Abstract
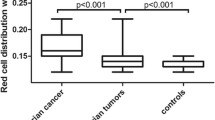
We assessed the diagnostic accuracy of a newly developed laboratory score—based on CA125, platelet count (PLT), C-reactive protein (CRP), and fibrinogen levels—in the preoperative diagnosis of adnexal mass. In this retrospective single-center study, we analyzed records of 142 patients with 54 malignant (38 %) and 88 benign (62 %) ovarian tumors. Preoperative levels of CA125, PLT, CRP, and fibrinogen were dichotomized according to the common cutoff values (CA125, 35 U/ml; PLT, 350/nl; CRP, 5.0 mg/l; fibrinogen, 400 mg/dl), resulting in “1” for results above the cutoff and “0” for results within the normal ranges. The values (1 or 0) were summarized to a “low” (0–2) or “high” (3–4) score. Its diagnostic accuracy was compared to the “gold standard,” CA125. All parameters differed significantly between malignant and benign cases. The score was false positive in 5/88 (5.7 %) and false negative in 13/54 (24 %) of cases. Conversely, CA125 was false positive in 18/88 (20.4 %) and false negative in 4/54 (7.4 %). The diagnostic accuracy of CA125 (>35 U/ml) was sensitivity 0.93, specificity 0.80, positive predictive value (PPV) 0.74, negative predictive value (NPV) 0.95, and positive likelihood ratio (weighted by prevalence) (+LH/p) 2.78. The diagnostic accuracy of the score was sensitivity 0.76, specificity 0.94, PPV 0.89, NPV 0.86, and +LH/p 8.2. In conclusion, the score is easy to use and generates no additional costs. It provides a better specificity, PPV, and +LH/p than CA125. The sensitivity and NPV are lower, but acceptable. A validation of the score in a large patient cohort is needed.
Similar content being viewed by others
References
Lowe KA, Chia VM, Taylor A, O'Malley C, Kelsh M, Mohamed M, et al. An international assessment of ovarian cancer incidence and mortality. Gynecol Oncol. 2013;130:107–14.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108.
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JW, Comber H, et al. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer. 2013;49:1374–403.
American College of Obstetricians and Gynecologists. ACOG practice bulletin. Management of adnexal masses. Obstet Gynecol. 2007;110:201–14.
Moore RG, Brown AK, Miller MC, Skates S, Allard WJ, Verch T, et al. The use of multiple novel tumor biomarkers for the detection of ovarian carcinoma in patients with a pelvic mass. Gynecol Oncol. 2008;108:402–8.
Radosa MP, Camara O, Vorwergk J, Diebolder H, Winzer H, Mothes A, et al. Preoperative multimodal strategies for risk assessment of adnexal masses: analysis of 1362 cases in a gynecologic cancer center. Int J Gynecol Cancer. 2011;21:1056–62.
Padilla LA, Radosevich DM, Milad MP. Accuracy of the pelvic examination in detecting adnexal masses. Obstet Gynecol. 2000;96:593–8.
Manegold-Brauer G, Bellin AK, Tercanli S, Lapaire O, Heinzelmann-Schwarz V. The special role of ultrasound for screening, staging and surveillance of malignant ovarian tumors: distinction from other methods of diagnostic imaging. Arch Gynecol Obstet. 2014;289:491–8.
Timmerman D, Testa AC, Bourne T, Ameye L, Jurkovic D, Van Holsbeke C, et al. Simple ultrasound-based rules for the diagnosis of ovarian cancer. Ultrasound Obstet Gynecol. 2008;31:681–90.
Medeiros LR, Rosa DD, da Rosa MI, Bozzetti MC. Accuracy of CA 125 in the diagnosis of ovarian tumors: a quantitative systematic review. Eur J Obstet Gynecol Reprod Biol. 2009;142:99–105.
Partheen K, Kristjansdottir B, Sundfeldt K. Evaluation of ovarian cancer biomarkers HE4 and CA-125 in women presenting with a suspicious cystic ovarian mass. J Gynecol Oncol. 2011;22:244–52.
Wang J, Gao J, Yao H, Wu Z, Wang M, Qi J. Diagnostic accuracy of serum HE4, CA125 and ROMA in patients with ovarian cancer: a meta-analysis. Tumour Biol. 2014;35:6127–38.
Ortiz-Muñoz B, Aznar-Oroval E, García García A, Covisa Peris A, Perez Ballestero P, Sanchez Yepes M, et al. HE4, Ca125 and ROMA algorithm for differential diagnosis between benign gynaecological diseases and ovarian cancer. Tumour Biol. 2014;35:7249–58.
Moss EL, Hollingworth J, Reynolds TM. The role of CA125 in clinical practice. J Clin Pathol. 2005;58:308–12.
He RH, Yao WM, Wu LY, Mao YY. Highly elevated serum CA-125 levels in patients with non-malignant gynecological diseases. Arch Gynecol Obstet. 2011;283 Suppl 1:107–10.
Macedo AC, da Rosa MI, Lumertz S, Medeiros LR. Accuracy of serum human epididymis protein 4 in ovarian cancer diagnosis: a systematic review and meta-analysis. Int J Gynecol Cancer. 2014;24:1222–31.
Grenache DG, Heichman KA, Werner TL, Vucetic Z. Clinical performance of two multi-marker blood tests for predicting malignancy in women with an adnexal mass. Clin Chim Acta. 2015;438:358–63.
Jacob F, Meier M, Caduff R, Goldstein D, Pochechueva T, Hacker N, et al. No benefit from combining HE4 and CA125 as ovarian tumor markers in a clinical setting. Gynecol Oncol. 2011;121:487–91.
Chalas E, Welshinger M, Engellener W, Chumas J, Barbieri R, Mann WJ. The clinical significance of thrombocytosis in women presenting with a pelvic mass. Am J Obstet Gynecol. 1992;166:974–7.
Friebe Z, Watrowski R, Bembnista M, Rokowska A, Włosiińska J, Dembińska M. Correlation between platelet count and CA-125 in ovarian cancer. Ginekol Pol. 2005;76:187–94. Polish.
Gungor T, Kanat-Pektas M, Sucak A, Mollamahmutoglu L. The role of thrombocytosis in prognostic evaluation of epithelial ovarian tumors. Arch Gynecol Obstet. 2009;279:53–6.
Stone RL, Nick AM, McNeish IA, Balkwill F, Han HD, Bottsford-Miller J, et al. Paraneoplastic thrombocytosis in ovarian cancer. N Engl J Med. 2012;366:610–8.
Man YN, Wang YN, Hao J, Liu X, Liu C, Zhu C, et al. Pretreatment plasma D-dimer, fibrinogen, and platelet levels significantly impact prognosis in patients with epithelial ovarian cancer independently of venous thromboembolism. Int J Gynecol Cancer. 2015;25:24–32.
Cho MS, Bottsford-Miller J, Vasquez HG, Stone R, Zand B, Kroll MH, et al. Platelets increase the proliferation of ovarian cancer cells. Blood. 2012;120:4869–72.
Lee M, Kim SW, Nam EJ, Yim GW, Kim S, Kim YT. The impact of pretreatment thrombocytosis and persistent thrombocytosis after adjuvant chemotherapy in patients with advanced epithelial ovarian cancer. Gynecol Oncol. 2011;122:238–41.
Allensworth SK, Langstraat CL, Martin JR, Lemens MA, McGree ME, Weaver AL, et al. Evaluating the prognostic significance of preoperative thrombocytosis in epithelial ovarian cancer. Gynecol Oncol. 2013;130:499–504.
Qiu J, Yu Y, Fu Y, Ye F, Xie X, Lu W. Preoperative plasma fibrinogen, platelet count and prognosis in epithelial ovarian cancer. J Obstet Gynaecol Res. 2012;38:651–7.
Hefler-Frischmuth K, Lafleur J, Hefler L, Polterauer S, Seebacher V, Reinthaller A, et al. Plasma fibrinogen levels in patients with benign and malignant ovarian tumors. Gynecol Oncol. 2015;136:567–70.
Simpson-Haidaris PJ, Rybarczyk B. Tumors and fibrinogen. The role of fibrinogen as an extracellular matrix protein. Ann N Y Acad Sci. 2001;936:406–25.
Polterauer S, Grimm C, Seebacher V, Concin N, Marth C, Tomovski C, et al. Plasma fibrinogen levels and prognosis in patients with ovarian cancer: a multicenter study. Oncologist. 2009;14:979–85.
Hefler-Frischmuth K, Hefler LA, Heinze G, Paseka V, Grimm C, Tempfer CB. Serum C-reactive protein in the differential diagnosis of ovarian masses. Eur J Obstet Gynecol Reprod Biol. 2009;147:65–8.
Hefler LA, Concin N, Hofstetter G, Marth C, Mustea A, Sehouli J, et al. Serum C-reactive protein as independent prognostic variable in patients with ovarian cancer. Clin Cancer Res. 2008;14:710–4.
Watrowski R. Simple laboratory score improves the pre-operative diagnosis of adnexal tumors. Arch Gynecol Obstet. 2012;286 Suppl 1:S105. German.
Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, et al. Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. Ann Intern Med. 2003;138:40–4.
Tavassoli FA, Devilee P, editors. World Health Organization classification of tumours: pathology and genetics of tumours of the breast and female genital organs. Lyon: IARC Press; 2003.
Pecorelli S, Benedet JL, Creasman WT, Shepherd JH. FIGO staging of gynecologic cancer. 1994-1997 FIGO Committee on Gynecologic Oncology. International Federation of Gynecology and Obstetrics. Int J Gynaecol Obstet. 1999;65:243–9.
Lowry R. VassarStats: Website for Statistical Computation. (http://vassarstats.net).
Donach M, Yu Y, Artioli G, Banna G, Feng W, Bast Jr RC, et al. Combined use of biomarkers for detection of ovarian cancer in high-risk women. Tumour Biol. 2010;31:209–15.
Partridge EE, Spencer EB. Assessing the risk of ovarian malignancy in asymptomatic women with abnormal findings: “tilting at windmills”. Gynecol Oncol. 2014;135:1–2.
Moore RG, Hawkins DM, Miller MC, Landrum LM, Gajewski W, Ball JJ, et al. Combining clinical assessment and the Risk of Ovarian Malignancy Algorithm for the prediction of ovarian cancer. Gynecol Oncol. 2014;135:547–51.
Acknowledgments
We thank Prof. Johannes Forster (University of Freiburg) for his assistance in preparing the study project. We thank Ms. Barbara Steffan and Christine Rauch (Central Laboratory of the St. Josefskrankenhaus, Freiburg) for providing the technical specifications of the laboratory methods.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Compliance with ethical standards
Approval for this study has been obtained from the Institutional Review Board of the University of Freiburg, Germany (Reference No. 192/12).
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Watrowski, R., Zeillinger, R. Simple laboratory score improves the preoperative diagnosis of adnexal mass. Tumor Biol. 37, 4343–4349 (2016). https://doi.org/10.1007/s13277-015-4280-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4280-7