Abstract
Introduction
To investigate the effects of glucose abnormality on outcomes of hospitalized coronavirus disease 2019 (COVID-19) patients in Japan.
Methods
This study retrospectively analyzed 393 COVID-19 patients admitted at Juntendo University Hospital. Patients were divided into subgroups according to history of diabetes and blood glucose (BG) levels and subsequently compared in terms of in-hospital death, invasive ventilation, or a composite of both.
Results
Patients with glucose abnormality demonstrated more risk factors for serious COVID-19, such as high body mass index, dyslipidemia, and hypertension, and higher biomarkers for inflammation compared to those with normal BG levels. Oxygen inhalation and steroid use were more frequent among patients with than without glucose abnormality. Invasive ventilation was more frequent in patients with diabetes (9.5% vs. 3.2%, p = 0.033) and BG ≥ 140 mg/dl (11.0% vs. 3.1%, p = 0.009) compared with those without diabetes and BG < 140 mg/dl, respectively. Logistic regression analysis showed that BG ≥ 140 mg/dl was a risk factor for invasive ventilation [odds ratio (OR) 2.87, 95% CI 1.04–7.68, p = 0.037] or the composite outcome (OR 3.03, 95% CI 1.21–7.38, p = 0.015) even after adjusting for by age and gender. Kaplan-Meier analysis showed that glucose abnormality was significantly associated with invasive ventilation and that BG ≥ 140 mg/dl was a risk factor for invasive ventilation [hazard ratio (HR) 2.68, 95% CI 1.05–6.82, p = 0.039] and the composite of death and invasive ventilation (HR 2.77, 95% CI 1.21–6.37, p = 0.016) regardless of age and gender.
Conclusions
Glucose abnormality, particularly BG ≥ 140 mg/dl, was associated with serious outcomes among Japanese COVID-19 patients, suggesting the need to consider high BG as a major risk factor for poor clinical course also in Japan.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The effects of glucose abnormality on COVID-19 patients in Japan remain unknown. |
393 patients with COVID-19 in Juntendo University Hospital were retrospectively analyzed. |
What was learned from the study? |
Patients with blood glucose ≥ 140 mg/dl were associated with in-hospital deterioration. |
Glucose abnormality was considered a risk factor for poor COVID-19 outcomes in Japan. |
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has ravaged the world for almost 2 years now, with the accumulated total number of deaths exceeding 5 million [1]. In Japan, cumulative numbers of patients diagnosed with COVID-19 has reached 1.5 million as of September 2021 [2]. Patients with risk factors, such as obesity [3], hypertension [4], and dyslipidemia [5], have been reported to experience serious COVID-19 complications, with glucose abnormality, including diabetes, also having been considered a major comorbidity associated with poor COVID-19 clinical course [6]. In the New York City area between March and April 2020, diabetes was one of the most common (33.8%) comorbidities in 5700 COVID-19 patients [7]. A retrospective study of critically ill patients with COVID-19 admitted to ICUs in Italy found that 68% of the subjects had as least one comorbidity, with diabetes also being a major one (17%) along with hypertension and dyslipidemia [8]. While COVID-19 subjects with type 2 diabetes required more intensive medical care and showed significantly higher mortality rates than those without diabetes, a report has also shown that well-controlled blood glucose (BG) levels were associated with low mortality rates compared to poorly controlled BG levels in Hubei, China, where the first cases of pneumonia of unknown origin had been reported [9]. The aforenoted reports suggested that diabetes plays an important role as a risk factor for poor outcomes in patients with COVID-19. However, only a few reports in Japan have examined the relationship between glucose abnormality and COVID-19 prevalence and prognosis. Considering the differences in prevalence of COVID-19 patients between Japan and other countries where governments are struggling to control the number of newly diagnosed patients, examining the impact of glucose abnormality on COVID-19 prognosis should provide a useful perspective on future treatments. From these perspectives, we retrospectively investigated the clinical records of in-hospital COVID-19 patients at Juntendo University Hospital, Tokyo, to clarify the effects of glucose abnormality on outcomes of hospitalized COVID-19 patients in Japan.
Methods
Patients
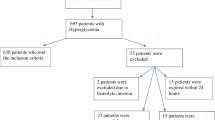
Our study protocol was in accordance with the principles outlined in the Declaration of Helsinki for the use of human tissue or subjects and was approved by the ethics committee of Juntendo University Hospital, a 1051-bed university-affiliated hospital in Tokyo (approval no. E21-0012-H01). We utilized the opt-out method to obtain consent for this study from 453 COVID-19-positive patients admitted at Juntendo University Hospital from July 2020 to June 2021. COVID-19 was diagnosed using chest computed tomography and reverse transcription-polymerase chain reaction (RT-PCR) based on the Clinical Management of Patients with COVID-19 (5th edition) published by the Ministry of Health, Labour and Welfare and the National Institute of Infectious Diseases [10]. We retrospectively extracted baseline characteristics, including medical history, laboratory findings, severity on admission, and in-hospital treatment, from the medical records. Baseline characteristics and laboratory data on admission were available for 393 patients without missing values (Fig. 1). COVID-19 severity was classified as mild (patients with SpO2 ≥ 96%, no respiratory symptoms), moderate I (patients with 93% < SpO2 < 96%, shortness of breath and pneumonia findings), moderate II (patients with SpO2 ≤ 93%, oxygen therapy required), and severe (admission to ICU or mechanical ventilation required) [10]. All patients received the standard protocol upon admission, including oxygen therapy, steroid therapy with dexamethasone, and pulse therapy with methylprednisolone. Favipiravir was administered following the recommended protocol as indicated in the Japanese Guideline [10] based on previous trials [11]. Only a limited number of patients received baricitinib after the Ministry of Health, Labour, and Welfare issued an emergency use authorization in April 2021. Patients received insulin therapy as needed due to steroid-induced hyperglycemia, with a target BG range of 140–180 mg/dl [12]. All eligible patients were categorized as having glucose abnormality based on history of diabetes or BG level ≥ 140 mg/dl on admission following the latest American Diabetes Association and American Association of Clinical Endocrinologists consensus [13] and definition of hyperglycemia for hospitalized patients [14] (Fig. 1).
Study Outcomes
The study outcomes were in-hospital death, invasive ventilation defined as mechanical ventilation, or the composite of death and invasive ventilation (Fig. 1). These outcomes were analyzed in previous reports to assess the severity of COVID-19 [15].
Statistical Analysis
Categorical variables were expressed as frequency rates and percentages (%), whereas continuous variables were expressed as mean (SD). Categorical variables between groups were compared using the Fisher exact test, whereas continuous variables were analyzed using the Student t test. Survival curves were plotted using the Kaplan-Meier method. Univariable analysis was performed to determine risk factors for the outcomes, and age- and gender-adjusted multivariable logistic regression analysis was performed focusing on BG, whereas age- and gender-adjusted multivariable Cox regression models were also used to investigate risk factors during hospitalization. Multivariable analysis was performed as exploratory because of the limited number of events, and power calculation was not performed considering that this research is exploratory. All statistical analyses were performed using the R Studio version 1.4.1717, with a p value of < 0.05 indicating statistical significance.
Results
Patient Characteristics on Admission
To examine the effects of glucose abnormality on COVID-19 prognosis, patients diagnosed with COVID-19 who were admitted at Juntendo University Hospital were categorized based on their history of diabetes and BG levels. Among the 393 patients without any missing data, 84 had a history of diabetes and 73 had a BG level ≥ 140 mg/dl (Table 1). Three patients had been diagnosed with type 1 diabetes, and four patients had been treated by steroid therapy because of other comorbidities on admission. As expected, subjects with history of diabetes and BG ≥ 140 mg/dl had similar risk factors, such as higher age, higher body mass index, and history of hypertension or dyslipidemia. Patients with mild symptoms upon admission had a more normal BG level compared to those with worse symptoms.
Laboratory Findings on Admission
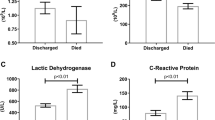
As shown in Table 2, patients with history of diabetes showed higher HbA1c [5.73 (0.44) vs. 7.64 (1.89), p < 0.001] and BG [106.1 (23.3) vs. 168.5 (72.7), p < 0.001] levels than those without a history of diabetes. Moreover, patients with history of diabetes [5.14 (2.03) vs. 5.91 (2.68), p = 0.005] as well as subjects with BG level ≥ 140 mg/dl [5.17 (1.91) vs. 5.91 (3.14), p = 0.009] demonstrated higher white blood cell counts than those without a history of diabetes or BG level < 140 mg/dl. Moreover, patients with diabetes [67.01 (12.43) vs. 72.18 (11.69), p = 0.001] as well as those with BG level ≥ 140 mg/dl [67.17 (12.34) vs. 72.22 (12.15), p = 0.002] had a higher neutrophil percentage than their counterparts. In contrast, the percentage of lymphocytes was lower in patients with diabetes [23.76 (10.66) vs. 19.79 (9.37), p = 0.002], as well as in subjects with BG levels ≥ 140 mg/dl [23.64 (10.54) vs. 19.71 (9.85), p = 0.004], than in their counterparts. Serum creatinine levels were higher in subjects with than without a history of diabetes [0.93 (1.28) vs. 2.11 (3.49), p < 0.001], whereas they were comparable between patients with increased and decreased BG levels. In contrast, no significant difference in aspartate aminotransferase (AST) levels was observed between patients with and without diabetes [35.64 (27.42) vs. 34.76 (21.46), p = 0.786] and BG level greater and less than 140 mg/dl [34.33 (26.11) vs. 40.36 (26.41), p = 0.077]. C-reactive protein was higher in subjects with diabetes [3.12 (4.18) vs. 5.36 (6.02), p < 0.001] and BG level ≥ 140 mg/dl [3.28 (4.48) vs. 5.02 (5.43), p = 0.004], whereas those with BG level ≥ 140 mg/dl had higher levels of ferritin [544.71 (642.09) vs. 758.34 (585.03), p = 0.01] and KL-6 [279.28 (207.14) vs. 371.45 (316.52), p = 0.002] than their counterparts.
In-Hospital Patient Treatment
All patients received the standard COVID-19 protocol, including oxygen therapy, steroid therapy, and antiviral treatment. As shown in Table 3, oxygen therapy was more frequently performed in patients with diabetes [106 (34.3) vs. 44 (52.4), p = 0.003] and BG ≥ 140 mg/dl [109 (34.1) vs. 41 (56.2), p = 0.001] compared to their counterparts. Steroid therapy, including pulse therapy, was also more prevalent in subjects with diabetes [142 (46.0) vs. 51 (60.7), p = 0.019] and BG ≥ 140 mg/dl [144 (45.0) vs. 49 (67.1), p = 0.001] compared to their counterparts. In contrast, treatment with antiviral drugs, such as remdesivir, baricitinib, and favipiravir, was comparable between all subgroups.
Clinical Outcomes
Next, we retrospectively analyzed the patient outcomes. As shown in Table 4, mortality rates were comparable between those with and without a history of diabetes [8 (2.6) vs. 4 (4.8), odds ratio (OR) 1.87, 95% confidence interval (CI) 0.40–7.22, p = 0.294] and between those with BG greater and less than 140 mg/dl [7 (2.2) vs. 5 (6.8), OR 3.27 (95% CI 0.79–12.4), p = 0.052]. In contrast, patients with glucose abnormality received more intensive respiratory treatment given that invasive ventilation was more frequently performed in subjects with history of diabetes [10 (3.2) vs. 8 (9.5), OR 3.13 (95% CI 1.03–9.16), p = 0.033] and those with BG ≥ 140 mg/dl [10 (3.1) vs. 8 (11.0), OR 3.80 (1.25–11.2), p = 0.009] compared to their counterparts. Accordingly, the composite outcomes, death and invasive ventilation, were more prevalent among patients with BG ≥ 140 mg/dl [13 (4.1) vs. 10 (13.7), OR 3.73 (95% CI 1.40–9.69), p = 0.004] compared to those with BG < 140 mg/dl.
Relationship Between Glucose Abnormality and Clinical Outcomes
We then performed a retrospective analysis on various factors on admission to examine which parameter is important for predicting the prognosis of COVID-19 patients. Among the factors shown in Tables 1 and 2, higher mortality rates were observed among older patients and those with higher white blood cell counts, neutrophil percentages, blood urea nitrogen levels, C-reactive protein levels, Krebs von den Lungen 6 (KL-6), and BG levels, whereas univariable analysis showed that lower percentages of lymphocytes, albumin levels, total protein levels, red blood cell count, and hemoglobin were significantly correlated with mortality (Table 5). Invasive ventilation was significantly associated with advanced age; male gender; systolic blood pressure; history of diabetes; chronic obstructive pulmonary disease; atherosclerotic cardiovascular disease (ASCVD); and higher white blood cell counts, neutrophil percentages, C-reactive protein, γ-glutamyltransferase (γGTP), ferritin, AST, alanine transaminase (ALT), BG, HbA1c, and KL-6. Meanwhile, lower percentages of lymphocytes, albumin, and total protein were associated with invasive ventilation. Moreover, advanced age, ASCVD, and higher white blood cell counts, neutrophil percentages, C-reactive protein, γGTP, ferritin, BG, AST, KL-6, and blood urea nitrogen were relevant to the composite outcome. Furthermore, the composite outcome was significantly associated with lower lymphocyte percentages, albumin, and total protein. Given that BG ≥ 140 mg/dl was commonly detected as a significantly related factor for the outcomes, we further examined whether BG ≥ 140 mg/dl remained a risk factor for poor COVID-19 clinical course after adjusting for age and gender. Multivariable logistic regression analysis revealed that BG ≥ 140 mg/dl was an independent risk factor for invasive ventilation [OR 2.87 (1.04–7.68), p = 0.037] and the composite outcome of death and invasive ventilation [OR 3.03 (1.21–7.38), p = 0.015] even after adjusted for age and gender (Table 5).
Finally, we performed Kaplan-Meier analysis to investigate the rates of mortality and invasive respiratory treatment during in-hospital treatment (Fig. 2). Notably, mortality rates were comparable between patients with and without a history of diabetes (4/84 vs. 8/309, log-rank test p value = 0.8) and between those with BG greater and less than 140 mg/dl (5/73 vs. 7/320, log-rank test p value = 0.3). In contrast, invasive ventilation was more frequently performed in patients with diabetes (8/84 vs. 10/309, log-rank test p value = 0.02) and BG ≥ 140 mg/dl (8/73 vs. 10/320, log-rank test p value = 0.006) compared to their counterparts. As for the composite outcome of death or invasive ventilation, patients with BG ≥ 140 mg/dl manifested poorer outcomes (10/73 vs. 13/320, log-rank test p value = 0.004) compared to those with BG < 140 mg/dl. The aforementioned data also indicated that high BG on admission was also associated with poor COVID-19 clinical course during in-hospital treatment. Thus, we investigated whether high BG was an independent risk factor for poor COVID-19 clinical course. As shown in Table 6, Cox regression analysis adjusted by age and gender found that BG ≥ 140 mg/dl was significantly associated with invasive ventilation [HR 2.68 (1.05–6.82), p = 0.039] and its composite with death [HR 2.77 (1.21–6.37), p = 0.016].
Kaplan-Meier curves of the event-free probability in patients with COVID-19 for outcomes. Probabilities of death, invasive ventilation, and the composite of death and invasive ventilation were analyzed between subgroups based on glucose abnormality. p values evaluated by log-rank test are shown in each plot. The lower and upper bounds of the 95% confidence interval are also displayed. The probability of invasive ventilation was significantly higher among all subgroups with glucose abnormality
Discussion
This retrospective study of COVID-19 patients in Japan revealed that high BG on admission was significantly associated with in-hospital intensive respiratory treatment. In contrast, no significant differences in mortality rates were observed from the perspective of glucose abnormality determined through either a history of diabetes or BG ≥ 140 mg/dl. Our findings also demonstrated that BG ≥ 140 mg/dl was an important factor significantly associated with death, invasive ventilation, and their composite in common, and an independent risk factor for invasive ventilation and a composite of death and invasive ventilation even after adjusted for age and gender. Kaplan-Meier analysis also demonstrated that BG ≥ 140 mg/dl was significantly associated with frequent invasive ventilation and a composite of death and invasive ventilation during in-hospital treatment, with multivariable Cox regression analysis identifying BG ≥ 140 mg/dl as a risk factor even after adjusted for age and gender.
According to a systematic review and meta-analysis including > 14,000 patients and 18 studies in the early pandemic from January 2019 to April 2020, around 11.5% of COVID-19 patients had a history of diabetes [16]. However, it should be noted that most of the earlier reports were from China, where the outbreak began. In the USA, around 33% of the subjects hospitalized for COVID-19 had history of diabetes based on the US Centers for Disease Control and Prevention COVID-19-Associated Hospitalization Surveillance Network (COVID-NET) [17], suggesting that geographic and chronological effects had an impact on variations in the prevalence of COVID-19 patients with diabetes. In the current study, 21% of our patients had diabetes, which is reasonable considering the number of previously hospitalized COVID-19 patients.
Reports have also shown that other comorbidities, such as obesity, hypertension, and dyslipidemia, which are all significantly associated with glucose abnormality, were prevalent in COVID-19-patients. Our data showed that those comorbidities were significantly associated with a history of diabetes and BG ≥ 140 mg/dl, suggesting that the combination of these risk factors may contribute to poor outcomes among COVID-19 patients with glucose abnormality.
Notably, among patients with glucose abnormality, only a few had mild symptoms on admission, which may be, however, due to the bias that subjects with risk factors are preferentially admitted to the hospital.
Laboratory examination on admission revealed that data associated with inflammatory response, such as high white blood cell counts, C-reactive protein, and high percentage of neutrophils, were significantly increased among patients with glucose abnormality. In particular, factors implying existence of acute inflammation, such as C-reactive protein, ferritin, and KL-6, were all associated with BG ≥ 140 mg/dl, suggesting that the aforementioned parameters reflected severe inflammation in the lungs, resulting in deterioration of pneumonia and need for invasive ventilation.
From the perspective of treatment, more patients with glucose abnormality received oxygen therapy and required steroid therapy than those without glucose abnormality. The use of glucocorticoids, such as dexamethasone, has been recommended as a standard medical treatment for COVID-19 [18], and pulse therapy has been considered as an effective approach for acute respiratory distress syndrome [19], which was widely administered to prevent the deterioration of respiratory functions in COVID-19 patients with glucose abnormality admitted at our hospital. In contrast, the use of antiviral reagents was comparable between patients with and without glucose abnormality given that those were prescribed as standard therapy irrelevant of the presence of glucose abnormality.
Surprisingly, no significant difference in mortality was observed between those with and without glucose abnormality determined based on a history of diabetes and BG ≥ 140 m/dl. The overall mortality rates based on our analyzed data was 12/393 (3.0%), which was considerably lower than that (15.8%) reported in the USA from March through November 2020 [20]. The low mortality rates might increase the difficultly of detecting statistically significant differences between patients with and without glucose abnormality. In contrast, more patients with glucose abnormality, estimated using either history of diabetes or BG ≥ 140 mg/dl, received intensive respiratory therapy compared to those without glucose abnormality. It has been widely accepted that high BG is a remarkable predictor of poor outcomes in infectious diseases, such as pneumonia [21], with studies recommending that BG be controlled between 140 and 180 mg/dl in the intensive care unit avoiding hypoglycemia [12]. In addition, there was no significant difference in proportion of the patients with invasive ventilation between those with BG levels ≥ 180 mg/dl (4/34, 11.8%) and subjects with 140 ≤ BG < 180 mg/dl (4/39, 10.3%) in this study.
Finally, after searching for a factor predicting the prognosis of COVID-19 from our data, we found that high BG on admission was significantly associated with poorer outcomes of COVID-19. Interestingly, the association between BG level ≥ 140 mg/dl and invasive ventilation and the composite outcome of death and invasive ventilation remained significant even after adjusted for age and gender, suggesting that glucose abnormality was one of the determinant factors for poor in-hospital clinical course of COVID-19. However, we could not exclude the effects of certain confounding factors as the low event rates in our data allowed for the limited number of factors to be analyzed using multivariable analysis [22]. Furthermore, the same trend was confirmed in the Kaplan-Meier analysis followed by Cox regression analysis, indicating the significance of estimating BG and the hypothesis that BG control during the initial phase could have favorable effects on the prognosis of COVID-19.
The current study has some limitations worth noting. (1) This was a single-center, retrospective study. (2) Another limitation was the relatively short study duration, which assessed confinements within a year, reflecting the first–fourth waves in Japan [2]. By December 2021, Japan had experienced five waves of COVID-19 infection since the start of the pandemic. The spread of the Delta variant was observed in Osaka by March 2021 and then reported in Tokyo by June 2021. (3) We should have considered the effects of vaccination [23] and novel antiviral treatments, which could have been administered during the research period [24]. Vaccination with the Pfizer-BioNTech mRNA and Moderna COVID-19 (mRNA-1273) vaccines started on April 2021 in Japan.
Conclusions
In summary, the current study revealed that glucose abnormality on admission, determined based on BG level, was significantly associated with poor COVID-19 clinical course such as invasive ventilation in Japan. Considering that only a few reports are available for discussing the relationship between glucose abnormality and prognosis of COVID-19 in Japan, this research is of significance to some extent as it suggests that high BG level is an important predicting factor for in-hospital severity of COVID-19 during treatment, consistent with previous reports in other countries. A future research direction is a prospective intervention study to prove that controlling BG levels may help improve the prognosis of COVID-19.
References
World Health Organization. Coronavirus disease (COVID-19) pandemic. https://www.who.int/emergencies/diseases/novel-coronavirus-2019. Accessed 30 Sept 2021.
Office for COVID-19 and other emerging infectious disease control, cabinet secretariat, government of Japan. COVID-19 Information and Resources. https://corona.go.jp/en/. Accessed 30 Sept 2021.
Tartof SY, Qian L, Hong V, et al. Obesity and mortality among patients diagnosed with COVID-19: results from an integrated health care organization. Ann Intern Med. 2020;173:773–81.
Li B, Yang J, Zhao F, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. 2020;109:531–8.
Hariyanto TI, Kurniawan A. Dyslipidemia is associated with severe coronavirus disease 2019 (COVID-19) infection. Diabetes Metab Syndr. 2020;14:1463–5.
Feldman EL, Savelieff MG, Hayek SS, Pennathur S, Kretzler M, Pop-Busui R. COVID-19 and diabetes: a collision and collusion of two diseases. Diabetes. 2020;69:2549–65.
Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized With COVID-19 in the New York City Area. JAMA. 2020;323:2052–9.
Grasselli G, Zangrillo A, Zanella A, et al. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region. Italy JAMA. 2020;323:1574–81.
Zhu L, She Z-G, Cheng X, et al. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;31(6):1068-1077.e3.
Office for COVID-19 and other emerging infectious disease control. Clinical management of patients with COVID-19. A Guide for front-line healthcare workers. Version 5.0. https://www.mhlw.go.jp/content/000785119.pdf. Accessed 30 Sept 2021.
Manabe T, Kambayashi D, Akatsu H, Kudo K. Favipiravir for the treatment of patients with COVID-19: a systematic review and meta-analysis. BMC Infect Dis. 2021;21:489.
Finfer S, Chittock DR, NICE SUGER Study Investigators. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360:1283–97.
Moghissi ES, Korytkowski MT, DiNardo M, et al. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care. 2009;32(6):1119–31.
American Diabetes Association. 15. Diabetes care in the hospital: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(Suppl 1):S193–202.
Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. New Engl J Med. 2019;382:NEJMoa2002032.
Singh AK, Gillies CL, Singh R, et al. Prevalence of comorbidities and their association with mortality in patients with COVID-19: a systematic review and meta-analysis. Diabetes Obes Metab. 2020;22:1915–24.
Ko JY, Danielson ML, Town M, et al. Risk Factors for COVID-19-associated hospitalization: COVID-19-associated hospitalization surveillance network and behavioral risk factor surveillance system. Clin Infect Dis. 2020;72:ciaa1419.
World Health Organization. Therapeutics and COVID-19: living guideline. https://www.who.int/publications/i/item/WHO-2019-nCoV-therapeutics-2021.3. Accessed 30 Sept 2021.
Prescott HC, Rice TW. Corticosteroids in COVID-19 ARDS: evidence and hope during the pandemic. JAMA. 2020;324:1292–5.
Roth GA, Emmons-Bell S, Alger HM, et al. Trends in patient characteristics and COVID-19 in-hospital mortality in the United States during the COVID-19 Pandemic. Jama Netw Open. 2021;4:e218828.
Lepper PM, Ott S, Nüesch E, et al. Serum glucose levels for predicting death in patients admitted to hospital for community acquired pne. BMJ. 2012;344:e3397.
Hosmer DW, Lemeshow S, May S. Applied survival analysis. 2nd ed. Hoboken: Wiley; 2007.
Tregoning JS, Flight KE, Higham SL, Wang Z, Pierce BF. Progress of the COVID-19 vaccine effort: viruses, vaccines and variants versus efficacy, effectiveness and escape. Nat Rev Immunol. 2021;21:626–36.
Ledford H. COVID antiviral pills: what scientists still want to know. Nature. 2021;599:358–9.
Acknowledgements
Funding
The journal’s Rapid Service Fee was funded by Center for Therapeutic Innovations in Diabetes, Juntendo University School of Medicine.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Y.N. and T.M. designed the study. Y.N. and M.H. researched data. Y.N, M.H, and Y.M. contributed to the data analysis. T.M contributed to the statistical analysis. T.N. and H.W. contributed to the discussion. Y.N. wrote the manuscript. All the authors reviewed and edited the manuscript.
Medical Writing, Editorial, and Other Assistance
We thank Satoshi Kurihara for technical assistance. We appreciate all medical members of Juntendo University Hospital for their devotion to the care of the patients and insightful suggestions and discussions. English editorial assistance in the preparation of this article was provided by Enago, the editing brand of Crimson Interactive Pvt. Ltd under Advance Editing B2C.
Disclosures
Yuya Nishida, Tomoya Mita, Makoto Hiki, Yasushi Matsushita, Toshio Naito, and Hirotaka Watada declare that they have nothing to disclose.
Compliance with Ethics Guidelines
Our study protocol was in accordance with the Declaration of Helsinki for the use of human tissue or subjects and was approved by the ethics committee of Juntendo University Hospital (approval# E21-0012-H01). We utilized the opt-out method to obtain consent for this study.
Data Availability
The datasets analyzed during the current study are not publicly available due to confidentiality and privacy of personal data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Nishida, Y., Mita, T., Hiki, M. et al. Retrospective Study on the Effects of Glucose Abnormality on COVID-19 Outcomes in Japan. Diabetes Ther 13, 325–339 (2022). https://doi.org/10.1007/s13300-022-01206-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01206-2