Abstract
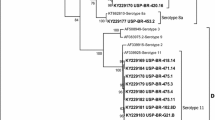
The present study was carried out for detection and molecular characterization of fowl adenoviruses (FAdVs) associated with hydropericardium syndrome or inclusion body hepatitis in commercial broiler chickens. The FAdVs were detected in liver samples from 33 commercial broiler chicken flocks by polymerase chain reaction (PCR) using hexon gene specific primers. The restriction enzyme analysis using StyI, BsiWI, MluI, AspI, BglI and ScaI enzymes of all the 33 FAdV-positive samples revealed FAdV-4 in 10 samples, FAdV-8 in five samples, FAdV-2 and FAdV-12 in two samples each, and FAdV-5 and FAdV-6 in one sample each. Twelve samples revealed the digestion pattern for more than one serotypes with FAdV-8 and FAdV-5, FAdV-8 and FAdV-7, FAdV-8 and FAdV-6, FAdV-8 and FAdV-12 being the predominant combinations indicating mixed infection. The serotypes FAdV-2 and FAdV-5 have not been detected previously in the country. The purified PCR products of FAdVs of four samples (HR 1, HR 2, HR 3 and HR 4) were cloned and sequenced. Phylogenetic analysis revealed that FAdVs of all four samples clustered in separate groups consistent with the REA pattern. In conclusion, this study reveals the presence of FAdV-2, FAdV-4, FAdV-5, FAdV-6, FAdV-7, FAdV-8 and FAdV-12 in broiler chickens affected with hydropericardium syndrome or inclusion body hepatitis in India.


Similar content being viewed by others
References
Asthana M, Chandra R, Kumar R. Hydropericardium syndrome: current state and future developments. Arch Virol. 2013;158:921–31.
Chandra R, Dixit VP, Kumar M. Inclusion body hepatitis in domesticated and wild birds: a review. Indian J Virol. 1998;14:1–12.
Chiocca S, Kurzbauer R, Schaffner G, Baker A, Mautner V, Cotton M. The complete DNA sequence and genomic organization of the avian adenovirus CELO. J Virol. 1996;70:2939–49.
Choi KS, Kye SJ, Kim JY, Jeon WJ, Lee EK, Park KY, Sung HW. Epidemiological investigation of outbreaks of fowl adenovirus infection in commercial chickens in Korea. Poult Sci. 2012;91:2502–6.
Dar A, Gomis S, Shirley I, Mutwiri G, Brownlie R, Potter A, Gerdts V, Tikoo SK. Pathotypic and molecular characterization of a fowl adenovirus associated with inclusion body hepatitis in Saskatchewan chickens. Avian Dis. 2012;56:73–81.
Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA. Virus taxonomy-eighth report of the international committee on the taxonomy of viruses. San Diego: Elsevier Acad Press; 2005.
Ganesh K, Suryanarayana VVS, Raghavan R, Gowda S. Nucleotide sequence of L1 and part of P1 of hexon gene of fowl adenovirus associated with hydropericardium hepatitis syndrome differs with the corresponding region of other fowl adenoviruses. Vet Microbiol. 2001;78:1–11.
Gomis S, Goodhope AR, Ojkic AD, Willson P. Inclusion body hepatitis as a primary disease in broilers in Saskatchewan, Canada. Avian Dis. 2006;50:550–5.
Kim JN, Byun SH, Kim MJ, Kim J, Sung HW, Mo IP. Outbreaks of hydropericardium syndrome and molecular characterization of Korean fowl adenoviral isolates. Avian Dis. 2008;52:526–30.
Mase M, Nakamura K, Imada T. Characterization of fowl adenovirus serotype 4 isolated from chickens with hydropericardium syndrome based on analysis of the short fiber protein gene. J Vet Diagn Invest. 2010;22:218–23.
Mazaheri A, Prusas C, Vob M, Hess M. Some strains of serotype 4 fowl adenoviruses cause inclusion body hepatitis and hydropericardium syndrome in chickens. Avian Pathol. 1998;27:269–76.
Meulemans G, Boschmans M, Van den Berg TP, Decaesstecker M. Polymerase chain reaction combined with restriction enzyme analysis for detection and differentiation of fowl adenovirus. Avian Pathol. 2001;24:693–706.
Meulemans G, Couvreur B, Decaesstecker M, Boschmans M, Van den Berg TP. Phylogenetic analysis of fowl adenoviruses. Avian Pathol. 2004;33:164–70.
Mittal D, Khokhar RS, Jindal N. Diagnosis of the inclusion body hepatitis–hydropericardium syndrome using conventional techniques. Haryana Vet. 2011;50:53–6.
Nakamura K, Mase M, Yamamoto Y, Takizawa K, Kabeya M, Wakuda T, Matsuda M, Chikuba T, Yamamoto Y, Ohyama T, Takahashi K, Sato N, Akiyama N, Honma H, Imai K. Inclusion body hepatitis caused by fowl adenovirus in broiler chickens in Japan, 2009–2010. Avian Dis. 2011;55:719–23.
Ojkic D, Martin E, Swinton J, Vaillancourt JP, Boulianne M, Gomis S. Genotyping of Canadian isolates of fowl adenoviruses. Avian Pathol. 2008;37:95–100.
Parthiban M, Manoharan S, Roy P, Chandran NDJ, Aruni AW, Koteeswaran A. Nucleotide sequence analysis of the L1 loop variable region of hexon gene of fowl adenovirus 4 isolates from India. Acta Virol. 2005;49:65–8.
Philippe C, Grgic H, Ojkic D, Nagy E. Serologic monitoring of a broiler breeder flock previously affected by inclusion body hepatitis and testing of the progeny for vertical transmission of fowl adenoviruses. Can J Vet Res. 2007;71:98–102.
Rahul S, Kataria JM, Senthilkumar N, Dhama K, Sylvester SA, Uma R. Association of fowl adenovirus serotype 12 with hydropericardium syndrome of poultry in India. Acta Virol. 2005;49:139–43.
Singh A, Oberoi MS, Grewal GS, Hafez HM, Hess M. The use of PCR combined with restriction enzyme analysis to characterize fowl adenovirus field isolates from northern India. Vet Res Commun. 2002;26:577–85.
Singh A, Oberoi MS, Jand SK, Singh B. Epidemiology of inclusion body hepatitis in poultry in northern India from 1990 to 1994. Rev sci tech Off int Epiz. 1996;15(3):1053–60.
Steer PA, O’Rourke D, Ghorashi SA, Noormohammadi AH. Application of high-resolution melting curve analysis for typing of fowl adenoviruses in field cases of inclusion body hepatitis. Aust Vet J. 2011;89:184–92.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
13337_2013_183_MOESM1_ESM.jpg
Supplemental Fig. 1a Alignment of deduced amino acid sequences of hexon genevariable region of fowl adenovirus serotypes. Amino acid residuesdiffering from majority consensus are boxed. Amino acid positionsare as per Chiocca et al. [3] (JPG 4266 kb)
13337_2013_183_MOESM2_ESM.jpg
Supplemental Fig. 1b Alignment of deduced amino acid sequences of hexon genevariable region of fowl adenovirus serotypes. Amino acid residuesdiffering from majority consensus are boxed. Amino acid positionsare as per Chiocca et al. [3] (JPG 2469 kb)
Rights and permissions
About this article
Cite this article
Mittal, D., Jindal, N., Tiwari, A.K. et al. Characterization of fowl adenoviruses associated with hydropericardium syndrome and inclusion body hepatitis in broiler chickens. VirusDis. 25, 114–119 (2014). https://doi.org/10.1007/s13337-013-0183-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-013-0183-7