Abstract
Objective
To evaluate the usefulness of procalcitonin (PCT) as a point-of-care testing to screen for radiographic pneumonia among children with influenza-like illness (ILI) and prolonged fever.
Methods
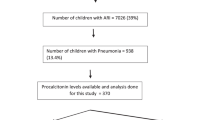
A prospective cohort study conducted at the pediatric emergency department of a tertiary hospital. Point-of-care testing for PCT was determined for 185 children aged 3 months to < 18 years with ILI and fever lasting > 4 days seen during the flu season in 2020. A chest radiograph (CXR) was performed for patients with PCT > 0.5 ng/mL.
Results
PCT value was > 0.5 ng/mL in 46 (24.9%) patients; a CXR was ordered in all cases except one and 14 (31.1%) of them had radiographic pneumonia (all had a PCT value > 0.7 ng/mL). Among the 139 (75.1%) patients with a PCT value ≤ 0.5 ng/mL, 137 (98.6%) were managed in the outpatient with symptomatic treatment; the remaining two cases warranted a CXR which was unremarkable in both. At evolution, no radiographic pneumonia was diagnosted in any of them.
Conclusion
PCT is a useful tool for point-of-care testing in patients with ILI and fever > 4 days to guide the indication for CXR to rule out radiographic pneumonia and helps in avoiding unnecessary radiation exposure.
Similar content being viewed by others
References
Goldman RD. Pediatric influenza in the emergency department: diagnosis and management. Pediatr Emerg Med Pract. 2021;18:1–20.
Lahti E, Peltola V, Virkki R, Ruuskanen O. Influenza pneumonia. Pediatr Infect Dis J. 2006;25:160–4.
Guirado Rivas C, Castrillón Cabaleiro I, Trenchs Sainz de la Maza V, Hernández-Bou S, Luaces Cubells C. Cost-effectiveness of chest radiographs in children with influenza-like illness and prolonged fever. Emerg Pediatr. 2022;1:69–72.
Shah SN, Bachur RG, Simel DL, Neuman MI. Does this child have pneumonia? the rational clinical examination systematic review. JAMA. 2017;318:462–71.
Rambaud-Althaus C, Althaus F, Genton B, Dacremont V. Clinical features for diagnosis of pneumonia in children younger than 5 years: a systematic review and meta-analysis. Lancet Infect Dis. 2015;15:439–50.
Rees CA, Basnet S, Gentile A, et al; World Health Organization PREPARE study group. An analysis of clinical predictive values for radiographic pneumonia in children. BMJ Glob Health. 2020;5:e002708.
Toikka P, Irjala K, Juvén T, et al. Serum procalcitonin, C-reactive protein and interleukin-6 for distinguishing bacterial and viral pneumonia in children. Pediatr Infect Dis J. 2000;19:598–602.
Moulin F, Raymond J, Lorrot M, et al. Procalcitonin in children admitted to hospital with community-acquired pneumonia. Arch Dis Child. 2001;84:332–6.
Korppi M, Remes S, Heiskanen-Kosma T. Serum procalcitonin concentrations in bacterial pneumonia in children: a negative result in primary healthcare settings. Pediatr Pulmonol. 2003;35:56–61.
Flood RG, Badik J, Aronoff SC. The utility of serum C-reactive protein in differentiating bacterial from non-bacterial pneumonia in children: a meta-analysis of 1230 children. Pediatr Infect Dis J. 2008;27:95–9.
Esposito S, Tagliabue C, Picciolli I, et al. Procalcitonin measurements for guiding antibiotic treatment in pediatric pneumonia. Respir Med. 2011;105:1939–45.
Hoshina T, Nanishi E, Kanno S, Nishio H, Kusuhara K, Hara T. The utility of biomarkers in differentiating bacterial from non-bacterial lower respiratory tract infection in hospitalized children: difference of the diagnostic performance between acute pneumonia and bronchitis. J Infect Chemother. 2014;20:616–20.
Banerjee R. Procalcitonin does not have clinical utility in children with community-acquired pneumonia. JAC Anti-microb Resist. 2021; 3:dlab152.
Khan DA, Rahman A, Khan FA. Is Procalcitonin Better than C-Reactive Protein for Early Diagnosis of Bacterial Pneumonia in Children? J Clin Lab Analn. 2010;24:1–5.
Tsou PY, Rafael J, Ma YK, et al. Diagnostic accuracy of procalcitonin for bacterial pneumonia in children - a syste matic review and meta-analysis. Infect Dis (Lond). 2020; 52:683–97.
Stockmann C, Ampofo K, Killpack J, et al. Procalcitonin accurately identifies hospitalized children with low risk of bacterial community-acquired pneumonia. J Ped Infect Dis Soc. 2018;7:46–53.
Nascimento-Carvalho CM, Cardoso MR, Barral A, et al. Procalcitonin is useful in identifying bacteraemia among children with pneumonia. Scand J Infect Dis. 2010;42: 644–9.
Don M, Valent F, Korppi M, et al. Efficacy of serum procalcitonin in evaluating severity of community-acquired pneumonia in childhood. Scand J Infect Dis. 2007; 39: 129–37.
Marès J, Rodrigo C, Moreno-Pérez D, et al. Recommendations for the management of influenza in pediatrics (2009–2010). An Pediatr (Engl Ed). 2010;72:144.e1–e12
Dieckmann RA, Brownstein D, Gausche-Hill M. The pediatric assessment triangle: a novel approach for the rapid evaluation of children. Pediatr Emerg Care. 2010;26:312–5.
Nelson KA, Morrow C, Wingerter SL, Bachur RG, Neuman MI. Impact of chest radiography on antibiotic treatment for children with suspected pneumonia. Pediatr Emerg Care. 2016;32:514–9.
Lipsett SC, Hirsch AW, Monuteaux MC, Bachur RG, Neuman MI. Development of the novel Pneumonia Risk Score to predict radiographic pneumonia in children. Pediatr Infect Dis J. 2022;41:24–30.
Klein EJ, Koenig M, Diekema DS, Winters W. Discordant radiograph interpretation between emergency physicians and radiologists in a pediatric emergency department. Pediatr Emerg Care. 1999;15:245–8.
Williams GJ, Macaskill P, Kerr M,et al. Variability and accuracy in interpretation of consolidation on chest radiography for diagnosing pneumonia in children under 5 years of age. Pediatr Pulmonol. 2013;48:1195–200.
Lipshaw MJ, Florin TA, Krueger S, et al. Factors associated with antibiotic prescribing and outcomes for pediatric pneumonia in the emergency department. Pediatr Emerg Care. 2021;37:e1033–e1038.
Funding
Funding: This study was partly supported by Brahms Diagnostica, GmbH, Berlin, Germany, who provided the Thermo Scientific™ B·R·A·H·M·S PCT™ direct test instrument and kits for the study. The study sponsor played no role in the study design, data collection, data analysis, or interpretation of results; writing of the report, or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
Contributors: SHB: Conceptualized and designed the study, analyzed, and interpreted data, and drafted the initial manuscript; VT: Conceptualized and designed the study, was involved in obtaining ethics approval, analyzed, and interpreted data and revised the manuscript; CG, IC: Conceptualized the study, enrolled patients and did data collection; CL: Conceptualized the study and provided critical review of the original and subsequent manuscript drafts. All authors approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Ethics Clearance: The Sant Joan de Déu Research Foundation Ethics Committee; PIC-166-18, dated Jan 24, 2019.
Competing interests: None stated.
Rights and permissions
About this article
Cite this article
Hernández-Bou, S., Trenchs, V., Guirado, C. et al. Procalcitonin as Point-of-Care Testing Modality for the Diagnosis of Pneumonia in Children With Influenza-like Illness. Indian Pediatr 61, 57–61 (2024). https://doi.org/10.1007/s13312-024-3089-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-024-3089-0