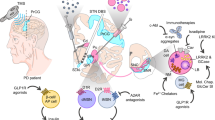
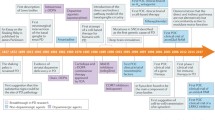
Abstract
Parkinson disease is an inexorably progressive neurodegenerative disorder. Multiple attempts have been made to establish therapies for Parkinson disease which provide neuroprotection or disease modification—two related, but not identical, concepts. However, to date, none of these attempts have succeeded. Many challenges exist in this field of research, including a complex multisystem disorder that includes dopaminergic and non-dopaminergic features; poorly understood and clearly multifaceted disease pathogenic mechanisms; a lack of reliable animal models; an absence of effective biomarkers of disease state, progression, and target engagement; and the confounding effects of potent symptomatic therapy. In this article, we will review previous, ongoing, and potential future trials designed to alter the progressive course of the disease from the perspective of the targeted underlying pathogenic mechanisms.

Similar content being viewed by others
References
Lang AE. Clinical trials of disease-modifying therapies for neurodegenerative diseases: the challenges and the future. Nat Med 2010;16:1223–1226.
Yacoubian TA, Standaert DG. Biochim Biophys Acta 2009;1792:676–687.
Alam ZI, Jenner A, Daniel SE, et al. Oxidative DNA damage in the parkinsonian brain: an apparent selective increase in 8-hydroxyguanine levels in substantia nigra. J Neurochem 1997;69:1196–1203.
Dexter DT, Sian J, Rose S, et al. Indices of oxidative stress and mitochondrial function in individuals with incidental Lewy body disease. Ann Neurol 1994;35:38–44.
Hastings TG, Lewis DA, Zigmond MJ. Reactive dopamine metabolites and neurotoxicity: implications for Parkinson’s disease. Adv Exp Med Biol 1996;387:97–106.
The Parkinson Study Group. Effect of deprenyl on the progression of disability in early Parkinson’s disease. N Engl J Med 1989;321:1364–1371.
Olanow CW, Hauser RA, Gauger L, et al. The effect of deprenyl and levodopa on the progression of signs and symptoms in Parkinson’s disease. Ann Neurol 1995;38:771–777.
Pålhagen S, Heinonen E, Hägglund J, et al. Selegiline slows the progression of the symptoms of Parkinson disease. Neurology 2006;66:1200–1206.
Bar-Am O, Weinreb O, Amit T, Youdim MB. The neuroprotective mechanism of 1-(R)-aminoindan, the major metabolite of the anti-parkinsonian drug rasagiline. J Neurochem 2010;112:1131–1137.
Parkinson Study Group. A controlled, randomized, delayed-start study of rasagiline in early Parkinson disease. Arch Neurol 2004;61:561–566.
Olanow CW, Rascol O, Hauser R, et al. A double-blind, delayed start trial of rasagiline in Parkinson’s disease. N Engl J Med 2009;361:1268–1278.
Rascol O, Fitzer-Attas CJ, Hauser R et al. A double-blind, delayed-start trial of rasagiline in Parkinson’s disease (the ADAGIO study): prespecified and post-hoc analyses of the need for additional therapies, changes in UPDRS scores, and non-motor outcomes. Lancet Neurol 2011;10:415–423.
Jenner P, Langston JW. Explaining ADAGIO: a critical review of the biological basis for the clinical effects of rasagiline. Mov Disord 2011;26:2316–2323.
Deas E, Wood NW, Plun-Favreau H. Mitophagy and Parkinson’s disease: the PINK1-parkin link. Biochim Biophys Acta 2011;1813:623–633.
Schapira AH, Cooper JM, Dexter D, Jenner P, Clark JB, Marsden CD. Mitochondrial complex I deficiency in Parkinson’s disease. Lancet 1989;1:1269.
Schapira AHV, Cooper JM, Dexter D, et al. Mitochondrial complex I deficiency in Parkinson’s disease. J Neurochem 1990;54:823–827.
Mizuno Y, Ohta S, Tanaka M, et al. Deficiencies in complex I subunits of the respiratory chain in Parkinson’s disease. Biochem Biophys Res Commun 1989;163:1450–1455.
Beal MF. Bioenergetic approaches for neuroprotection in Parkinson’s disease. Ann Neurol 2003;53:S39-S47.
Shults CW, Oakes D, Kieburtz K, et al. Effects of coenzyme Q10 in early Parkinson disease: evidence of slowing of the functional decline. Arch Neurol 2002;59:1541–1550.
Effects of Coenzyme Q10 (CoQ) in Parkinson Disease (QE3). Clinicaltrials.gov. Available at: www.http://clinicaltrials.gov/ct2/show/NCT00740714. Accessed August 16, 2013.
Snow BJ, Rolfe FL, Lockhart MM, et al. A double-blind, placebo controlled study to assess the mitochondria-targeted antioxidant MitoQ as a disease- modifying therapy in Parkinson’s disease. Mov Disord 2010;25:1670–1674.
Matthews RT, Ferrante RJ, Klivenyi P, et al. Creatine and cyclocreatine attenuate MPTP neurotoxicity. Exp Neurol 1999;157:142–149.
Bender A, Koch W, Elstner M, et al. Creatine supplementation in Parkinson disease: a placebo controlled randomized pilot trial. Neurology 2006;67:1262–1264.
NINDS NET-PD Investigators. A randomized, double-blind, futility clinical trial of creatine and minocycline in early Parkinson disease. Neurology 2006;66:664–671.
Elm JJ; NINDS NET-PD Investigators. Design innovations and baseline findings in a long-term Parkinson’s trial: the National Institute of Neurological Disorders and Stroke Exploratory Trials in Parkinson’s Disease Long-Term Study-1. Mov Disord 2012;27:1513–1521.
Olanow CW, Jenner P, Brooks D. Dopamine agonists and neuroprotection in Parkinson’s disease. Ann Neurol 1998;44:167–174.
Whone A, Watts R, Stoessl J, et al. Slower progression of Parkinson’s disease with ropinirole versus levodopa: the REAL-PET study: Ann Neurol 2003;54:93–101.
Parkinson Study Group. Dopamine transporter brain imaging to assess the effects of Pramipexole vs levodopa Parkinson disease progression. JAMA 2002;287:1653–1661.
Ravina B, Eidelberg D, Ahlskog JE, et al. The role of radiotracer imaging in Parkinson disease. Neurology 2005;64:208–215.
Olanow CW, Agid Y, Mizuno Y, et al. Levodopa in the treatment of Parkinson’s disease: current controversies. Mov Disord 2004;19:997–1005.
Fahn S. Is levodopa toxic? Neurology 1996;47:S184-195.
Barzilai A, Melamed E, Shirvan A. Is there a rationale for neuroprotection against dopamine toxicity in Parkinson’s disease? Cell Mol Neurobiol 2001;21:215–235.
Murer MG, Dziewczapolski G, Menalled LB et al. Chronic levodopa is not toxic for remaining dopamine neurons, but instead promotes their recovery, in rats with moderate nigrostriatal lesions. Ann Neurol 1998;43:561–575.
Datla KP, Blunt SB, Dexter DT. Chronic L-DOPA administration is not toxic to the remaining dopaminergic nigrostriatal neurons, but instead may promote their functional recovery, in rats with partial 6-OHDA or FeCl(3) nigrostriatal lesions. Mov Disord 2001;16:424–434.
Fahn S, Oakes D, Shoulson I, et al. Levodopa and the progression of Parkinson’s disease. N Engl J Med 2004;351:2498–2508.
Olanow CW, Jankovic J. Neuroprotective therapy in Parkinson’s disease and motor complications: a search for a pathogenesis-targeted, disease-modifying strategy. Mov Disord 2005;20:S3-10.
Schapira AH, McDermott MP, Barone P, et al. Pramipexole in patients with early Parkinson’s disease (PROUD): a randomised delayed-start trial. Lancet Neurol 2013;12:747–755.
Howells DW, Porritt MJ, Wong JY, et al. Reduced BDNF mRNA expression in the Parkinson’s disease substantia nigra. Exp Neurol 2000;166:127–135.
Chauhan NB, Siegel GJ, Lee JM. Depletion of glial cell line-derived neurotrophic factor in substantia nigra neurons of Parkinson’s disease brain. J Chem Neuroanat 2001;21:277–288.
Mogi M, Togari A, Kondo T, et al. Brain-derived growth factor and nerve growth factor concentrations are decreased in the substantia nigra in Parkinson’s disease. Neurosci Lett 1999;270:45–48.
Cheng FC, Ni DR, Wu MC, Kuo JS, Chia LG. Glial cell line-derived neurotrophic factor protects against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced neurotoxicity in C57BL/6 mice. Neurosci Lett 1998;252:87–90.
Fox CM, Gash DM, Smoot MK, Cass WA. Neuroprotective effects of GDNF against 6-OHDA in young and aged rats. Brain Res 2001;896:56–63.
Decressac M, Kadkhodaei B, Mattsson B, Laguna A, Perlmann T, Björklund A. α-Synuclein-induced down-regulation of Nurr1 disrupts GDNF signaling in nigral dopamine neurons. Sci Transl Med 2012;4:156–163.
Gill SS, Patel NK, Hotton GR, et al. Direct brain infusion of glial cell line-derived neurotrophic factor in Parkinson disease. Nat Med 2003;9:589–595.
Lang AE, Gill S, Patel NK, et al. Randomized controlled trial of intraputamenal glial cell line-derived neurotrophic factor infusion in Parkinson disease. Ann Neurol 2006;59:459–466.
AAV2-GDNF for Advanced Parkinson’s Disease. Clinicaltrials.gov. Available at: www.http://clinicaltrials.gov/ct2/show/NCT01621581. Accessed August 16, 2013.
Marks WJ Jr, Bartus RT, Siffert J, et al. Gene delivery of AAV2-neurturin for Parkinson’s disease: a double-blind, randomised, controlled trial. Lancet Neurol 2010;9:1164–1172.
Investigation of Cogane (PYM50028) in Early-stage Parkinson’s Disease (CONFIDENT-PD). Clinicaltrials.gov. Available at: www.http://clinicaltrials.gov/ct2/show/NCT01060878. Accessed August 16, 2013.
Steiner JP, Connolly MA, Valentine HL, et al. Neurotrophic actions of nonimmunosuppressive analogues of immunosuppressive drugs FK506, rapamycin and cyclosporin A. Nat Med 1997;3:421–428.
Khan Z, Ferrari G, Kasper M, et al. The nonimmunosuppressive immunophilin ligand GPI-1046 potently stimulates regenerating axon growth from adult mouse dorsal root ganglia cultured in Matrigel. Neuroscience 2002;114:601–609.
Zhang C, Steiner JP, Hamilton GS, Hicks TP, Poulter MO. Regeneration of dopaminergic function in 6-hydroxydopamine-lesioned rats by neuroimmunophilin ligand treatment. J Neurosci 2001;21:RC156.
Tanaka K, Yoshioka M, Miyazaki I, Fujita N, Ogawa N. GPI1046 prevents dopaminergic dysfunction by activating glutathione system in the mouse striatum. Neurosci Lett 2002;321:45–48.
Tanaka K, Fujita N, Ogawa N. Immunosuppressive (FK506) and non-immunosuppressive (GPI1046) immunophilin ligands activate neurotrophic factors in the mouse brain. Brain Res 2003;970:250–253.
Poulter MO, Payne KB, Steiner JP. Neuroimmunophilins: a novel drug therapy for the reversal of neurodegenerative disease? Neuroscience 2004;128:1–6.
Mody I, MacDonald JF. NMDA receptor-dependent excitotoxicity: the role of intracellular Ca2+ release. Trends Pharmacol Sci 1995;16:356–359.
Jankovic J, Hunter C. A double-blind, placebo-controlled and longitudinal study of riluzole in early Parkinson’s disease. Parkinsonism Relat Disord 2002;8:271–276.
Rascol O, Olanow W, Brooks D, Koch G, Truffinet P, Bejuit R. A 2-year multicenter, placebo-controlled, double-blind, parallel group study of the effect of riluzole on Parkinson’s disease progression. Mov Disord 2002;17 (Suppl. 5):A80.
Parkinson Study Group PRECEPT Investigators. Mixed lineage kinase inhibitor CEP-1347 fails to delay disability in early Parkinson disease. Neurology 2007;69:1480–1490.
Olanow CW, Schapira AH, LeWitt PA, et al. TCH346 as a neuroprotective drug in Parkinson’s disease: a double-blind, randomised, controlled trial. Lancet Neurol 2006;5:1013–1020.
Tikka T, Fiebich BL, Goldsteins G, Keinanen R, Koistinaho J. Minocycline, a tetracycline derivative, is neuroprotective against excitotoxicity by inhibiting activation and proliferation of microglia. J Neurosci 2001;21:2580–2588.
Tikka TM, Koistinaho JE. Minocycline provides neuroprotection against N-methyl-D-aspartate neurotoxicity by inhibiting microglia. J Immunol 2001;166:7527–7533.
Du Y, Ma Z, Lin S, et al. Minocycline prevents nigrostriatal dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease. Proc Natl Acad Sci U S A 2001;98:14669–14674.
He Y, Appel S, Le W. Minocycline inhibits microglial activation and protects nigral cells after 6-hydroxydopamine injection into mouse striatum. Brain Res 2001;909:187–193.
Schneider JS, Gollomp SM, Sendek S, Colcher A, Cambi F, Du W. A randomized, controlled, delayed start trial of GM1 ganglioside in treated Parkinson’s disease patients. J Neurol Sci 2013;324:140–148.
Bezard E, Yue Z, Kirik D, Spillantini MG. Animal models of Parkinson’s disease: limits and relevance to neuroprotection studies. Mov Disord 2013;57:61–70.
Olanow CW, Kordower J. Modeling Parkinson’s disease. Ann Neurol 2009;66:432–436.
Dawson TM, Ko HS, Dawson VL. Genetic animal models of Parkinson’s disease. Neuron 2010;66:646–661.
Stocchi F, Olanow CW. Obstacles to the development of a neuroprotective therapy for Parkinson’s disease. Mov Disord 2013;28:3–7.
Luk KC, Kehm V, Carroll J, Zhang B, O’Brien P, Trojanowski JQ, Lee VM. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 2012;338:949–953.
Kieburtz K. Issues in neuroprotection clinical trials in Parkinson’s disease. Neurology 2006;66:S50-57.
Lang AE, Melamed E, Poewe W, Rascol O. Trial designs used to study neuroprotective therapy in Parkinson’s disease. Mov Disord 2013;28:86–95.
The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. Mov Disord 2003;18:738–750.
Goetz CG, Tilley BC, Shaftman SR, et al., for the Movement Disorder Society UPDRS Revision Task Force. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;23:2129–2170.
Lang AE, Obeso JA. Time to move beyond nigrostriatal dopamine deficiency in Parkinson’s disease. Ann Neurol 2004;55:761–765.
Shi M, Bradner J, Hancock AM, et al. Cerebrospinal fluid biomarkers for Parkinson disease diagnosis and progression. Ann Neurol 2011;69:570–580.
Rentzos M, Nikolaou C, Andreadou E, et al. Circulating interleukin-10 and interleukin-12 in Parkinson’s disease. Acta Neurol Scand 2009;119:332–337.
Agarwal PA, Stoessl AJ. Biomarkers for Trials of Neuroprotection in Parkinson’s Disease. Mov Disord 2013;28:71–85.
Dragalin V. An introduction to adaptive designs and adaptation in CNS trials. Eur Neuropsychopharmacol. 2011;21:153–158.
Michell AW, Lewis SJ, Foltynie T, Barker RA. Biomarkers and Parkinson’s disease. Brain 2004;127:1693–1705.
Dexter DT, Wells FR, Lees AJ, et al. Increased nigral iron content and alterations in other metal ions occurring in brain in Parkinson’s disease. J Neurochem 1989;52:1830–1836.
Efficacy and Safety of the Iron Chelator Deferiprone in Parkinson’s Disease (FAIR-PARK-I). Clinicaltrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT00943748. Accessed August 16, 2013.
Ames BN, Cathcart R, Schwiers E, Hochstein P. Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci U S A 1981;78:6858–6862.
de Lau LM, Koudstaal PJ, Hofman A, Breteler MM. Serum uric acid levels and the risk of Parkinson disease. Ann Neurol 2005;58:797–800.
Weisskopf MG, O’Reilly E, Chen H, Schwarzschild MA, Ascherio A. Plasma urate and risk of Parkinson’s disease. Am J Epidemiol 2007;166:561–567.
Alonso A, Rodriguez LA, Logroscino G, Hernan MA. Gout and risk of Parkinson disease: a prospective study. Neurology 2007;69:1696–1700.
Schwarzschild MA, Schwid SR, Marek K, et al. Serum urate as a predictor of clinical and radiographic progression in Parkinson disease. Arch Neurol 2008;65:716–723.
Gao X, Chen H, Choi HK, Curhan G, Schwarzschild MA, Ascherio A. Diet, urate, and Parkinson’s disease risk in men. Am J Epidemiol 2008;167:831–838.
Safety of Urate Elevation in Parkinson’s Disease (SURE-PD). ClinicalTrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT00833690. Accessed August 16, 2013.
Yürekli VA, Gürler S, Nazıroğlu M, Uğuz AC, Koyuncuoğlu HR. Zonisamide attenuates MPP+−induced oxidative toxicity through modulation of Ca2+ signaling and caspase-3 activity in neuronal PC12 cells. Cell Mol Neurobiol 2013;33:205–212.
Choudhury ME, Moritoyo T, Kubo M, et al. Zonisamide-induced long-lasting recovery of dopaminergic neurons from MPTP-toxicity. Brain Res 2011;1384:170–178.
Study of Zonisamide in Early Parkinson Disease (ZONIST). ClinicalTrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT01766128. Accessed August 16, 2013.
Hauser RA, Lyons KE, McClain T, et al. Randomized, doubleblind, pilot evaluation of intravenous glutathione in Parkinson’s disease. Mov Disord 2009;24:979–983.
Okun MS, Lang A, Jankovic J. Reply: Based on the available randomized trial patients should say no to glutathione for Parkinson’s disease. Mov Disord. 2010;25:961–962.
Intranasal Glutathione in Parkinson’s Disease (inGSH in PD). ClinicalTrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT01398748. Accessed August 16, 2013.
N-Acetylcysteine for Neuroprotection in Parkinson’s Disease (NAC for PD). ClinicalTrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT01470027. Accessed August 16, 2013.
Guo S, Yan J, Yang T, Yang X, Bezard E, Zhao B. Protective effects of green tea polyphenols in the 6-OHDA rat model of Parkinson’s disease through inhibition of ROS-NO pathway. Biol Psychiatry 2007;62:1353–1362.
Efficacy and Safety of Green Tea Polyphenol in De Novo Parkinson’s Disease Patients. ClinicalTrials.gov. Available at: http://www.clinicaltrials.gov/ct2/show/NCT00461942. Accessed August 16, 2013.
Ascherio A, Zhang SM, Hernan MA, et al. Prospective study of caffeine consumption and risk of Parkinson’s disease in men and women. Ann Neurol 2001;50:56–63.
Palacios N, Gao X, McCullough ML, et al. Caffeine and risk of Parkinson’s disease in a large cohort of men and women. Mov Disord 2012;27:1276–82.
Schwarzschild MA, Agnati L, Fuxe K, Chen JF, Morelli M. Targeting adenosine A2A receptors in Parkinson’s disease. Trends Neurosci 2006;29:647–654.
Kalia LV, Brotchie JM, Fox SH. Novel nondopaminergic targets for motor features of Parkinson’s disease: review of recent trials. Mov Disord 2013;28:131–144.
A Placebo- and Active-Controlled Study of Preladenant in Early Parkinson’s Disease (P05664 AM5). ClinicalTrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT01155479. Accessed August 16, 2013.
Tansey MG, McCoy MK, Frank-Cannon TC. Neuroinflammatory mechanisms in Parkinson’s disease: potential environmental triggers, pathways, and targets for early therapeutic intervention. Exp Neurol 2007;208:1–25.
McGeer EG, McGeer PL. The role of anti-inflammatory agents in Parkinson’s disease. CNS Drugs 2007;21:789–797.
McGeer PL, Itagaki S, Boyes BE, McGeer EG. Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 1988;38:1285–1291.
McGeer PL, Schwab C, Parent A, Doudet D. Presence of reactive microglia in monkey substantia nigra years after 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine administration. Ann Neurol 2003;54:599–604.
Sherer TB, Betarbet R, Kim JH, Greenamyre JT. Selective microglial activation in the rat rotenone model of Parkinson’s disease. Neurosci Lett 2003;341:87–90.
Mogi M, Harada M, Kondo T, et al. Interleukin-1 beta, interleukin-6, epidermal growth factor and transforming growth factor-alpha are elevated in the brain from parkinsonian patients. Neurosci Lett 1994;180:147–150.
Goldknopf IL, Sheta EA, Bryson J, et al. Complement C3c and related protein biomarkers in amyotrophic lateral sclerosis and Parkinson’s disease. Biochem Biophys Res Commun 2006;342:1034–1039.
Esposito E, Di Matteo V, Benigno A, Pierucci M, Crescimanno G, Di Giovanni G. Non-steroidal anti-inflammatory drugs in Parkinson’s disease. Exp Neurol 2007;205:295–312.
Chen H, Zhang SM, Hernan MA, et al. Nonsteroidal anti-inflammatory drugs and the risk of Parkinson disease. Arch Neurol 2003;60:1059–1064.
Chen H, Jacobs E, Schwarzschild MA, et al. Nonsteroidal antiinflammatory drug use and the risk for Parkinson’s disease. Ann Neurol 2005;58:963–967.
Bower JH, Maraganore DM, Peterson BJ, Ahlskog JE, Rocca WA. Immunologic diseases, anti-inflammatory drugs, and Parkinson disease: a case–control study. Neurology 2006;67:494–496.
Hernan MA, Logroscino G, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and the incidence of Parkinson disease. Neurology 2006;66:1097–1099.
Selley ML. Simvastatin prevents 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced striatal dopamine depletion and protein tyrosine nitration in mice. Brain Res 2005;1037:1–6.
Di Napoli P, Taccardi AA, Oliver M, De Caterina R. Statins and stroke: evidence for cholesterol independent effects. Eur Heart J 2002;23:1908–1921.
Wolozin B, Wang SW, Li NC, Lee A, Lee TA, Kazis LE. Simvastatin is associated with a reduced incidence of dementia and Parkinson’s disease. BMC Med 2007;5:20.
Wahner AD, Bronstein JM, Bordelon YM, Ritz B. Statin use and the risk of Parkinson disease. Neurology 2008;70:1418–1422.
de Lau LM, Koudstaal PJ, Hofman A, Breteler MM. Serum cholesterol levels and the risk of Parkinson’s disease. Am J Epidemiol 2006;164:998–1002.
Huang X, Chen H, Miller WC, et al. Lower low-density lipoprotein cholesterol levels are associated with Parkinson’s disease. Mov Disord 2007;22:377–381.
Carta AR, Pisanu A. Modulating microglia activity with PPAR-γ agonists: a promising therapy for Parkinson’s disease? Neurotox Res 2013;23:112–123.
Pioglitazone in Early Parkinson’s Disease. ClinicalTrials.gov. Available at: http://www.clinicaltrials.gov/ct2/show/NCT01280123. Accessed August 16, 2013.
Chan CS, Gertler TS, Surmeier DJ. A molecular basis for the increased vulnerability of substantia nigra dopamine neurons in aging and Parkinson’s disease. Mov Disord 2010;25:S63-70.
Pasternak B, Svanström H, Nielsen NM, Fugger L, Melbye M, Hviid A. Use of calcium channel blockers and Parkinson’s disease. Am J Epidemiol 2012;175:627–635.
Marras C, Gruneir A, Rochon P, et al. Dihydropyridine calcium channel blockers and the progression of parkinsonism. Ann Neurol 2012;71:362–369.
Simuni T, Borushko E, Avram MJ, et al. Tolerability of isradipine in early Parkinson’s disease: a pilot dose escalation study. Mov Disord 2010;25:2863–2866.
Lashuel HA, Overk CR, Oueslati A, Masliah E. The many faces of α-synuclein: from structure and toxicity to therapeutic target. Nat Rev Neurosci 2013;14:38–48.
Visanji NP, Brooks PL, Hazrati LN, Lang AE. The prion hypothesis in Parkinson’s disease: Braak to the future. Acta Neuropathol Comm 2013;1:2.
Kalia LV, Kalia SK, McLean PJ, Lozano AM, Lang AE. α-Synuclein oligomers and clinical implications for Parkinson disease. Ann Neurol 2013;73:155–169.
Tolerability and Safety of Subcutaneous Administration of Two Doses of AFFITOPE® PD01A in Early Parkinson’s Disease. Clinicaltrials.gov. Available at: http://clinicaltrials.gov/ct2/show/NCT01568099. Accessed August 16, 2013.
Bodles AM, El-Agnaf OM, Greer B, Guthrie DJ, Irvine GB. Inhibition of fibril formation and toxicity of a fragment of alpha-synuclein by an N-methylated peptide analogue. Neurosci Lett 2004;359:89–93.
Amer DA, Irvine GB, El-Agnaf OM. Inhibitors of alpha-synuclein oligomerization and toxicity: a future therapeutic strategy for Parkinson’s disease and related disorders. Exp Brain Res 2006;173:223–233.
Kalia SK, Kalia LV, McLean PJ. Molecular chaperones as rational drug targets for Parkinson’s disease therapeutics. CNS Neurol Disord Drug Targets 2010;9:741–753.
Qiao L, Hamamichi S, Caldwell KA, et al. Lysosomal enzyme cathepsin D protects against alpha-synuclein aggregation and toxicity. Mol Brain 2008;1:17.
Yu D, Pendergraff H, Liu J, et al. Single-stranded RNAs use RNAi to potently and allele-selectively inhibit mutant huntingtin expression. Cell 2012;150:895–908.
Gilks WP, Abou-Sleiman PM, Gandhi S, et al. A common LRRK2 mutation in idiopathic Parkinson’s disease. Lancet 2005;365:415–416.
Healy DG, Falchi M, O’Sullivan SS, et al. Phenotype, genotype, and worldwide genetic penetrance of LRRK2-associated Parkinson’s disease: a case–control study. Lancet Neurol 2008;7:583–590.
Lesage S, Ibanez P, Lohmann E, et al. G2019S LRRK2 mutation in French and North African families with Parkinson’s disease. Ann Neurol 2005;58:784–787.
Ozelius LJ, Senthil G, Saunders-Pullman R, et al. LRRK2 G2019S as a cause of Parkinson’s disease in Ashkenazi Jews. N Engl J Med 2006;354:424–425.
West AB, Moore DJ, Biskup S, et al. Parkinson’s disease-associated mutations in leucine-rich repeat kinase 2 augment kinase activity. Proc Natl Acad Sci U S A 2005;102:16842–16847.
West AB, Moore DJ, Choi C, et al. Parkinson’s disease-associated mutations in LRRK2 link enhanced GTP-binding and kinase activities to neuronal toxicity. Hum Mol Genet 2007;16:223–232.
Ramsden N, Perrin J, Ren Z, et al. Chemoproteomics-based design of potent LRRK2-selective lead compounds that attenuate Parkinson’s disease-related toxicity in human neurons. ACS Chem Biol 2011;6:1021–1028.
Liu Z, Hamamichi S, Lee BD, et al. Inhibitors of LRRK2 kinase attenuate neurodegeneration and Parkinson-like phenotypes in Caenorhabditis elegans and Drosophila Parkinson’s disease models. Hum Mol Genet 2011;20:3933–3942.
Aviles-Olmos I, Dickson J, Kefalopoulou Z, et al. Exenatide and the treatment of patients with Parkinson’s disease. J Clin Invest 2013 May 20 [Epub ahead of print].
Efficacy of Transdermal Nicotine, on Motor Symptoms in Advanced Parkinson’s Disease (NICOPARK2). Clinicaltrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT00873392. Accessed August 16, 2013.
Disease-modifying Potential of Transdermal NICotine in Early Parkinson’s Disease (NIC-PD). Clinicaltrials.gov. Available at: www.clinicaltrials.gov/ct2/show/ NCT01560754. Accessed August 16, 2013.
Xue YQ, Zhao LR, Guo WP, Duan WM. Intrastriatal administration of erythropoietin protects dopaminergic neurons and improves neurobehavioral outcome in a rat model of Parkinson’s disease. Neuroscience 2007;146:1245–1258.
Pedroso I, Bringas ML, Aguiar A, et al. Use of Cuban recombinant human erythropoietin in Parkinson’s disease treatment. MEDICC Rev 2012;14:11–17.
McCollum M, Ma Z, Cohen E, et al. Post-MPTP treatment with granulocyte colony-stimulating factor improves nigrostriatal function in the mouse model of Parkinson’s disease. Mol Neurobiol 2010;41:410–419.
Study of the Neuro-protective Effect of Granulocyte-colony Stimulating Factor on Early Stage Parkinson’s Disease. Clinicaltrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT01227681. Accessed August 16, 2013.
The ParkCycle Study. Clinicaltrials.gov. Available at: www.clinicaltrials.gov/ct2/show/ NCT01562496. Accessed August 16, 2013.
Pahwa R, Factor SA, Lyons KE, et al. Practice parameter: treatment of Parkinson disease with motor fluctuations and dyskinesia (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2006;66:983–995.
Fox SHKR, Lim SY, Ravina B, et al. The Movement Disorder Society evidence-based medicine review update: treatments for the motor symptoms of Parkinson’s disease. Mov Disord 2011;26:S2-S41.
Anderson VC, Burchiel KJ, Hogarth P, Favre J, Hammerstad JP. Pallidal vs subthalamic nucleus deep brain stimulation in Parkinson disease. Arch Neurol 2005;62:554–560.
Follett KA, Weaver FM, Stern M, et al. Pallidal versus subthalamic deep-brain stimulation for Parkinson’s disease. N Engl J Med 2010;362:2077–2091.
Lozano AM, Dostrovsky J, Chen R, Ashby P. Deep brain stimulation for Parkinson’s disease: disrupting the disruption. Lancet Neurol 2002;1:225–231.
Kita H., Kitai S.T. Efferent projections of the subthalamic nucleus in the rat: light and electron microscopic analysis with the PHA-L method. J. Comp. Neurol 1987;260:435–452.
Temel Y, Blokland A, Steinbusch HW, Visser-Vandewalle V. The functional role of the subthalamic nucleus in cognitive and limbic circuits. Prog Neurobiol 2005;76:393–413.
Rodriguez MC, Obeso JA, Olanow CW. Subthalamic nucleus-mediated excitotoxicity in Parkinson’s disease: a target for neuroprotection. Ann Neurol 1998;44:S175-S188.
Temela Y, Visser-Vandewalle V, Kapland S, et al. Protection of nigral cell death by bilateral subthalamic nucleus stimulation. Brain Res 2006;1120:100–105.
Deep Brain Stimulation (DBS) for Early Stage Parkinson’s Disease (PD). Clinicaltrials.gov. Available at: www.clinicaltrials.gov/ct2/show/NCT00282152. Accessed August 16, 2013.
LeWitt PA, Rezai AR, Leehey MA, et al. AAV2-GAD gene therapy for advanced Parkinson’s disease: a double-blind, sham-surgery controlled, randomised trial. Lancet Neurol 2011;10:309–319.
Park HJ, Lee PH, Bang OY, Lee G, Ahn YH. Mesenchymal stem cells therapy exerts neuroprotection in a progressive animal model of Parkinson’s disease. J Neurochem 2008;107:141–151.
Sadan O, Bahat-Stromza M, Barhum Y, et al. Protective effects of neurotrophic factor-secreting cells in a 6-OHDA rat model of Parkinson disease. Stem Cells Dev 2009;18:1179–1190.
Brundin P, Strecker RE, Lindvall O, et al. Intracerebral grafting of dopamine neurons. Experimental basis for clinical trials in patients with Parkinson’s disease. Ann N Y Acad Sci 1987; 495,473-496.
Nishimura K, Takahashi J. Therapeutic application of stem cell technology toward the treatment of Parkinson’s disease. Biol Pharm Bull 2013;36:171–175.
Mendez I, Dagher A, Hong M, et al. Simultaneous intrastriatal and intranigral fetal dopaminergic grafts in patients with Parkinson disease: a pilot study. Report of three cases. J Neurosurg 2002;96:589–596.
Gross RE, Watts RL, Hauser RA, et al. Intrastriatal transplantation of microcarrier-bound human retinal pigment epithelial cells versus sham surgery in patients with advanced Parkinson’s disease: a double-blind, randomised, controlled trial. Lancet Neurol 2011;10:509–519.
Freed CR, Greene PE, Breeze RE, et al. Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N Engl J Med 2001;344:710–719.
Olanow CW, Goetz CG, Kordower JH, et al. A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson’s disease. Ann Neurol 2003;54:403–414.
Kriks S, Shim JW, Piao J, et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature 2011;480:547–551.
Karussis D, Petrou P, Kassis I. Clinical experience with stem cells and other cell therapies in neurological diseases. J Neurol Sci 2013;324:1–9.
Lang AE, Obeso JA. Challenges in Parkinson’s disease: restoration of the nigrostriatal dopamine system is not enough. Lancet Neurol 2004;3:309–316.
Olanow CW, Kordower JH, Lang AE, Obeso JA. Dopaminergic transplantation for Parkinson’s disease: current status and future prospects. Ann Neurol 2009;66:591–596.
Tetrud JW, Langston JW. The effect of deprenyl (selegiline) on the natural history of Parkinson’s disease. Science 1989;245:519–522.
The Parkinson Study Group. Effects of tocopherol and deprenyl on the progression of disability in early Parkinson’s disease. N Engl J Med 1993;328:176–183.
Myllylä VV, Sotaniemi KA, Vuorinen JA, Heinonen EH. Selegiline as initial treatment in de novo parkinsonian patients. Neurology 1992;42:339–343.
Parkinson Study Group. Impact of deprenyl and tocopherol treatment on Parkinson’s disease in DATATOP subjects not requiring levodopa. Ann Neurol 1996;39:29–36.
Pålhagen S, Heinonen EH, Hägglund J, et al. Selegiline delays the onset of disability in de novo parkinsonian patients. Swedish Parkinson Study Group. Neurology 1998;51:520–525.
NINDS NET-PD Investigators. A randomized clinical trial of coenzyme Q(10) and GPI-1485 in early Parkinson disease. Neurology 2007;68:20–28.
Parkinson Study Group. Pramipexole vs levodopa as initial treatment for Parkinson disease: A randomized controlled trial. Parkinson Study Group. JAMA 2000;284:1931–1938.
The GPI 1485 Study Group. GPI 1485, a neuroimmunophilin ligand, fails to alter disease progression in mild to moderate Parkinson’s disease. Mov Disord 2006;21:A1009.
Ribeiro MJ, Stoessl AJ, Brooks D, et al. Evaluation of a potential neurotrophic drug on the progression of Parkinson disease with 18FDOPA. J Nucl Med 2009;50(Suppl. 2):A125.
Schneider JS, Sendek S, Daskalakis C, Cambi F. GM1 ganglioside in Parkinson’s disease: results of a five year open study. J Neurol Sci 2010;292:45–51.
Acknowledgments
LVK is supported by a Canadian Institutes of Health Research Clinician-Scientist Award. AEL holds the Jack Clark Chair in Parkinson’s Disease Research at the University of Toronto. The authors have no real or perceived conflicts of interest. Full conflict of interest disclosures are available in the electronic supplementary material for this article.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article.
Since the review and acceptance of this manuscript, the NINDS has stopped the NET‐PD long‐term study of creatine for treatment of early stage PD. According to the NET‐PD website, an interim analysis revealed evidence for futility. Continued follow‐up of subjects was not expected to demonstrate a statistically significant difference between creatine and placebo. Hence, the study’s Data Safety Monitoring Board recommended termination of the study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 511 kb)
Rights and permissions
About this article
Cite this article
AlDakheel, A., Kalia, L.V. & Lang, A.E. Pathogenesis-Targeted, Disease-Modifying Therapies in Parkinson Disease. Neurotherapeutics 11, 6–23 (2014). https://doi.org/10.1007/s13311-013-0218-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13311-013-0218-1