Abstract
Management of diverticular abscess (DA) is still controversial. Antibiotic therapy is indicated in abscesses ≤ 4 cm, while percutaneous drainage/surgery in abscesses > 4 cm. The study aims to assess the role of antibiotics and surgical treatments in patients affected by DA. We retrospectively analyzed 100 consecutive patients with DA between 2013 and 2020, with a minimum follow-up of 12 months. They were divided into two groups depending on abscess size ≤ or > 4 cm (group 1 and group 2, respectively). All patients were initially treated with intravenous antibiotics. Surgery was considered in patients with generalized peritonitis at admission or after the failure of antibiotic therapy. The primary endpoint was to compare recurrence rates for antibiotics and surgery. The secondary endpoint was to assess the failure rate of each antibiotic regimen resulting in surgery. In group 1, 31 (72.1%) patients were conservatively treated and 12 (27.9%) underwent surgery. In group 2, percentages were respectively 50.9% (29 patients) and 49.1% (28 patients). We observed 4 recurrences in group 1 and 6 in group 2. Recurrence required surgery in 3 patients/group. We administered amoxicillin-clavulanic acid to 74 patients, piperacillin-tazobactam to 14 patients and ciprofloxacin + metronidazole to 12 patients. All patients referred to surgery had been previously treated with amoxicillin-Powered by Editorial Manager® and ProduXion Manager® from Aries Systems Corporation clavulanic acid. No percutaneous drainage was performed in a hundred consecutive patients. Surgical treatment was associated with a lower risk of recurrence in patients with abscess > 4 cm, compared to antibiotics. Amoxicillin-clavulanic acid was associated with a higher therapeutic failure rate than piperacillin-tazobactam/ciprofloxacin + metronidazole.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diverticular abscess (DA) is the most frequent complication of acute colonic diverticulitis (AD). It is usually diagnosed in approximately 15–20% [1,2,3,4] of AD cases, mostly using contrast-enhanced-computed-tomography scan (CT-scan) [5]. DAs are classified according to location and size and the most widely used classifications for staging are the best-known Hinchey’s [6] and the most recent one from the World Society of Emergency Surgery (WSES) [7]. According to Hinchey’s modified classification, DA can be confined in pericolic fat, smaller than 5 cm (stage Ib) or distant intra-abdominal/retroperitoneal at least 5 cm in size (stage II). Differently, according to WSES, the Ib stage indicates the presence of an abscess ≤ 4 cm, while the IIa stage indicates an abscess > 4 cm.
While for the uncomplicated forms of AD conservative treatment is established, and the treatment of the severe cases is surely surgical, the management of intra-abdominal DAs is still controversial. Conservative treatment is currently suggested in small abscesses (size ≤ 4 cm), while percutaneous drainage (PD) or surgery are suggested in larger abscesses [8,9,10]. Here the role of surgery is debated, being necessary to distinguish between elective and emergency surgery. Elective surgery is considered for patients after the resolution of acute episodes and consists of colic resection and primary anastomosis (PA). The indication is based on the quality of life, the number of acute episodes and the wellness intervals, the immunity status and the acceptance of possible complications [7]. On the contrary, the indication for emergency surgery is accepted in case of severe complications of AD, such as extensive abdominal collections, or entero-visceral fistulas, as well as after the failure of conservative treatment, such as antibiotics or percutaneous drainage. Emergency surgery is burdened by a high mortality rate, often due to co-morbidities or sepsis, rather than the technique itself.
The study aimed to assess the role of antibiotics and surgery in patients affected by DA and the effectiveness of different antibiotic regimens.
Methods
We retrospectively analysed a prospective database, including all consecutive patients with contrast-enhanced CT scan diagnosis of acute Hinchey Ib–II and WSES Ib–IIa diverticulitis of the left colon (from the splenic flexure to the rectum), admitted to our hospital between January 1st, 2013, and December 31st, 2020. Exclusion criteria were age < 18 years, co-presence of other acute diseases, and use of oral anticoagulant therapy. We considered a minimum follow-up period of 12 months after discharge from the hospital. The study was conducted in good clinical practice according to the Helsinki Declaration of 1975 and subsequent modifications. The information has been processed after checking the presence of informed consent to the processing of personal data for purpose of scientific research.
After CT scan diagnosis and staging, all patients were initially referred to conservative treatment. In our series there were no cases of generalized peritonitis; otherwise, such patients would have been referred for upfront surgery.
Conservative treatment consisted of intravenous (i.v.) administration of amoxicillin + clavulanic acid (2.2 g dose every 8 h per 6 days), or ciprofloxacin (400 mg dose every 12 h per 6 days) + metronidazole (500 mg dose every 8 h per 6 days), or piperacillin + tazobactam (4.5 g dose every 8 h per 5 days). The choice of the antibiotic regimen was based on the preference of the medical doctor responsible for the patient’s management. The antibiotic therapy was defined as successful when we observed a resolution of the acute infection, the regression of symptoms and the normalization of inflammatory indices. We considered the antibiotic therapy to fail when observing persistent symptoms, increased white blood cell (WBC) count and C-reactive protein (CRP) dosage after 48 h from the start of treatment. Failure of antibiotic therapy led to surgical treatment. This consisted of colic resection with PA with or without a protective stoma or Hartmann’s procedure (HP) according to the operator’s choice.
Per each patient, the following characteristics were considered: age, gender, body mass index (BMI), Charleston Comorbidity Index (CCI), smoking, family history of diverticulitis disease (DD), comorbidities, AD location, abscess location, abscess size (mm), white blood cells (WBC) count at admission, history of AD, previous surgery for AD.
Endpoints
The primary endpoint was to compare recurrence rates for conservative (antibiotics) and surgical therapy in patients affected by DA. The secondary endpoint was to assess the failure rate of each antibiotic regimen resulting in surgery.
Subgroups
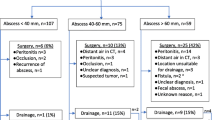
The series was divided into two groups according to WSES classification: patients with DA size ≤ 4 cm (group 1) and patients with DA size > 4 cm (group 2). Each group was further divided based on treatment: antibiotic therapy (sub-group 1A/2A) vs antibiotic + surgery (sub-group 1S/2S) (Fig. 1).
Statistical analysis
Data were analysed using descriptive and inferential statistics. Categorical variables were expressed as absolute/relative frequencies, while the continuous ones as mean (standard deviation) or median (range). Statistical analysis was conducted with a chi-square test for categorical variables. For continuous variables, Student’s t test and Mann–Whitney U test were used, as appropriate. The significance level was set at 5% for all variables. All statistics were performed with SPSS 22.0 (SPSS Inc., Chicago, IL).
Results
A hundred consecutive patients were included in the study. The mean age of the whole series was 61 years (± 15), with a little prevalence of women (51%). Median CCI at diagnosis was 2 (0–8). All patients’ characteristics are reported in Table 1. The most common comorbidity was cardiovascular disease (22%). In the majority of cases, AD was located in the sigmoid colon (94%), while the abscess was in most of the cases pericolic/mesocolic (68%).
Forty-three patients were included in group 1 (abscess ≤ 4 cm), while 57 were assigned to group 2 (abscess > 4 cm). In group 1, 31 patients (72.1%) were successfully treated with antibiotic therapy and 12 (27.9%) required surgery. In group 2, 29 (50.9%) cases received only antibiotic therapy and 28 (49.1%) underwent surgery after medical therapy failure during the same hospitalization.
Table 2 shows the comparison of the subgroups (antibiotic therapy A vs antibiotic + surgery S in each main group) for patients’ and disease characteristics. In group 1, the subgroups resulted homogeneous for all the variables considered except for “history of AD”, which was significantly more frequent both overall (n = 2 vs n = 16, p < 0.001) and in subgroup S (n = 1 vs n = 11, p < 0.001). All the 12 cases who underwent surgery were subjected to PA. In a single case, a protective loop ileostomy was performed. In group 2, subgroups A and S presented a significant difference in terms of median abscess size (55 mm vs 65 mm, p = 0.003). Groups 2A and 2S resulted homogeneous for all the other variables analyzed. Among the patients of subgroup 2S, 26 (92.9%) underwent PA (3 protected by loop ileostomy), while in the other 2 (7.1%) an HP was performed. Patients undergoing HP were > 70 years of age, both with a past history of AD, with abscess size > 50 mm and ASA IV. Also, at first observation they had body temperature > 38 °C and WBC > 15 × 109 cells. In the 1S subgroup laparotomy was performed in 2 (16.7%) patients, laparoscopy in 7 (58.3%) patients and laparoscopy converted into an open approach in 3 (25%) patients. In the subgroup 2S percentages were respectively 39.3% (11 patients), 42.9% (12 patients) and 17.8% (5 patients). Characteristics of patients who underwent surgery in the two groups are reported in Tables 3, 4.
No patient developed severe postoperative complications (Clavien–Dindo ≥ 3) and no patient required blood transfusions. Clavien–Dindo I events occurred in group 1, resolved by analgesic, antipyretic, antiemetic therapy alone. In group 2 the Clavien–Dindo II events were mainly related to ileus, in all cases resolved through the use of prokinetic drugs and/or placement of a nasogastric tube. The mean length of hospital stay was 8 days (± 4) for group 1 and 10 days (± 4) for group 2 (p = 0.033). A single patient required readmission for a phlegmon of the abdominal wall, successfully treated with i.v. antibiotic therapy. Stoma reversal was performed within 6 months from initial surgery in all cases and no surgical revision was necessary after primary surgery. None of the 6 patients with a stoma experienced complications during their subsequent hospitalization for stoma reversal surgery. The 3 patients, who experienced recurrence surgically treated with PA, did not develop post-operative complications.
With a minimum follow-up of 12 months (mean 36 months), 10 recurrences (16.7%) were observed in patients initially treated with antibiotic therapy only, none after resective surgery. Four recurrences occurred in group 1 (all patients previously treated with antibiotic therapy). Three of four patients required surgery and underwent PA, while in a single case conservative treatment was successfully retried. In group 2 we observed 6 recurrences, all in subgroup A. Three patients underwent PA, other 2 conservative treatments with i.v. antibiotic therapy, while in one case outpatient antibiotic therapy was successfully administered. Table 5 reports a comparison of the subgroups for recurrence rate.
The analysis of the association between the type of antibiotic and conservative therapy failure highlighted that in both groups all the patients referred to surgery had been previously treated with i.v. amoxicillin-clavulanic acid (Table 6).
Discussion and conclusions
DD is a frequent gastrointestinal disorder in Western Countries [11]. More than two hundred thousand people request hospital assistance every year for DD, impacting significantly on costs for the National Health System [12]. About 4% of these patients experience AD during their lifetime [13]. Their treatment is conservative in most cases, sometimes even handled as an outpatient [14, 15]. In complicated forms with purulent or faecal peritonitis, extensive bowel perforation or widespread abdominal collections, surgery is mandatory [7,8,9]. The preferred treatment for localized abdominal abscesses, found in about 25% of complicated ADs, is still debated [4]. Currently, recent guidelines recommend antibiotic therapy for abscesses up to 3 or 4 cm in diameter. When reachable, PD is recommended for those larger than 3 or 4 cm [7,8,9]. Nevertheless, little evidence is available to support this. Data regarding short and long-term outcomes of percutaneous treatments are heterogeneous and derive mainly from retrospective series. Many of these studies report no difference in success rate comparing PD vs antibiotic therapy [16, 17]. Even the assessment of the risk of recurrence is controversial. A systematic review has reported this to be overall higher in the antibiotic treatment group compared to PD, while for larger abscesses > 5 cm PD was associated with an increased risk of recurrence [18]. A recent large observational study reported that abscesses > 3 cm were associated with a higher short-term treatment failure compared to < 3 cm, while abscesses > 5 cm had an increased indication of surgery during short-term follow-up [19]. In addition, PD is not immune from complications, such as entero-cutaneous or entero-visceral fistulas, which sometimes require surgical treatment [20, 21]. Based on these controversial concerns, the practice of PD for DA never got into a routine at our Institution, although an extremely skilled interventional radiology department is available 24/7. Our policy is both due to the good results of resective surgery when antibiotics were not successful in controlling sepsis and for the uncertain management of each patient once the acute episode has been resolved.
In cases of complicated AD, Hartmann’s procedure is generally the most performed surgery, but a high rate of permanent colostomy is registered [22]. As an alternative, in selected cases laparoscopic lavage has been proposed [9]. The advantages and disadvantages of this technique have been extensively discussed in the literature, due to the conflicting results of the main trials SCANDIV, DILALA, LADIES and subsequent meta-analyses [23,24,25,26]. Due to the worse outcomes and the complications reported in some trials, guidelines suggest that laparoscopic lavage should not be considered as the first choice in diverticular peritonitis, being left as an option in selected cases [27,28,29,30,31].
Resection and PA with or without diverting stoma, initially performed as elective surgery, has been reconsidered in an emergency setting only in recent years. According to most recent guidelines, PA should represent the first therapeutic choice even in cases of severely complicated diverticulitis, such as in Hinchey III and IV disease, reserving HP and laparoscopic lavage only to high-risk patients with ASA scores ≥ 3 [32, 33]. Several randomized trials compared the outcomes of HP versus PA in this situation, finding no differences for in-hospital mortality or major complications rate (Clavien–Dindo IIIb–V). Diverting ileostomies, performed in some series in about one-third of the patients only, were more likely reversed than end colostomies [34, 35].
Similarly, even more we are convinced that for Hinchey Ib and IIa not responding to antibiotic therapy direct surgical resection with PA should be the preferred option. In a series of a hundred consecutive patients affected by DA, 40 not responding to antibiotic therapy underwent colonic resection. Short-term results showed a low incidence of complications and very few stomas required. The analysis of long-term data revealed that all stomas were reversed within a few months, with minimal impact on quality of life, together with a lower risk of recurrence within one year compared to conservative therapy.
In the past, surgery for AD was confined to an emergency setting due to the high perioperative morbidity and mortality. Nevertheless, these are more often dependent on clinical conditions, such as co-morbidities and sepsis, rather than the surgical technique itself. Currently, surgery is considered in patients with DAs Hinchey Ib and IIa after the failure of other treatments (antibiotics, percutaneous). We believe in these cases the role of surgery should be reconsidered as an effective one-way therapy with minimal sequelae. Hinchey Ib and IIa should be equated to symptomatic recurrent AD burdening quality of life. The present series shows that when peritonitis is confined and patients have a good performance status, PA experiences a very low rate of complications and need for temporary stomas with comparable results to elective delayed surgery. Indeed, percutaneous drainage may allow resolution of the acute episode and delay elective surgery in a referral centre. However, most peripheral centres are not equipped with interventional radiology even in daytime. Furthermore, percutaneous drainage would probably delay the timing of the operation, exposing the patient to a higher risk of complications, due both to the percutaneous treatment itself and to a prolonged infectious state.
So why not consider surgery as a treatment of choice, if, at the right timing, it can have good short- and long-term results? There is an increasing need for new contributions to this debate. Further data should contribute to evidence about the effectiveness of PA and PD for DA, supporting the definition of specific indications for both. Based on the aforementioned, an in-depth analysis of the role of different antibiotics regimens is also mandatory. Antibiotics should be effective against Grams—and anaerobes and oral use should be preferred over i.v. [9]. Although the choice of the molecule was depending on the operator’s preference, we observed that i.v. amoxicillin-clavulanic acid was significantly associated with failure of conservative therapy both in group 1 (p = 0.019) and in group 2 (p < 0.001). Furthermore, all patients who underwent surgery received amoxicillin-clavulanic acid initially. On the contrary, i.v. ciprofloxacin + metronidazole or piperacillin-tazobactam were associated with better infectious control compared to amoxicillin-clavulanic acid in group 2 (p = 0.023 and p = 0.004, respectively). When this would be confirmed in further studies, a more appropriate selection of antibiotics regimens may lead to a consistent reduction in the failure of sepsis control.
What also emerged from our work is that patients undergoing surgery were more likely to have a past history of acute uncomplicated diverticulitis. This is in contrast with previous studies [36], in which the history of diverticulitis is considered protective against the incidence of complicated diverticulitis. However, this result was not decisive in opting for surgery, which was instead adopted exclusively after the failure of antibiotic therapy.
Strengths of our study include a collection of a perspective database, sample homogeneity due to a reduced patient recruitment time and absence of missing data. Nonetheless, important limitations should be highlighted. Firstly, the monocentric retrospective nature of this work is the main source of bias. The sample consists of a small number of patients, although we believe it could be representative of the short recruitment time in our study. Finally, our study lacks antibiotic therapy management guidelines. Antibiotic regimen is left to the choice of each doctor at the time the patient enters the emergency room.
In conclusion, opting for resective surgery at the failure of antibiotic therapy in patients with DA, we observed a significant reduction in recurrence within 1 year, compared to antibiotic therapy, in patients with abscesses > 4 cm. Therefore, we suggest performing further studies comparing resective surgery with primary anastomosis to percutaneous drainage policies.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ambrosetti P (2012) Value of CT for acute left-colonic diverticulitis: the surgeon’s view. Dig Dis 30:51–55
Eglinton T, Nguyen T, Raniga S, Dixon L, Dobbs B, Frizelle FA (2010) Patterns of recurrence in patients with acute diverticulitis. Br J Surg 97(6):952–957
Trenti L, Kreisler E, Galvez A, Golda T, Frago R, Biondo S (2015) Long-term evolution of acute colonic diverticulitis after successful medical treatment. World J Surg 39(1):266–274
Devaraj B, Liu W, Tatum J, Cologne K, Kaiser AM (2016) Medically treated diverticular abscess associated with high risk of recurrence and disease complications. Dis Colon Rectum 59(3):208–215
Kaiser AM, Jiang JK, Lake JP, Ault G, Artinyan A, Gonzalez-Ruiz C, Essani R, Beart RW Jr (2005) The management of complicated diverticulitis and the role of computed tomography. Am J Gastroenterol 100(4):910–917
Hinchey EJ, Schaal PG, Richards GK (1978) Treatment of perforated diverticular disease of the colon. Adv Surg 12:85–109
Sartelli M, Catena F, Ansaloni L et al (2016) WSES guidelines for the management of acute left sided colonic diverticulitis in the emergency setting. World J Emerg Surg 11:37
Francis NK, Sylla P, Abou-Khalil M et al (2019) EAES and SAGES 2018 consensus conference on acute diverticulitis management: evidence-based recommendations for clinical practice. Surg Endosc 33(9):2726–2741
Schultz JK, Azhar N, Binda GA et al (2020) European society of coloproctology: guidelines for the management of diverticular disease of the colon. Colorectal Dis Suppl 2:5–28
Sartelli M, Weber DG, Kluger Y et al (2020) 2020 update of the WSES guidelines for the management of acute colonic diverticulitis in the emergency setting. World J Emerg Surg 15(1):32
Bugiantella W, Rondelli F, Longaroni M, Mariani E, Sanguinetti A, Avenia N (2015) Left colon acute diverticulitis: an update on diagnosis, treatment and prevention. Int J Surg 13:157–164
Peery AF, Crockett SD, Murphy CC et al (2019) Burden and cost of gastrointestinal, liver, and pancreatic diseases in the United States: update 2018. Gastroenterology 156(1):254-272.e11
Shahedi K, Fuller G, Bolus R, Cohen E, Vu M, Shah R, Agarwal N, Kaneshiro M, Atia M, Sheen V, Kurzbard N, van Oijen MG, Yen L, Hodgkins P, Erder MH, Spiegel B (2013) Long-term risk of acute diverticulitis among patients with incidental diverticulosis found during colonoscopy. Clin Gastroenterol Hepatol 11(12):1609–1613
Cirocchi R, Randolph JJ, Binda GA, Gioia S, Henry BM, Tomaszewski KA, Allegritti M, Arezzo A, Marzaioli R, Ruscelli P (2019) Is the outpatient management of acute diverticulitis safe and effective? A systematic review and meta-analysis. Tech Coloproctol 23(2):87–100
van Dijk ST, Bos K, de Boer MGJ, Draaisma WA, van Enst WA, Felt RJF, Klarenbeek BR, Otte JA, Puylaert JBCM, van Geloven AAW, Boermeester MA (2018) A systematic review and meta-analysis of outpatient treatment for acute diverticulitis. Int J Colorectal Dis 33(5):505–512
Mali J, Mentula P, Leppäniemi A, Sallinen V (2019) Determinants of treatment and outcomes of diverticular abscesses. World J Emerg Surg 14:31
Brandt D, Gervaz P, Durmishi Y, Platon A, Morel P, Poletti PA (2006) Percutaneous CT scan-guided drainage vs. antibiotherapy alone for Hinchey II diverticulitis: a case–control study. Dis Colon Rectum 49(10):1533–8
Gregersen R, Mortensen LQ, Burcharth J, Pommergaard HC, Rosenberg J (2016) Treatment of patients with acute colonic diverticulitis complicated by abscess formation: a systematic review. Int J Surg 35:201–208
Lambrichts DPV, Bolkenstein HE, van der Does DCHE, Dieleman D, Crolla RMPH, Dekker JWT, van Duijvendijk P, Gerhards MF, Nienhuijs SW, Menon AG, de Graaf EJR, Consten ECJ, Draaisma WA, Broeders IAMJ, Bemelman WA, Lange JF (2019) Multicentre study of non-surgical management of diverticulitis with abscess formation. Br J Surg 106(4):458–466
Singh B, May K, Coltart I, Moore NR, Cunningham C (2008) The long-term results of percutaneous drainage of diverticular abscess. Ann R Coll Surg Engl 90(4):297–301
Elagili F, Stocchi L, Ozuner G et al (2015) Antibiotics alone instead of percutaneous drainage as initial treatment of large diverticular abscess. Tech Coloproctol 19(2):97–103
Vermeulen J, Coene PP, Van Hout NM, van der Harst E, Gosselink MP, Mannaerts GH, Weidema WF, Lange JF (2009) Restoration of bowel continuity after surgery for acute perforated diverticulitis: should Hartmann’s procedure be considered a one-stage procedure? Colorectal Dis 11(6):619–624
Angenete E, Thornell A, Burcharth J, Pommergaard HC, Skullman S, Bisgaard T, Jess P, Läckberg Z, Matthiessen P, Heath J, Rosenberg J, Haglind E (2016) Laparoscopic lavage is feasible and safe for the treatment of perforated diverticulitis with purulent peritonitis: the first results from the randomized controlled trial DILALA. Ann Surg 263(1):117–122
Schultz JK, Yaqub S, Wallon C, Blecic L, Forsmo HM, Folkesson J, Buchwald P, Körner H, Dahl FA, Øresland T, SCANDIV Study Group (2015) Laparoscopic lavage vs primary resection for acute perforated diverticulitis: the SCANDIV randomized clinical trial. JAMA 314(13):1364–75
Vennix S, Musters GD, Mulder IM, Swank HA, Consten EC, Belgers EH, van Geloven AA, Gerhards MF, Govaert MJ, van Grevenstein WM, Hoofwijk AG, Kruyt PM, Nienhuijs SW, Boermeester MA, Vermeulen J, van Dieren S, Lange JF, Bemelman WA, Ladies trial colloborators, (2015) Laparoscopic peritoneal lavage or sigmoidectomy for perforated diverticulitis with purulent peritonitis: a multicentre, parallel-group, randomised, open-label trial. Lancet 386(10000):1269–1277
Cirocchi R, Di Saverio S, Weber DG, Taboła R, Abraha I, Randolph J, Arezzo A, Binda GA (2017) Laparoscopic lavage versus surgical resection for acute diverticulitis with generalised peritonitis: a systematic review and meta-analysis. Tech Coloproctol 21(2):93–110
Binda GA, Bonino MA, Siri G, Di Saverio S, Rossi G, Nascimbeni R, Sorrentino M, Arezzo A, Vettoretto N, Cirocchi R, LLO Study Group (2018) Multicentre international trial of laparoscopic lavage for Hinchey III acute diverticulitis (LLO Study). Br J Surg 105(13):1835–1843
Ceresoli M, Coccolini F, Montori G, Catena F, Sartelli M, Ansaloni L (2016) Laparoscopic lavage versus resection in perforated diverticulitis with purulent peritonitis: a meta-analysis of randomized controlled trials. World J Emerg Surg 11(1):42
Penna M, Markar SR, Mackenzie H, Hompes R, Cunningham C (2018) Laparoscopic lavage versus primary resection for acute perforated diverticulitis: review and meta-analysis. Ann Surg 267(2):252–258
Galbraith N, Carter JV, Netz U, Yang D, Fry DE, McCafferty M, Galandiuk S (2017) Laparoscopic lavage in the management of perforated diverticulitis: a contemporary meta-analysis. J Gastrointest Surg 21(9):1491–1499
Biffl WL, Moore FA, Moore EE (2019) The evolving role of laparoscopic lavage and drainage. J Trauma Acute Care Surg 86(2):376
Lambrichts DP, Edomskis PP, van der Bogt RD, Kleinrensink GJ, Bemelman WA, Lange JF (2020) Sigmoid resection with primary anastomosis versus the Hartmann’s procedure for perforated diverticulitis with purulent or fecal peritonitis: a systematic review and meta-analysis. Int J Colorectal Dis 35(8):1371–1386
Constantinides VA, Heriot A, Remzi F, Darzi A, Senapati A, Fazio VW, Tekkis PP (2007) Operative strategies for diverticular peritonitis: a decision analysis between primary resection and anastomosis versus Hartmann’s procedures. Ann Surg 245(1):94–103
Oberkofler CE, Rickenbacher A, Raptis DA, Lehmann K, Villiger P, Buchli C, Grieder F, Gelpke H, Decurtins M, Tempia-Caliera AA, Demartines N, Hahnloser D, Clavien PA, Breitenstein S (2012) A multicenter randomized clinical trial of primary anastomosis or Hartmann’s procedure for perforated left colonic diverticulitis with purulent or fecal peritonitis. Ann Surg 256(5):819–26
Bridoux V, Regimbeau JM, Ouaissi M, Mathonnet M, Mauvais F, Houivet E, Schwarz L, Mege D, Sielezneff I, Sabbagh C, Tuech JJ (2017) Hartmann’s procedure or primary anastomosis for generalized peritonitis due to perforated diverticulitis: a prospective multicenter randomized trial (DIVERTI). J Am Coll Surg 225(6):798–805
Binda GA, Amato A, Serventi A, Arezzo A (2012) Clinical presentation and risks. Dig Dis 30(1):100–107
Funding
Open access funding provided by Università degli Studi di Torino within the CRUI-CARE Agreement. No funding was necessary for this work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the realization of the study, each in one or more phases of the work elaboration process. Study conception and design: AA. Material preparation and data collection: FO, AF, GP, SV, GM. Analysis and interpretation of data: LDB, AA, AN. Drafting of manuscript: AA, AN, LDB, MM. Critical revision of manuscript: AA, MM. All authors commented on previous versions of the manuscript and read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Alberto Arezzo, Antonella Nicotera, Luca Domenico Bonomo, Francesco Olandese, Simona Veglia, Alice Ferguglia, Giuseppe Pentassuglia, Giuseppe Mingrone, Mario Morino have no conflict of interest.
Ethical and Informed Consent
The study was performed in compliance with ethical standards and informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arezzo, A., Nicotera, A., Bonomo, L.D. et al. Outcomes of surgical treatment of diverticular abscesses after failure of antibiotic therapy. Updates Surg 75, 855–862 (2023). https://doi.org/10.1007/s13304-023-01509-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-023-01509-4