Abstract
The ongoing global pandemic of the coronavirus disease 2019 (COVID-19) has placed a severe strain on the management of chronic conditions like diabetes. Optimal glycemic control is always important, but more so in the existing environment of COVID-19. In this context, timely insulinization to achieve optimal glycemic control assumes major significance. However, given the challenges associated with the pandemic like restrictions of movement and access to healthcare resources, a simple and easy way to initiate and optimize insulin therapy in people with uncontrolled diabetes is required. With this premise, a group of clinical experts comprising diabetologists and endocrinologists from India discussed the challenges and potential solutions for insulin initiation, titration, and optimization in type 2 diabetes mellitus (T2DM) during the COVID-19 pandemic and how basal insulin can be a good option in this situation owing to its unique set of advantages like lower risk of hypoglycemia, ease of training, need for less monitoring, better adherence, flexibility of using oral antidiabetic drugs, and improved quality of life compared to other insulin regimens. The panel agreed that the existing challenges should not be a reason to delay insulin initiation in people with uncontrolled T2DM and provided recommendations, which included potential solutions for initiating insulin in the absence or restriction of in-person consultations; the dose of insulin at initiation; the type of insulin preferred for simplified regimen and best practices for optimal titration to achieve glycemic targets during the pandemic. Practical and easily implementable tips for patients and involvement of stakeholders (caregivers and healthcare providers) to facilitate insulin acceptance were also outlined by the expert panel. Simplified and convenient insulin regimens like basal insulin analogues are advised during and following the pandemic in order to achieve glycemic control in people with uncontrolled T2DM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Optimal glycemic control is always important, but more so in the existing environment of COVID-19. In this context, timely insulinization to achieve optimal glycemic control assumes major significance. |
However, given the challenges associated with the pandemic like restrictions of movement and access to healthcare resources, a simple and easy way to initiate and optimize insulin therapy in people with uncontrolled diabetes is required. |
A group of clinical experts comprising diabetologists and endocrinologists from India discussed the challenges and potential solutions for insulin initiation, titration, and optimization in type 2 diabetes mellitus (T2DM) during the COVID-19 pandemic and how basal insulin can be a good option in this situation owing to its unique set of advantages. |
Simplified and convenient insulin regimens like basal insulin analogues are advised during and following the pandemic in order to achieve glycemic control in people with uncontrolled T2DM. |
Digital Features
This article is published with digital features to facilitate understanding of the article. You can access the digital features on the article’s associated Figshare page. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13227848.
Introduction
The emergence and rapid spread of the coronavirus disease 2019 (COVID-19) has disrupted healthcare delivery systems across the world. The condition, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), first emerged in late November 2019 and has now assumed pandemic proportions on a global scale [1]. Statistics from the World Health Organization as of August 20, 2020 show that over 21 million people worldwide have been affected with COVID-19 and that over 735,000 people have died with the disease. Data indicates higher mortality risk in people with COVID-19 who have co-morbidities such as diabetes [2].
The data from the pandemic is still evolving, but all evidence suggests that people with co-morbidities such as diabetes, cardiovascular disease, and hypertension are at a heightened risk of experiencing adverse outcomes due to COVID-19 [2].
Studies have shown that people with uncontrolled type 2 diabetes mellitus (T2DM) are at increased risk of severity of complications and mortality due to COVID-19 [3]. A predictive analysis based on pre-pandemic glycemic status in India has demonstrated that glycemic control and diabetes-related complications are likely to worsen with increased duration of lockdown. This may further increase the load on the already stretched healthcare system [4].
Therefore, achieving and maintaining adequate glycemic control during and following the pandemic situation is of utmost importance. Continuing to follow lifestyle modifications needs to be stressed upon by the clinicians during and after the pandemic. Despite its efficacy in achieving glycemic control, the initiation of insulin therapy is often delayed in people with T2DM in India [5]. It is anticipated that delaying insulin initiation is even more common during the COVID-19 pandemic because of the presence of overarching concerns during these times which include [6]:
-
Restricted movement
-
Reduced access to healthcare resources including medications and glucose test strips
-
Missed follow-ups including laboratory tests and visits to healthcare providers
-
Restricted time for interaction leading to lack of counselling and training
-
Challenges with remote monitoring of blood glucose levels
-
Lack of adequate troubleshooting including correction of injection technique and proper use of insulin devices
In view of these unprecedented challenges, a panel of experts comprising diabetologists and endocrinologists from India developed an opinion paper on the need for insulin initiation during and following the COVID-19 pandemic. The brief practical recommendations authored by this group are elucidated in this paper. The expert panel recommends that clinicians follow local government guidelines for management of T2DM including the protocol for insulin initiation during and after the COVID-19 pandemic.
Methods
The RAND/UCLA Appropriateness Method (RAM) was used to arrive at an agreement on various practical issues of insulinization during the COVID-19 pandemic. Developed at the Research and Development (RAND) Corporation/University of California Los Angeles (UCLA) in the 1980s, RAM combines available scientific evidence with collective judgement of experts. The RAM technique obtains agreement from an expert group through rating appropriateness of well-defined clinical situations, on the basis of the appropriateness scale. Agreement is rated as “appropriate”, “uncertain”, or “inappropriate” according to the situation being discussed [7]. On the basis of this technique, a questionnaire was developed to seek clinical insights from the expert panel on initiation, titration, and optimization of basal insulin analogue in times of the COVID-19 pandemic. The questionnaire was circulated prior to a virtual meeting and experts were requested to rate the responses as “appropriate”, “uncertain”, or “inappropriate”. Further, the experts discussed the responses to the questionnaire and provided their inputs during the virtual meeting. The points on which the experts reached agreement and considered the advantage of basal insulin analogue over other insulin regimens during these challenging times are listed here. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Review Results
Challenges and Potential Solutions for Insulin Initiation During the COVID-19 Pandemic
Delay in initiating insulin therapy has been a major challenge in real-world clinical practice. Timely initiation of insulin can help mitigate unwanted complications and should be adopted for treating uncontrolled T2DM in the pandemic. In fact, a recent opinion paper by Hirsch and Gaudiani recommends earlier initiation of basal insulin therapy in uncontrolled T2DM. The authors argue that the American Diabetes Association (ADA) 2020 guidelines for diabetes treatment offer little “guidance regarding how treatment might differ based on how much a patient’s hemoglobin A1c (HbA1c) is above the treatment target after metformin is started.” They advocate the use of basal insulin after metformin as a second-line therapy if HbA1c ≥ 9.0% [8].
The expert panel discussed and debated three key questions surrounding insulin initiation during the ongoing COVID-19 pandemic:
-
1.
What are the main challenges related to insulin initiation at present and anticipated in the months ahead?
-
2.
What are the solutions to address these challenges?
-
3.
What are the tips for insulin initiation for primary care physicians and specialists in these times?
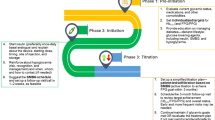
The summary of the expert opinion put forth by the panel for these questions is elucidated in Table 1 and Fig. 1.
Which Insulin is Recommended for Simplicity and Ease of Use at Initiation During the COVID-19 Pandemic?
Complex regimens, need for multiple injections, and side effects can hamper the person’s involvement and trust in insulin therapy. A simple and practical therapeutic approach helps empower the person, provides the context for targeting the goals in the management of diabetes mellitus, and boosts the confidence of the person [9]. This holds true in the current pandemic scenario as well.
The 2020 American Diabetes Association Standards of Care stress upon the fact that a basal insulin analogue is the most convenient insulin to initiate in T2DM [10]. A further advantage with basal insulin analogues like glargine 100 U/mL (Gla-100), detemir, glargine 300 U/mL (Gla-300), and degludec (IDeg) is that they involve only one injection per day and do not require adherence to inflexible meal patterns or quantity and composition of the diet [9]. It is also pertinent to note that newer second-generation basal insulin analogues like Gla-300 and IDeg have lower glycemic variability, greater flexibility in dosing, and a sustained glucose-lowering effect with lesser risk of hypoglycemia [11].
The expert panel discussed and debated three key questions surrounding the type of insulin to be used at initiation during the ongoing COVID-19 pandemic:
-
1.
Does basal insulin offer advantages for initiation during the pandemic?
-
2.
What dose should be used to initiate basal insulin and what is the preferred channel?
-
3.
When should the first follow-up be requested? What is the preferred channel?
The expert opinion put forth by the panel for these questions is summarized in Table 2 and Fig. 2.
What Are the Recommendations for Simple and Easy Self-monitoring of Blood Glucose and Insulin Titration During the COVID-19 Pandemic?
Self-monitoring of blood glucose (SMBG) constitutes one of the key tools in T2DM management in general and in insulin therapy in particular. This is because, once initiated, non-optimally titrated insulin leads to treatment dissatisfaction, discontinuation, and suboptimal glycemic control [11].
Safer and easier titration is facilitated by basal insulin compared with other insulin regimens, which works well with different titration algorithms [11]. The first head-to-head randomized controlled trial between the two second-generation basal insulins, Gla-300 and IDeg-100, in insulin-naïve people with T2DM has indicated lower risk of hypoglycemia during the titration period (initial 12 weeks) with Gla-300 [12].
Further, basal insulin analogues can effectively address glycemic variability by controlling the peaks and troughs of glucose levels. The peaks are addressed by targeting the fasting glucose which in turn minimizes postprandial excursions [13]. The troughs are addressed as there is lesser risk of hypoglycemia with basal insulin analogues as compared with Neutral Protamine Hagedorn (NPH) insulin and premix or co-formulations [14,15,16].
Individual preference and convenience are important parameters in initiating insulin during the pandemic. In this context, it must be noted that basal insulin analogues offer more flexible options than premix or co-formulations and are associated with increased treatment persistence and adherence [17, 18]. Research has also suggested that persons receiving once-daily dosing had approximately 44% more adherent days compared with those receiving twice-daily dosing [19]. The once-daily dosing offered by basal insulin analogues is also an attractive option for minimizing the risk of exposure to COVID-19 for healthcare providers and allied staff as has been pointed out by a recent study [20].
The expert panel discussed and debated the following key questions on SMBG and titration options during the ongoing COVID-19 pandemic:
-
1.
When should one start the conversation about titration?
-
2.
What is the minimum frequency of monitoring/SMBG?
-
3.
When should one start titration and what is the preferred channel?
-
4.
What is the preferred default titration algorithm?
-
5.
How long is the average “active” titration phase?
-
6.
What is the parameter to be monitored for titration?
-
7.
What are the points in the insulin journey where in-person consultation is important?
-
8.
What is the minimum follow-up frequency during the active titration phase?
-
9.
What are the profiles for patient-led titration?
-
10.
What is the maximum dose up to which basal insulin should be titrated?
-
11.
What changes should be recommended for the maintenance phase (post “active” titration) for monitoring, in-person visit requirement, etc.?
-
12.
What are the important points emphasized post “active” titration?
The expert opinion put forth by the panel for these questions is summarized in Table 3 and Fig. 3.
Conclusion
Routine care of diabetes has been adversely impacted during the COVID-19 pandemic in India. Increased stress levels, reduced physical activity, restrictions of movement, and limited access to healthcare associated with the pandemic could contribute to worsening outcomes in people with T2DM. The existing challenges should not be a reason to delay insulin initiation in people with uncontrolled T2DM. To overcome these challenges, simplified and convenient insulin regimens like basal insulin analogues are advised during and following the pandemic. The expert panel opined that basal insulin analogues may be preferred during and following the pandemic in order to achieve glycemic control owing to the unique set of advantages such as lower risk of hypoglycemia, ease of training, need for less monitoring, better adherence, flexibility of using oral antidiabetic drugs, and improved quality of life compared to other insulin regimens.
References
Coronavirus disease 2019 (COVID-19) Situation Report-94. World Health Organization. 2020. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200423-sitrep-94-covid-19.pdf?sfvrsn=b8304bf0_4. Accessed 5 Oct 2020.
Bornstein SR, Rubino F, Khunti K, et al. Practical recommendations for the management of diabetes in patients with COVID-19. Lancet Diabetes Endocrinol. 2020;8:546–50.
Hussain A, Bhowmik B, do Vale Moreira NC. COVID-19 and diabetes: knowledge in progress. Diabetes Res Clin Pract. 2020;162:108142.
Ghosal S, Sinha B, Majumder M, et al. Estimation of effects of nationwide lockdown for containing coronavirus infection on worsening of glycosylated haemoglobin and increase in diabetes-related complications: a simulation model using multivariate regression analysis. Diabetes Metab Syndr. 2020;14:319–23.
Kalra S, Bajaj S, Sharma SK, et al. A practitioner’s toolkit for insulin motivation in adults with type 1 and type 2 diabetes mellitus: evidence-based recommendations from an international expert panel. Diabetes Ther. 2020;11:585–606.
Managing diabetes during the COVID-19 pandemic. https://www.cebm.net/covid-19/managing-diabetes-during-the-covid-19-pandemic/. Accessed 6 July 2020.
The RAND/UCLA appropriateness method user’s manual. https://www.rand.org/content/dam/rand/pubs/monograph_reports/2011/MR1269.pdf. Accessed 6 July 2020.
Hirsch IB, Gaudiani LM. Using insulin to treat poorly controlled type 2 diabetes in 2020. JAMA. 2020. https://doi.org/10.1001/jama.2020.1303.
Ghosh S, Ghosh R. Glargine-300: an updated literature review on randomized controlled trials and real-world studies. World J Diabetes. 2020;11:100–14.
American Diabetes Association. Standards of medical care in diabetes—2020. https://care.diabetesjournals.org/content/43/Supplement_1/S98.full-text.pdf. Accessed 6 July 2020.
Jain SM, Seshadri K, Unnikrishnan AG, et al. Best practices and tools for titrating basal insulins: expert opinion from an Indian panel via the modified Delphi consensus method. Diabetes Ther. 2020;11:621–32.
Rosenstock J, Cheng A, Ritzel R, et al. More similarities than differences testing insulin glargine 300 units/mL versus insulin degludec 100 units/mL in insulin-naive type 2 diabetes: the randomized head-to-head BRIGHT trial. Diabetes Care. 2018;41(10):2147–54. https://doi.org/10.2337/dc18-0559.
Kalra S, Gupta Y, Unnikrishnan AG. Flexibility in insulin prescription. Indian J Endocrinol Metab. 2016;20:408–11.
Hanefeld M, Koehler C, Hoffmann C, et al. Effect of targeting normal fasting glucose levels with basal insulin glargine on glycaemic variability and risk of hypoglycaemia: a randomized, controlled study in patients with early type 2 diabetes. Diabet Med. 2010;27:175–80.
Janka HU, Plewe G, Riddle MC, et al. Comparison of basal insulin added to oral agents versus twice-daily premixed insulin as initial insulin therapy for type 2 diabetes. Diabetes Care. 2005;28:254–9. https://doi.org/10.2337/diacare.28.2.254.
Kumar S, Jang HC, Demirağ NG, et al. Efficacy and safety of once-daily insulin degludec/insulin aspart compared with once-daily insulin glargine in participants with type 2 diabetes: a randomized, treat-to-target study. Diabet Med. 2017;34:180–8. https://doi.org/10.1111/dme.13125.
Kumar A, Franek E, Wise J, et al. Efficacy and safety of once-daily insulin degludec/insulin aspart versus insulin glargine (U100) for 52 weeks in insulin-naïve patients with type 2 diabetes: a randomized controlled trial. PLoS One. 2016;11:e0163350.
Baser O, Tangirala K, Wei W, et al. Real-world outcomes of initiating insulin glargine-based treatment versus premixed analog insulins among US patients with type 2 diabetes failing oral antidiabetic drugs. Clinicoecon Outcomes Res. 2013;5:497–505.
Saini SD, Schoenfeld P, Kaulback K, et al. Effect of medication dosing frequency on adherence in chronic diseases. Am J Manag Care. 2009;15:e22-33.
Singh AK, Khunti K. Assessment of risk, severity, mortality, glycemic control and antidiabetic agents in patients with diabetes and COVID-19: a narrative review. Diabetes Res Clin Pract. 2020;165:108266.
Acknowledgements
All authors had full access to the articles reviewed in this manuscript, have read and reviewed the final draft of this manuscript and take complete responsibility for the integrity and accuracy of this manuscript. Before submission to the journal, Sanofi was given the opportunity to review the manuscript. However, the authors remain responsible for all content and editorial decisions. The content published herein solely represents the views and opinions of the authors and does not necessarily represent the views or opinion of Sanofi and/or its affiliates. The details published herein are intended for informational, educational, academic and/or research purposes and are not intended to substitute for professional medical advice, diagnosis or treatment. This expert opinion article is not intended to supplement, or supersede, the most up to date guidance related to the COVID-19 outbreak from international and local health authorities. No product mentioned in this expert opinion article is indicated for the prevention or treatment of COVID-19.
Funding
This expert opinion initiative is supported by Sanofi India. Medical writing and the journal’s rapid service fee were paid for by Sanofi India. The authors received no honoraria from Sanofi directly or indirectly related to the development of this publication.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Medical Writing and Editorial Assistance
We would like to thank Dr Rajshri Mallabadi from BioQuest Solutions Pvt. Ltd, Bangalore, for providing medical writing assistance and editorial support in the preparation of this manuscript, which was paid for by Sanofi, India.
Disclosures
Manoj Chawla, Sunil M Jain, Jothydev Kesavadev, Brij M Makkar, Vijay Viswanathan, Mangesh Tiwaskar, Aravind R Sosale, Vijay Negalur, Kirtikumar D Modi, Mukulesh Gupta, Surinder Kumar, Santosh Ramakrishnan, Nilakshi Deka and Nirmalya Roy have nothing to disclose.
Compliance with Ethical Guidance
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Chawla, M., Jain, S.M., Kesavadev, J. et al. Insulinization in T2DM with Basal Analogues During COVID-19 Pandemic: Expert Opinion from an Indian Panel. Diabetes Ther 12, 133–142 (2021). https://doi.org/10.1007/s13300-020-00979-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-020-00979-8