, Isowaki A, Yamamoto M, Kimura M, Inada K, et al. Formulation of an ophthalmic lipid emulsion containing an anti-inflammatory steroidal drug, difluprednate. Int J Pharm. 2005;301:121–8.
Han S, Shen JQ, Gan Y, Geng HM, Zhang XX, Zhu CL, et al. Novel vehicle based on cubosomes for ophthalmic delivery of flurbiprofen with low irritancy and high bioavailability. Acta Pharmacol Sin. 2010;31:990–8. https://doi.org/10.1038/aps.2010.98.
Kaur IP, Garg A, Singla AK, Aggarwal D. Vesicular systems in ocular delivery: an overview. Int J Pharm. 2004;269:1–14. https://doi.org/10.1016/j.ijpharm.2003.09.016.
Siekmann B, Bunjes H, Koch MHJ, Westesen K. Preparation and structural investigations of colloidal dispersions prepared from cubic monoglyceride-water phases. Int J Pharm. 2002;244:33–43. https://doi.org/10.1016/s0378-5173(02)00298-3.
Gan L, Han S, Shen J, Zhu J, Zhu C, Zhang X, et al. Self-assembled liquid crystalline nanoparticles as a novel ophthalmic delivery system for dexamethasone: improving preocular retention and ocular bioavailability. Int J Pharm. 2010;396:179–87. https://doi.org/10.1016/j.ijpharm.2010.06.015.
Spicer PT, Hayden KL, Lynch ML, Ofori-Boateng A, Burnes JL. Novel process for producing cubic liquid crystalline nanoparticles (cubosomes). Langmuir. 2001;17:5748–56. https://doi.org/10.1021/la010161w.
Dian L, Yang Z, Li F, Wang Z, Pan X, Peng X, et al. Cubic phase nanoparticles for sustained release of ibuprofen: formulation, characterization, and enhanced bioavailability study. Int J Nanomedicine. 2013;8:845–54. https://doi.org/10.2147/IJN.S40547.
Li J, Wu L, Wu W, Wang B, Wang Z, Xin H, et al. A potential carrier based on liquid crystal nanoparticles for ophthalmic delivery of pilocarpine nitrate. Int J Pharm. 2013;455(1-2):75–84. https://doi.org/10.1016/j.ijpharm.2013.07.057.
Luzzate V, Husson F. The structure of the liquid crystalline phases of lipid water system J. Cell. 1962;12:209–19.
Boyd BJ. Character of drug release from cubosomes using the pressure ultrafiltration method. Int J Pharm. 2003;260:239–47.
Huang J, Peng T, Li Y, Zhan Z, Zeng Y, Huang Y, et al. Ocular cubosome drug delivery system for timolol maleate: preparation, characterization, cytotoxicity, ex vivo, and in vivo evaluation. AAPS PharmSciTech. 2017;18(8):2919–26.
Ali Z, Sharma PK, Warsi MH. Fabrication and evaluation of ketorolac loaded cubosome for ocular drug delivery. J Appl Pharm Sci. 2016;6(09):204–8.
Chen Y, Lu Y, Zhong Y, Wang Q, Wu W, Shen G. Ocular delivery of cyclosporine A based on glyceryl monooleate/poloxamer 407 liquid crystalline nanoparticles: preparation, characterization, in vitro corneal penetration and ocular irritation. J Drug Target. 2012;20(10):856–63.
Novelli V, Holzel H. Safety and tolerability of fluconazole in children. Antimicrob Agents Chemother. 1999;43:1955–60.
Magrath GN, Pulido JS, Montero J, Mason C, Wilson J. Cystoid macular edema secondary to fluconazole toxicity. Ocul Immunol Inflamm. 2010;18:472–4. https://doi.org/10.3109/09273948.2010.507320.
Price MF, LaRocco MT, Gentry LO. Fluconazole susceptibilities of Candida species and distribution of species recovered from blood cultures over a 5-year period. Antimicrob Agents Chemother. 1994;38:1422–4.
Abbasoğlu OE, Hoşal BM, Sener B, Erdemoğlu N, Gürsel E. Penetration of topical fluconazole into human aqueous humor. Exp Eye Res. 2001;72:147–51. https://doi.org/10.1006/exer.2000.0936.
Behrens-Baumann W, Klinge B, Rüchel R. Topical fluconazole for experimental candida keratitis in rabbits. Br J Ophthalmol. 1990;74:40–2 https://doi.org/10.1136/bjo.74.1.40.
Davies NM. Biopharmaceutical considerations in topical ocular drug delivery. Clin Exp Pharmacol Physiol. 2000;27:558–62. https://doi.org/10.1046/j.1440-1681.2000.03288.x.
Sasaki H, Yamamura K, Mukai T, Nishida K, Nakamura J, Nakashima M, et al. Enhancement of ocular drug penetration. Crit Rev Ther Drug Carrier Syst. 1999;16:85–146.
Habib FS, Fouad EA, Abdel-Rhaman MS, Fathalla D. Liposomes as an ocular delivery system of fluconazole: in-vitro studies. Acta Ophthalmol. 2010;88:901–4. https://doi.org/10.1111/j.1755-3768.2009.01584.x.
Gonjari ID, Hosmani AH, Karmarkar AB, Godage AS, Kadam SB, Dhabale BN. Formulation and evaluation of in situ gelling thermoreversible mucoadhesive gel of fluconazole. Drug Discov Ther. 2009;3(1):6–9.
Gratieri T, Gelfuso GM, de Freitas O, Rocha EM, Lopez RFV. Enhancing and sustaining the topical ocular delivery of fluconazole using chitosan solution and poloxamer/chitosan in situ forming gel. Eur J Pharm Biopharm. 2011;79:320–7. https://doi.org/10.1016/j.ejpb.2011.05.006.
Esposito E, Eblovi N, Rasi S, Drechsler M, Di Gregorio GM, Menegatti E, et al. Lipid-based supramolecular systems for topical application: a preformulatory study. AAPS PharmSci. 2003;5:62–76. https://doi.org/10.1016/j.jsps.2018.04.004.
Chung H, Kim J, Um JY, Kwon IC, Jeong SY. Self-assembled nanocubi as a carrier for peroral insulin delivery. Diabetologia. 2002;45:448–51. https://doi.org/10.1007/s00125-001-0751-z.
Teagarden DL, Anderson BD, Petre WJ. Determination of the pH-dependent phase distribution of prostaglandin E1 in a lipid emulsion by ultrafiltration. Pharm Res. 1988;5:482–7. https://doi.org/10.1023/a:1015961122982.
Davtyan TK, Melikyan LA, Nikoyan NA, Aleksanyan HP, Grigoryan NG. Development and validation of Simple RP-HPLC Method for intracellular determination of fluconazole concentration and its application to the study of Candida albicans azole resistance. Int J Anal Chem. 2015:1–8. https://doi.org/10.1155/2015/576250.
Saber S, Basuony M, Eldin S. Telmisartan ameliorates dextran sodium sulfate-induced colitis in rats by modulating NF-κB signalling in the context of PPARγ agonistic activity. Arch Biochem Biophys. 2019;671:185–95. https://doi.org/10.1016/j.abb.2019.07.014.
Banchroft J, Stevens A, Turner D. Theory and practice of histological techniques. 4th ed. New York: Churchill Livingstone; 1996.
Nasr M, Ghorab MK, Abdelazem A. In vitro and in vivo evaluation of cubosomes containing 5-fluorouracil for liver targeting. Acta Pharm Sin B. 2015;5(1):79–88. https://doi.org/10.1016/j.apsb.2014.12.001.
Wyatt DM, Dorschel D. A cubic-phase delivery system composed of glyceryl monooleate and water for sustained release of water-soluble drugs. Pharm Technol. 1992;16:116–30. https://doi.org/10.1155/2014/815981.
de Lima LS, Araujo MD, Quinàia SP, Migliorine DW, Garcia JR. Adsorption modeling of Cr, Cd and Cu on activated carbon of different origins by using fractional factorial design. Chem Eng J. 2011;166(3):881–9. https://doi.org/10.13168/AGG.2013.0035.
Ahmad A, Alkharfy KM, Wani TA, Raish M. Application of Box-Behnken design for ultrasonic-assisted extraction of polysaccharides from Paeonia emodi. Int J Biol Macromol. 2015;72:990–7. https://doi.org/10.1016/j.ijbiomac.2014.10.011.
Nazaruk E, Majkowska-Pilip A, Bilewicz R. Lipidic cubic-phase nanoparticles-cubosomes for efficient drug delivery to cancer cells. Chempluschem. 2017;82:570–5. https://doi.org/10.1002/cplu.201600534.
Sharma R, Kaur G, Kapoor DK. Fluconazole loaded cubosomal vesicles for topical delivery. Int J Drug Dev Res. 2015;7(3):32–41.
Gaballa SA, El Garhy OH, Abdelkader H. Cubosomes: composition, preparation, and drug delivery applications. J Adv Biomed Pharm Sci. 2020;3(1):1–9. https://doi.org/10.21608/jabps.2019.16887.1057.
Younes NF, Abdel-Halim SA, Elassasy AI. Corneal targeted sertaconazole nitrate loaded cubosomes: preparation, statistical optimization, in vitro characterization, ex vivo permeation and in vivo studies. Int J Pharm. 2018;553(1-2):386–97. https://doi.org/10.1016/j.ijpharm.2018.10.057.
Verma P, Ahuja M. Cubic liquid crystalline nanoparticles: optimization and evaluation for ocular delivery of tropicamide. Drug Deliv. 2016;23(8):3043–54. https://doi.org/10.3109/10717544.2016.1143057.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nasr, M., Teiama, M., Ismail, A. et al. In vitro and in vivo evaluation of cubosomal nanoparticles as an ocular delivery system for fluconazole in treatment of keratomycosis. Drug Deliv. and Transl. Res. 10, 1841–1852 (2020). https://doi.org/10.1007/s13346-020-00830-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-020-00830-4