Abstract
Ethanedinitrile is a chemical soil fumigant with promising efficacy against several key pests including weeds, nematodes, and soil-borne pathogens. The efficacy of 12 concentrations of ethanedinitrile, ranging from 8.7 to 1,751 mg kg− 1 soil, to control seeds and tubers of five weed species (Cyperus esculentus, C. rotundus, Malva parviflora, Portulaca oleracea, and Stellaria media), two nematode species (Globodera rostochiennsis and Tylenchulus semipenetrans), and four pathogenic fungal species (Fusarium oxysporum, Macrophomina phaseolina, Pythium ultimum, and Verticillium dahlia) were evaluated in acidic sand (pH: 5.6) and alkaline sandy loam (pH: 7.5–7.6), under controlled laboratory conditions. These pathogens and weeds are common in strawberry and vegetable fields and have been targeted by soil fumigants. Ethanedinitrile was injected into microcosms for 24 h. Lower doses of ethanedinitrile controlled fungal pathogens and nematodes better in acidic sand than in alkaline sandy loam. However, the reverse tended to be true for weed control as higher doses of ethanedinitrile in the acidic sand were required to control weeds than in the alkaline sandy loam. Results showed that ethanedinitrile can provide excellent control of soil-borne nematodes, pathogenic fungi, and key weed species in acidic sand and alkaline sandy loam.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Major impacts on crop productivity can occur via infestations from several fungal pathogens, plant-parasitic nematodes, and recalcitrant weed species. Common pathogens found in California fruit, vegetable, and ornamental cropping systems include Pythium ultimum, Verticillium dahliae, Macrophomina phaseolina, and Fusarium oxysporum (Boyd et al. 2017; Lecomte et al. 2016; Mace et al. 1981; Rosskopf et al. 2007; Subbarao et al. 2007). Weeds are also a problem in California vegetable production as they compete with crops for light, water, nutrients, and space. The most invasive weeds of vegetable crops are purple nutsedge (Cyperus rotundus) and yellow nutsedge (C. esculentus) (Webster 2010). Other weeds commonly found in California production fields are purslane (Portulaca oleracea), cheeseweed (Malva parviflora) (Rosskopf et al. 2007), and chickweed (Stellaria media). Nematodes that feed on plant material such as citrus nematode (Tylenchulus semipenetrans) and cyst nematode (Globodera rostochiensis) cause damage by feeding on roots or by predisposing plants to soil-borne pathogens.
Pre-plant soil fumigants are commonly used for broad-spectrum control of soil-borne pathogens, weeds, and nematodes in high-value crops including strawberries, tomatoes, and cut flowers. Methyl bromide (MeBr) was the most widely applied pre-plant soil fumigant due to its ability to control a broad spectrum of soil-borne pests, which enabled the production of acceptable commercial yields (Klein 1996). MeBr was identified as an ozone-depleting substance which led to its subsequent phase-out under the Montreal Protocol (UNEP 2006). Alternative fumigants available are 1,3-dichloropropene (1,3-D), allyl isothiocyanate, chloropicrin (Pic), and methyl isothiocyanate (MITC) generators such as metam sodium or metam potassium. However, these soil fumigant chemistries exhibit high boiling points and low vapour pressures (Ajwa et al. 2010) resulting in inconsistent control of many pests following soil fumigation with these alternatives (Boyd et al. 2016; Eure and Culpepper 2017; Hanson and Shrestha 2006; Mace et al. 1981; Subbarao et al. 2007; Wu et al. 2008; Zasada et al. 2010).
Ethanedinitrile (EDN) is a broad-spectrum fumigant with efficacy against many pathogens, weeds, and nematodes and has been recommended as a replacement to MeBr (Mattner et al. 2006). EDN is an ozone safe chemical registered in Australia for pre-plant soil fumigation and post-harvest treatment of timber and logs. Residue studies used for EDN registration showed that there are no toxic residues in the soil after 7 days. There are no EDN residues in harvested plants because it degrades within days in soil prior to planting the crop (Ajwa et al. 2016). The Solar Impulse Foundation, an organisation that independently assesses products and programs to address environmental challenges without compromising economic growth, recently approved EDN as an efficient solution to protect the environment. Like MeBr, EDN is a gas at ambient temperature and atmospheric pressure with a boiling point of -21 °C and high vapour pressure (3.9 × 103 mmg Hg at 21 °C) (Matheson Tri-Gas 2010). This enables a high degree of diffusion and distribution in the soil for effective pest control. EDN degrades within 2 weeks of application, allowing growers to plant a new crop quickly after fumigation with no residue in the harvested plant (Ajwa 2017). Field emission following drip or shank application under totally impermeable film (TIF) is very low (Ajwa 2017). Additionally, the fumigation equipment and application technology used for MeBr can be implemented for EDN application, increasing adoptability of this new chemical.
Successful control of pests under field conditions is highly dependent on soil conditions such as texture, pH, moisture, temperature, and organic matter content (Ajwa et al. 2010). This compound reacts with moisture to create cyanide, which targets cytochrome C oxidase and prevents the tissue from using oxygen (Leavesley et al. 2008). The fate of EDN in soil is largely controlled by temperature and pH solutions. Lai Wang et al. (1987) showed that C2N2 hydrolysis is faster in alkaline solution than acidic solution. Ajwa et al. (2016) presented hydrolysis rate constants at different temperature and pH values and showed that C2N2 is stable in pH solution at 10 °C, but the half-life decreases with higher temperature and/or soil solution pH. The half-life at 23 °C decreases from 28 days in pH 4.0 to 49 min in pH 7.0 and 4.5 min in pH 9.0 solution. Nevertheless, for a fumigant to be effective it must remain in contact with the target organism for sufficient time and in sufficient concentration to kill (Munnecke and Van Gundy 1979). Field evaluation of fumigant efficacy is expensive and time consuming due to variability found in natural conditions (Wang et al. 2004). Biological assays have been conducted under laboratory conditions to measure accurate and reliable dose response data for various soil fumigants (Hutchinson et al. 1999; Klose et al. 2007, 2008). This is evaluated based on a fumigant exposure index (CT) where C is the fumigant concentration and T is the time in hours (McKenry and Thomason 1976; Wang et al. 2004). From these data, an effective concentration required to obtain maximum control for the target organisms in field conditions can be predicted.
EDN has been shown to be effective against several weeds, soil-borne pathogens, and nematodes (Lecomte et al. 2016; Mattner et al. 2006; Ren et al. 2002, 2003; Rosskopf et al. 2007; Stevens et al. 2019). However, limited information is available on the dose-response of EDN against these pests. The objective of this research was to develop a dose-response to determine the EDN concentrations necessary to reduce the viability of five weed seeds and tubers, four fungal pathogens, and two nematodes by 50% (Lethal concentration, LC50) and 90% (LC90) in two soils that have different pH values under laboratory conditions. This study follows similar procedures to those outlined by Klose et al. (2007; 2008).
Materials and methods
EDN concentrations
EDN (Lucebni Zavody Draslovka, Havlickova 605, 280 02 Kolin, Czech Republic) (purity: 99.98%) was prepared by inflating Tedlar bags with pressurised EDN at 20oC. A known amount of EDN gas was taken from the Tedlar bags using a gas-tight syringe and injected into a pre-sealed 1-L incubation jar via septa installed on the jar lid. The jars (containing a total of 0.5 kg soil) received the following volumetric concentrations of EDN: 0, 2, 5, 10, 15, 20, 25, 50, 100, 150, 250, 400 µL EDN gas per jar. EDN concentration in the Tedlar bag was 2.189 mg mL-1 at 20oC. The volumetric amounts above were converted into mg kg-1 soil by multiplying the volumetric dose in each jar by (2.198 mg/0.5 kg). Therefore, EDN concentrations in the soils were 0, 8.8, 21.9, 43.8, 65.7, 87.6, 109.5, 218.9, 437.8, 656.7, 1,094.5, 1,751.2 mg kg-1 soil. These doses are equivalent to field application rates ranging from 0 to 3,923 kg ha-1. Potential field application rates range from 500 to 1200 kg ha-1.
Soil types
Two soils with different pH values were used in this study. The alkaline soil, referred to as the alkaline sandy loam, was collected from an agricultural field located at the USDA-University of California research facility in Salinas, California (36°37.434 N and 121°32.492 W). This soil has been used for strawberry and lettuce production for the past 20 years. Composite samples were collected within 100 m radius of this GPS location. The soil at this location had a pH of 7.5 to 7.6. Soil samples were collected at 0–15 cm depth. Soil sub-samples were homogenised and stored at room temperature in a plastic container. The alkaline soil type was a Chualar loamy sand soil (Fine loamy, Mixed, Thermic, Typic Argixerroll) soil containing 0.7% organic matter, 75% sand, 13% silt and 12% clay.
The acidic soil, referred to as the acidic sand, was collected from Bethel Farms in Fort Pierce, Florida (27°31’ 13.92 N and 80° 27’ 27.21 W) and was a sandy soil containing 96% sand, 1% silt and 3% clay with a pH of 5.6 and organic matter content of 0.8%. This soil is mainly used for commercial turfgrass production. Composite samples were collected within a 200 m radius of this GPS location at 0–15 cm depth. Soil sub-samples were homogenised and stored at room temperature in a plastic container. When ready for analysis, soils were air dried, passed through a 2 mm-aperture sieve, and thoroughly mixed.
Sample preparation
Ten grams of soils naturally infested with M. phaseolina (10 propagules g-1 soil) from an agricultural field were placed in nylon mesh bags. Similarly, ten grams of soils naturally infested with V. dahliae (10 microsclerotia g-1 soil), P. ultimum (500 propagules g-1 soil), and F. oxysporum (1,500 propagules g-1 soil) from another field were placed in individual nylon mesh bags.
Verticillum dahliae was identified by air-drying soils in the laboratory at 23 °C ± 2 °C. After thoroughly mixing and pulverising soil, 10 g was placed in snap cap vials and mixed with 2.5 mL of a 7.5 mg mL-1 DL-methionine solution. Vials were capped and incubated for 1 week in complete darkness at 30 °C. Following incubation, vials were opened and air-dried for 1 week at 22–24 °C. Each sample was pulverised and distributed onto petri dishes containing modified NP-10 selective medium using the modified Anderson sampler. With the Anderson sampler, 0.5 g of soil from each sample was distributed over six petri dishes and replicated twice. Plates were incubated in the dark at 22–24 °C for 3 weeks. Following incubation, the agar media was gently washed under running tap water to dislodge and remove soil particles. Washed petri dishes were examined for V. dahlia microsclerotia clusters using a dissecting microscope with transmitted light.
Pythium ultimum was identified by adding 1 g of air-dried soil to 10 mL of sterilised water blanks, vortexed, then 500 µL distributed evenly over a petri dish containing a semi-selective Pythium medium. The medium consisted of corn meal agar (17 g L-1, BBL, Kansas City, MO, USA) amended with 0.1% Tween 20 (Sigma-Aldrich, St. Louis, Missouri, USA) followed by 10 mg L-1 pimaricin (Sigma-Aldrich, St. Louis, Missouri, USA), 100 mg L-1 penicillin (Sigma-Aldrich, St. Louis, Missouri, USA), 250 mg L-1 ampicillin (Sigma-Aldrich, St. Louis, Missouri, USA), 10 mg L-1 rifampicin (Sigma-Aldrich, St. Louis, Missouri, USA), 50 mg L-1 rose bengal (Sigma-Aldrich, St. Louis, Missouri, USA), and 20 mg L-1 benomyl (Sigma-Aldrich, St. Louis, Missouri, USA) when medium had cooled to 50 °C. Following incubation at 25 °C for 24 h, colonies were visually identified using a dissecting microscope with transmitted light.
For F. oxysporum, soil samples were air-dried in the laboratory at 23 °C-21 °C), thoroughly mixed, and pulverised using mortar and pestle. Soil (15 g) was mixed with 50 mL of 0.05% water agar for 1 min to release spores from the soil. The soil suspension was then spread onto Komada’s modified medium, incubated at 25 °C for 1 week, and visually identified using a dissecting microscope with transmitted light.
Macrophomina phaseolina was identified by growing isolates on potato dextrose agar (39 g PDA dissolved in 1 L water). Methods used were described by Zveibil et al. (2012) and modified with 0.05 g rifampicin and 1 mL Tergitol NP-10 (Sigma-Aldrich, St. Louis, MO, USA) to suppress bacterial growth. PDA was autoclaved and cooled to 55 °C. Isolates were incubated on PDA for 1 week at 25 °C, then identified with a dissecting microscope with transmitted light.
A Glen County, California citrus orchard provided soil that was naturally infested with T. semipenetrans. Soil naturally infested with G. rostochiensis was obtained from a San Joaquin Valley, California potato field. All nematode species were extracted from the soil using a modified Baermann Funnel system (Ayoub 1977). Field soil (50 cm3) was kept on funnels at room temperature for 72 h. Nematodes were identified microscopically using methods described by Van Gundy (1958) and Baldwin et al. (1991). For these samples, small muslin bags (Hubco Soil Sample Bags, Forestry Suppliers, Inc., Jackson, Mississippi, USA) held 50 g of infested soil and were firmly tied. Each sample was placed in the microcosms described below. The total soil weight in the jar including inocula soils was 500 g.
The inocula bags were placed in the centre of the bulk soil inside each jar. The bulk soil was sieved (2 mm-aperture sieve) to remove large objects and debris, and then autoclaved and air dried before placing it inside the mesocosms. The microcosms consisted of 1 L jars. The bulk soil was preconditioned by adding water to adjust the moisture to 70% of the field water holding capacity. The pathogen inocula bags, weed seeds, and tubers were then placed in the centre of the bulk soil (approximately 425 g). The total soil weight in the jar including inocula soils was approximately 500 g. The mesocosm containing the soil and inocula was lightly tapped to reduce the amount of air gaps and to achieve a soil bulk density of 0.9 to 1.0 g/mL for the acidic sand and 1.1 to 1.2 g/mL for the alkaline sandy loam.
Weed seeds and tubers were obtained from commercial sources or harvested from fields. Stellaria media seeds were purchased (Herbiseed, New Farm, Mire Lane, West End, Twyford, RG10 0NJ, England). Cyperus esculentus, C. rotundus, M. parviflora, and Portulaca oleracea seeds were obtained from Salinas Valley, California, field weeds, and were transplanted and grown in a greenhouse or outdoor microplots. Up to 50 weed seeds per species were placed into each mesh bag (Delnet Applied Extrusion Technologies, Inc., Middletown, Delaware, USA). Weed seed germination assays were prepared as described by Haar et al. (2003). Twenty tubers of dormant C. esculentus and nutlets of C. rotundus were also placed into separate mesh bags. Following placement of infested soil and weed seeds, each bag was cut to dimensions of 5 cm × 5 cm and stored at room temperature for ≤ 5 days.
Microcosms were vented under a fume hood for 30 min at 20 °C following the 24 h fumigation event. Bags were rinsed with deionised water, sorted, and allowed to vent under a laminar flow hood for 6 h at 20 °C to discharge any residual fumigant. EDN gas inside each jar was measured with a high-speed gas chromatograph 5 to 10 min after EDN injection and 24 h after injection, immediately prior to venting the jars. The vapour phase in the mesocosms was collected by withdrawing 0.05 mL via an air-tight syringe and injected into a high-speed micro gas chromatograph with thermal conductivity detector (µGC-TCD, MTI model P200H with EZChrom 200 software, MTI Instruments, Fremont, CA, USA). The GC oven was operated isothermally (40 °C) to separate the analytes (C2N2 (EDN) and hydrogen cyanide (HCN)) into discrete peaks using MS-5 and OV-1 capillary columns. EDN and HCN eluting from the GC columns were identified and quantified by comparing the retention time and response with those of standards stored in a reference library data base. Concentrations were determined against calibration curves for EDN or HCN (> 99.9 purity).
Sample retrieval and data collection
Weed seed viability was assessed with tetrazolium chloride (TTC) upon removal (Grabe 1970). Seeds from each bag were sorted and placed on filter paper (Whatman no. 1) with 1 mL of deionised water in plastic petri dishes (100 mm diameter). Petri dishes were sealed and germinated at constant 21 °C in darkness for 20 to 24 h. Seeds used for the TTC viability test that had imbibed were cut and placed on filter paper moistened with 1 mL of a 0.1% (w:v) 2,3,5-TTC solution, then examined under the microscope. Remaining seeds were allowed to germinate in petri dishes for another 20 to 24 h prior to assessment. For C. esculentus and C. rotundus germination, tubers were planted in small pots containing moist sand and incubated inside a temperature-controlled incubator. The incubator was programmed to provide a 20/10°C day/night temperature cycle for two weeks. Water content in the germination pots was adjusted every 48 h. Viability was calculated as the percentage of viable seeds or tubers after fumigation compared to viability at the beginning of the experiment (time zero).
Mortality assessment of V. dahlia was carried out by air-drying soil at 20 °C and homogenising samples. Each sample had 10 g soil placed into a snap cap vial, combined with 2.5 mL of a DL-methionine solution (7.5 mg mL-1), and incubated for one week at 30 °C (Kapulnik et al. 1985). Samples were allowed to air dry for 1 week at 22 to 24 °C followed by placing samples onto NP-10 selective medium in petri dishes using the modified Anderson sampler (Butterfield and DeVay 1977; Kabir et al. 2004). Following incubation at 22 to 24 °C for 3 weeks, petri dishes were washed clear of soil particles and assessed for V. dahliae microsclerotia using a dissecting microscope with transmitted light.
Fusarium oxysporum and P. ultimum samples were assessed by vortexing 1 g of air-dried soil with 10 mL sterile water, then distributing 500 µL of the mixture onto a semi-selective Pythium medium with 5 petri plates and triplicates per sample, respectively. The medium used comprised of 17 g L-1 corn meal (BBL, Kansas City, Missouri). Immediately following autoclaving, once the medium had cooled to 50 °C, 0.1% Tween 20, ampicillin (250 mg L-1), penicillin (100 mg L-1), rose bengal (50 mg L-1), benomyl 50WP (20 mg L-1, DuPont, Wilmington, Delaware), pimaricin (10 mg L-1), and rifampicin (10 mg L-1) were added to the medium (Martin 1992). Plates were incubated for 24 h at 25 °C and washed free of soil particles prior to counting colonies. Colony forming units (CFU) were counted a second time after 24 h incubation.
Determination of M. phaseolina control was carried out using potato dextrose agar. PDA (39 g PDA dissolved in 1 L water) was modified using 0.05 g rifampicin and 1 mL Tergitol NP-10 (Zveibil et al. 2012). Media was autoclaved and allowed to cool to 55 °C before use. Isolates were plated and incubated on PDA for 1 week at 25 °C, then identified and counted with a dissecting microscope with transmitted light.
Citrus and cyst nematode population densities were assessed using a modified Baermann Funnel system (Ayoub 1977). Muslin bags with 50 cm3 of infested field soil were kept on funnels at room temperature for 72 h. Nematodes were identified as previously described and counted microscopically using a 10 mL aliquot from each sample.
Experimental design and statistical analysis
Two experiments were conducted between February and September 2018. These experiments were set up as a randomised complete block design with four replications. The effect of pH and fumigant concentration on the mortality of pathogens, nematodes, and weed seeds and tubers was assessed using multivariate analysis of variance (ANOVA). A 3-parameter sigmoidal logistic regression model (1) was used to describe the relationship between the percentage of pest mortality and the logarithm of the fumigant concentration. Model fit was assessed using R2 values and standard error estimates.
In this model, Y is pest mortality as a function of the logarithm of the EDN concentration (x). Sigmoidal logistic regression models were computed using SigmaPlot 2010 (Systat Software Inc., San Jose, California, USA). Determination of the EDN concentration required to achieve 50% (LC50) or 90% (LC90) pest mortality was carried out using non-linear logistic dose-response regressions with SAS probit procedure (SAS Institute, Inc., Cary, North Carolina). One-way ANOVA assessed the effect of pH on LC50 and LC90. When error probability of ≤ 5% (P ≤ 0.05) was revealed by the F test, means were separated using the Mann-Whitney U test (SPSS Inc., Chicago, Illinois, USA).
Results
EDN concentrations inside each jar were measured within 10 min of the initial EDN injection and again after 24 h, immediately prior to opening the jar. After 24 h, EDN concentrations in the alkaline sandy loam were negligible and were less than 10% of the initial measurements in the acidic sand for EDN doses below 220 mg kg− 1. At rates above 220 mg kg− 1, approximately 49% of the applied EDN partitioned into the solid/liquid phases of the acidic sand within 10 min after EDN injection (Figs. 1 and 2). In the alkaline sandy loam, approximately 23% of the applied EDN partitioned into the solid/liquid phases within 10 min. The amounts of EDN in jars receiving higher doses (> 400 mg kg− 1) were similar in both soils, indicating EDN saturation (data not shown).
Sensitivity of fungal pathogens and nematodes to soil fumigation with EDN varied substantially within each species studied. Sigmoidal dose-response curves for all fungal pathogens and nematodes described a good fit, with high adjusted R2 values (> 0.97), for increases in mortality rate with increased fumigant concentrations (Figs. 3 and 4). A lower slope (b) value of the sigmoidal models indicated increased mortality rate (Tables 1 and 2).
Response of (a) Fusarium oxysporum, (b) Macrophomina phaseolina, (c) Verticillium dahlia, and (d) Pythium ultimum to various ethanedinitrile (EDN) concentrations after 24 h in acidic sand and alkaline sandy loam. Closed circles indicate acidic sand, open triangles indicate alkaline loamy sand; bar markers indicate standard deviation
Probit regressions on logistic dose indicated that the minimum concentration necessary to eradicate 90% (LC90) of the target nematode and pathogenic fungus populations ranged from 14 to 78 mg EDN kg-1 in acidic sand and 58 to 180 mg EDN kg-1 in alkaline sandy loam (Tables 1 and 2). The sensitivity of these pests and pathogens decreased in the order T. semipenetrans < G. rostochiensis < F. oxysporum < M. phaseolina < V. dahlia < P.ultimum in acidic sand (Table 1). For the alkaline sandy loam, EDN sensitivity from most to least sensitive was in the order T. semipenetrans < P. ultimum < F. oxysporum = V. dahliae < G. rostochiensis < M. phaseolina (Table 2). Concentrations required to control 50% (LC50) of these pests and pathogens during the course of 24 h exposure at 20 ºC was < 53 and < 129 mg EDN kg-1 in acidic sand and alkaline loamy sand soil, respectively.
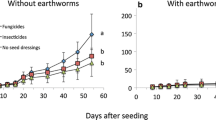
The sigmoidal dose-response models described a good fit for increased weed seed mortality rate with EDN concentrations for C. rotundus, P. oleracea, and S. media (Fig. 5). The minimum concentration required to kill 90% (LC90) of these weed seeds and tubers ranged between 56 and 318 mg EDN kg-1 in acidic sand (Table 1) and between 81 and 103 mg EDN kg-1 in alkaline sandy loam (Table 2). The minimum concentration required to control 50% (LC50) of these weeds in acidic sand ranged from 31 to 59 mg EDN kg-1 and 31 to 109 mg EDN kg-1 in alkaline sandy loam. Malva parviflora seeds and dormant C. esculentus tubers were least sensitive to fumigation with EDN among all species in this study (Tables 1 and 2). The sigmoidal model in both soils showed poor fit for these weeds (Fig. 5).
Response of (a) Cyperus rotundus, (b) Portulaca oleracea, (c) Stellaria media, (d) Malva parviflora, and (e) Cyperus esculentus to various ethanedinitrile (EDN) concentrations after 24 h in acidic sand and alkaline sandy loam. Closed circles indicate acidic sand, open triangles indicate alkaline loamy sand; bar markers indicate standard deviation
Discussion
EDN concentrations during incubation
When compared to the alkaline sandy loam, EDN concentrations in the acidic sand was approximately 47% higher at the same dose rate. Based on these laboratory results, we speculate that EDN degradation is at least partially dependent on soil pH, similar to previous results indicating fumigant degradation is faster for some fumigants in acidic soil versus alkaline soils (Qin et al. 2016). Many laboratory studies have also reported an effect of soil pH on adsorption and desorption of weakly acidic pesticides (Boivin et al. 2005; Kah et al. 2007; Ajwa et al. 2016) reported a half-life of 28 days and 6 min for hydrolysed EDN (20 to 23 °C) when pH was 4.0 and 9.0, respectively. While these results showed that soil pH plays a role in fumigant efficacy, other soil-based factors, such as organic matter, moisture, temperature, and microbial populations have also been shown to have an impact on fumigant efficacy but were not explored in this study. Studies have shown that soil moisture affects fumigant efficacy in sandy loam soils with the highest efficacy occurring when soil moisture was 14–15% (Haar et al. 2003; Zhang et al. 1998). Further studies have shown that adsorption behaviour of EDN in acidic, neutral, and alkaline soils was also impacted by soil organic matter (Zhou et al. 2019). In addition, the present experiments were performed under an artificially controlled environment in sealed glass jars which does not account for fumigant losses due to environmental and practical factors.
Due to the higher concentration of EDN in the acidic sand, it is expected that effective control of pests can occur at lower application rates. EDN in the alkaline sandy loam was unstable and is expected to degrade very quickly into hydrogen cyanide (HCN), which results in a lower concentration in the gaseous phase, but higher concentration in the liquid phase. Since HCN is soluble in water, it can penetrate the weed seeds imbibing water, resulting in higher weed mortality rates.
Dose-response to fungal pathogens and nematodes
In this study, pathogens and nematodes in the acidic sand required lower concentrations of EDN to provide 90% control of compared to concentrations required to achieve the same level of control in alkaline sandy loam. The higher dose required to control pathogens in neutral or alkaline sand can be attributed to the faster hydrolysis rate of EDN in the alkaline sandy loam than the acidic sand. However, concentrations required for 90% control of all pathogens and nematodes remained within the typical field application rates. These results agree with Waterford et al. (2006) which reported high efficacy of EDN against Pythium, Fusarium, Rhizoctonia, and Phytophthora species with and without soil. In addition, 90% control of P. ultimum with EDN required 78 mg kg-1 EDN, similar to our results (Klose et al. 2008).
The most sensitive fungal pathogen in this study was F. oxysporum. To control 50% of the population, 11 to 21 mg/kg-1 soil of EDN was required. Previous studies have reported a LC50 of 40 mg kg-1 soil for InLine (61% 1,3-D plus 33% Pic) (Klose et al. 2007).
Macrophomina phaseolina was the least sensitive to treatment with EDN, particularly in the alkaline sandy loam as it required 180 mg EDN kg-1 compared to 32 mg EDN kg-1 in the acidic sand. Boyd et al. (2017) reported the effect of EDN on M. phaseolina in production fields with pH of 6.5 and 7.6. In both soils, EDN provided excellent control of M. phaseolina. In addition, field studies conducted in Florida reported significantly reduced M. phaseolina populations when inoculated bags were placed at multiple locations in a raised bed in three soil types (Yu et al. 2020). These results agree with the current study, as field application rates are sufficient for control of M. phaseolina in a range of soils.
Furthermore, efficacy of EDN against plant-parasitic nematodes has been evaluated in laboratory and field studies. Rosskopf et al. (2007) reported 100% control of Meloidogyne incognita at the lowest EDN rate tested (20 mg L-1). Toxicity of EDN to Steinernema carpocapsae was also evaluated in a laboratory study, which reported an EDN concentration of 40 mg L-1 was required for 95% control when exposed for 5 h, while 100 mg MeBr L-1 was required for the same level of control (Waterford et al. 2006).
Dose response to weed seeds and tubers
These data agree with previous findings which reported adequate control of S. media and P. oleracea when treated with EDN. One laboratory study found that the lowest concentration tested (20 mg EDN L-1) was sufficient for complete control of P. oleracea (Rosskopf et al. 2007; Thalavaisundaram et al. 2015) also reported similar control of S. media when treated with EDN compared to MeBr.
Previous laboratory studies have found that the lowest concentration of EDN (20 mg L-1) significantly reduced C. rotundus and C. esculentus tubers compared to an untreated control (Rosskopf et al. 2007). In the present study, control of C. esculentus was achieved at a lower concentration in the acidic sand (pH: 7.5–7.6) compared to the alkaline sandy loam. Conversely, C. rotundus control in alkaline sandy loam (pH: 5.6) was achieved at lower concentrations than the acidic sand. Lesser efficacy against C. esculentus in alkaline sandy loam may be due to the physiological dormancy of the tubers. In alkaline sandy loam, it is likely that HCN is generated and penetrates tubers as they imbibe, therefore, dormant tubers may be more tolerant of EDN when applied in alkaline sandy loam. Despite these results, control of C. rotundus under field conditions in acidic sand with EDN at typical field use rates has shown to be highly effective (Stevens et al. 2019; Yu et al. 2020).
Malva parviflora seeds were not sensitive to EDN in this study. Malva parviflora is a recalcitrant weed with a hard seed coat, which reduces penetrability of fumigants. Malva parviflora has been reported to be resistant to applications of Pic, 1,3-D, 1,3-D:Pic mixtures, and MeBr:Pic mixtures (Fennimore et al. 2003; Haar et al. 2003). In a laboratory study, only 34% of seeds were killed when exposed to the highest rate of a 1:3-D:Pic mixture for 24 h (Klose et al. 2007). Additionally, the highest concentration of metam sodium was not sufficient to control seeds of M. parviflora (Klose et al. 2008). The authors reported M. parviflora was the only weed species that did not exhibit increased mortality when exposed to fumigant at higher soil temperatures (10 and 20 °C).
As the microcosms allowed pests to remain in constant soil type, moisture, and temperature, we can conclude that control of these pests was a function of EDN concentration and exposure time. While effects of soil moisture, temperature, and texture on fumigant efficacy have been explored (Hutchinson et al. 1999; Klose et al. 2007; Qiao et al. 2010; Zhang et al. 1997), the sole effect of soil pH has not been widely reported. These results suggest soil pH influences the sensitivity of the test pathogens and nematodes with higher mortality in acidic sand at lower EDN concentrations. Results from this study indicate that EDN provides excellent control of soil-borne pathogens, two nematodes, and select weed species at concentrations similar to typical field application rates.
References
Ajwa H (2017) EDN (Ethanedinitrile) Degradation in Soil After Shank and Drip Application Under TIF. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions
Ajwa H, Ntow WJ, Qin R, Gao S (2010) Properties of Soil Fumigants and their fate in the Environment, third edit. Elsevier Inc
Ajwa H, Xuan R, Guerrero J (2016) Fate of EDN Fumigant in Soil. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions. Orlando, FL
Ayoub S (1977) Plant Nematology: an Agricultural Training Aid. State of California, Department of Food and Agriculture
Baldwin JG (1991) M–OM, and NWR Manual of agricultural nematology
Boivin A, Cherrier R, Schiavon M (2005) A comparison of five pesticides adsorption and desorption processes in thirteen contrasting field soils. Chemosphere 61:668–676. https://doi.org/10.1016/j.chemosphere.2005.03.024
Boyd NS, Peres N, Baggio J et al (2017) Evaluation of EDN Fumigas for use in Strawberry. FSREF Res Rep 2017–2018:1–6
Boyd NS, Vallad GE, Noling JW (2016) Weed Control with Metam Potassium and Chloropicrin + 1,3-Dichloropropene Injected with Single and Dual Port Shanks. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions
Butterfield E, DeVay J (1977) Reassessment of soil assays for Verticillium dahliae. Phytopathology 67:1073–1078
Eure PM, Culpepper AS (2017) Bell Pepper and Weed Response to Dimethyl Disulfide Plus Chloropicrin and Herbicide Systems. Weed Technol 31:694–700. https://doi.org/10.1017/wet.2017.74
Fennimore S, Haar MJ, Ajwa H (2003) Weed control in strawberry provided by shank- and drip-applied methyl bromide alternative fumigants. HortScience 38:55–61
Grabe D (1970) Tetrazolium testing handbook for agricultural seeds. The handbook on seed testing. Association of Official Seed Analysts, p 62
Haar MJ, Fennimore S, Ajwa H, Winterbottom CQ (2003) Chloropicrin effect on weed seed viability. Crop Prot 22:109–115. https://doi.org/10.1016/S0261-2194(02)00119-9
Hanson BD, Shrestha A (2006) Weed control with methyl bromide alternatives. CAB reviews: perspectives in Agriculture, Veterinary Science. Nutr Nat Resour 1:1–13. https://doi.org/10.1079/PAVSNNR20061063
Hutchinson CM, McGiffen ME, Ohr HD et al (1999) Efficacy of methyl iodide soil fumigation for control of Meloidogyne incognita, Tylenchulus semipenetrans and Heterodera schachtii. Nematology 1:407–414. https://doi.org/10.1163/156854199508414
Kabir Z, Bhat RG, Subbarao KV (2004) Comparison of media for recovery of Verticillium dahliae from soil. Plant Dis 88:49–55. https://doi.org/10.1094/PDIS.2004.88.1.49
Kah M, Beulke S, Brown CD (2007) Factors influencing degradation of pesticides in soil. J Agric Food Chem 55:4487–4492. https://doi.org/10.1021/jf0635356
Kapulnik E, Quick J, DeVay J (1985) Germination of propagules of Verticilium dahliae in soil treated with methionine and other substances affecting ethylene production.Phytopathology75
Klein L (1996) Methyl Bromide as a Soil Fumigant. In: Bell C, Price N, Chakrabarti B (eds) The Methyl Bromide Issue. John Wiley & Sons, Inc., New York, pp 191–235
Klose S, Ajwa H, Browne GT et al (2008) Dose response of Weed Seeds, Plant-Parasitic Nematodes, and pathogens to twelve rates of Metam Sodium in a California soil. Plant Dis 92:1537–1546. https://doi.org/10.1094/PDIS-92-11-1537
Klose S, Ajwa H, Fennimore S et al (2007) Dose response of weed seeds and soilborne pathogens to 1,3-D and chloropicrin. Crop Prot 26:535–542. https://doi.org/10.1016/j.cropro.2006.05.004
Lai Wang Y, Lee HD, Beach MW, Margerum DW (1987) Kinetics of base hydrolysis of cyanogen and 1-cyanoformamide
Leavesley HB, Li L, Prabhakaran K et al (2008) Interaction of cyanide and nitric oxide with cytochrome c oxidase: implications for acute cyanide toxicity. Toxicol Sci 101:101–111. https://doi.org/10.1093/toxsci/kfm254
Lecomte C, Alabouvette C, Edel-Hermann V et al (2016) Biological control of ornamental plant diseases caused by Fusarium oxysporum: a review. Biol Control 101:17–30. https://doi.org/10.1016/j.biocontrol.2016.06.004
Mace M, Bell A, Beckman C (1981) Fungal Wilt Diseases of plants. Academic Press, New York
Martin FN (1992) The genus Pythium. In: Singleton L (ed) Methods for research on soil borne phytopathogenic fungi. Phytopathological Society, St. Paul, MN, pp 39–49
Matheson Tri-Gas Inc (2010) Material Safety Data Sheet. 1–7
Mattner SW, Gounder RK, Mann RC et al (2006) Ethanedinitrile (C2N2) - a novel soil fumigant for strawberry production. Acta Hortic 708:197–203
McKenry M, Thomason I (1976) Dosage values obtained following pre-plant fumigation for perennials. I. 1,3-dichloropropene nematicides in eleven field situations. Pestic Sci 7:521–534
Munnecke DE, Van Gundy SD (1979) Movement of fumigants in soil, dosage responses, and differential effects. Annu Rev Phytopathol 17:405–429. https://doi.org/10.1146/annurev.py.17.090179.002201
Qiao K, Wang HY, Shi XB et al (2010) Effects of 1,3-dichloropropene on nematode, weed seed viability and soil-borne pathogen. Crop Prot 29:1305–1310. https://doi.org/10.1016/j.cropro.2010.07.014
Qin R, Gao S, Ajwa H, Hanson BD (2016) Effect of application rate on fumigant degradation in five agricultural soils. Sci Total Environ 541:528–534. https://doi.org/10.1016/j.scitotenv.2015.09.062
Ren YL, Sarwar M, Wright EJ (2002) Development of Cyanogen for Soil Fumigation. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions. Orlando, FL
Ren YL, Waterford C, Matthiessen JN et al (2003) First Results from Ethanedinitrile (C2N2) Field Trials in Austrailia. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions. San Diego, CA
Rosskopf EN, Kokalis-Burelle N, Peterson GL, Waterford C (2007) Preliminary Investigation of Ethanedinitrile for Control of Weeds and Nematodes Important to Florida Production Systems. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions. San Diego, CA
Stevens MC, Freeman JH, Boyd NS (2019) Impact of Ethanedinitrile Rates and Application Method on Nutsedge Species and Tomato Root Galling. Crop Prot 116:1–6
Subbarao K, Kabir Z, Martin FN, Koike ST (2007) Management of soilborne diseases in strawberry using vegetable rotations. Plant Dis 91:964–972. https://doi.org/10.1094/PDIS-91-8-0964
Thalavaisundaram S, Mattner SW, Milinkovic M et al (2015) VIF Improves the Efficacy of EDN Fumigas in the Australian Strawberry Nursery Industry. In: Annual International Research Conference on MBr Alternatives and Emissions Reductions. San Diego, CA
UNEP UNEP (2006) Handbook for the Montreal protocol on substances that deplete the ozone layer-7th Edition. In: Sect. 1.1. Article 2H: Methyl Bromide. http://ozone.unep.org/Publications/MP_Handbook/Section_1.1_The_Montreal_Protocol/Article_2H.shtml. Accessed 12 May 2015
VanGundy SD (1958) The life history of the citrus nematode Tylenchulus semipenetrans Cobb. Nematologica 3:283–294
Wang D, He JM, Knuteson JA (2004) Concentration–time exposure index for modeling soil fumigation under various management scenarios. J Environ Qual 33:685. https://doi.org/10.2134/jeq2004.0685
Waterford C, Ren YL, Mattner SW, Sarwar M (2006) Ethanedinitrile (C2N2) - a Novel Soil Fumigant for Insect, Nematode, Pathogen, and Weed Control. Acta Hortic 708:197–203
Webster TM (2010) Weed Survey- Southern States. In: Southern Weed Science Society. pp 246–257
Wu T, Chellemi DO, Graham JH et al (2008) Comparison of soil bacterial communities under diverse agricultural land management and crop production practices. Microb Ecol 55:293–310. https://doi.org/10.1007/s00248-007-9276-4
Yu J, Baggio JS, Boyd NS et al (2020) Evaluation of ethanedinitrile (EDN) as a preplant soil fumigant in Florida strawberry production. Pest Manag Sci 76:1134–1141. https://doi.org/10.1002/ps.5626
Zasada IA, Halbrendt JM, Kokalis-Burelle N et al (2010) Managing nematodes without methyl bromide. Annu Rev Phytopathol 48:311–328. https://doi.org/10.1146/annurev-phyto-073009-114425
Zhang W Jr, Becker MEM JO, et al (1998) Effect of soil physical factors on methyl iodide and methyl bromide. Pestic Sci 53:71–79
Zhang WM, McGiffen ME, Becker JO et al (1997) Dose response of weeds to methyl iodide and methyl bromide. Weed Res 37:181–189. https://doi.org/10.1046/j.1365-3180.1997.d01-15.x
Zhou W, Du J, Li W et al (2019) Evaluation of the disappearance of cyanogen and hydrogen cyanide in different soil types using gas chromatography–mass spectrometry. Microchem J 151. https://doi.org/10.1016/j.microc.2019.104253
Zveibil A, Mor N, Gnayem N, Freeman S (2012) Survival, host-pathogen interaction, and management of Macrophomina phaseolina on strawberry in Israel. Plant Dis 96:265–272. https://doi.org/10.1094/PDIS-04-11-0299
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the following financial interests/personal relationships which may be considered as potential competing interests: Swaminathan Thalavaisundaram and Mary Stevens are employees of Draslovka Services. Husein Ajwa has no conflicts of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thalavaiasundaram, S., Ajwa, H. & Stevens, M.C. Dose-response of pests to ethanedinitrile dose-response of weed seeds, soil borne pathogens, and plant-parasitic nematodes to ethanedinitrile. Australasian Plant Pathol. 52, 133–143 (2023). https://doi.org/10.1007/s13313-023-00905-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-023-00905-z